Abstract
Background and Aim
Proximal colorectal cancer may arise from sessile serrated adenomas (SSAs). Recognition of these lesions during colonoscopy can optimize the endoscopic approach. We aimed to identify specific endoscopic features of SSA with conventional colonoscopy.
Methods
Patients undergoing screening colonoscopies from January 2011 to September 2012, in whom colonic polyps were found, were prospectively included in our study. Polyp morphology, location, polyp pit pattern (Kudo classification), and other previously reported features of SSA were evaluated. Histological examination was conducted independently by two pathologists. Multivariate analysis was performed to identify independent predictors of SSA.
Results
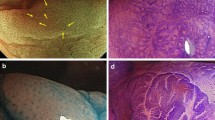
A total of 272 patients were included, and 440 polyps were evaluated (1.6 polyps per patient). Thirty-four polyps (8 %) were SSA, 135 (31 %) hyperplastic, and 249 (56 %) adenomas. The most prevalent endoscopic features of SSA were right-side location (94 %), type II Kudo pit pattern (91 %), mucus cap (41 %), flat morphology (29 %) and red-colored surface (26 %). Multivariate analysis revealed that flat morphology (p = 0.002, OR = 3.81 CI 1.53–9.09), red-colored surface (p < 0.001, OR = 12.97 CI 4.43–37.69), right-side location (p < 0.001, OR = 22.21 CI 5.09–135.94) and mucus cap (p < 0.001, OR 8.77 CI 3.76–20.44) were independent predictors of SSA.
Conclusion
We were able to identify specific features of SSA during conventional colonoscopy, which may help to identify, and therefore to optimize the endoscopic approach of these lesions.



Similar content being viewed by others
References
Leggett B, Whitehall V. Role of the serrated pathway in colorectal cancer pathogenesis. Gastroenterology. 2010;138:2088–2100.
Snover DC. Update on the serrated pathway to colorectal carcinoma. Hum Pathol. 2011;42:1–10.
Huang CS, Farraye FA, Yang S, O’Brien MJ. The clinical significance of serrated polyps. Am J Gastroenterol. 2011;106:229–240.
Vakiani E, Yantiss RK. Pathologic features and biologic importance of colorectal serrated polyps. Adv Anat Pathol. 2009;16:79–91.
Jass JR, Biden KG, Cummings MC, et al. Characterisation of a subtype of colorectal cancer combining features of the suppressor and mild mutator pathways. J Clin Pathol. 1999;52:455–460.
East JE, Saunders BP, Jass JR. Sporadic and syndromic hyperplastic polyps and serrated adenomas of the colon: classification, molecular genetics, natural history, and clinical management. Gastroenterol Clin North Am. 2008; 37:25–46, v.
Schreiner MA, Weiss DG, Lieberman DA. Proximal and large hyperplastic and nondysplastic serrated polyps detected by colonoscopy are associated with neoplasia. Gastroenterology. 2010;139:1497–1502.
Arain MA, Sawhney M, Sheikh S, et al. CIMP status of interval colon cancers: another piece to the puzzle. Am J Gastroenterol. 2010;105:1189.
Snover DC, Jass JR, Fenoglio-Preiser C, et al. Serrated polyps of the large intestine: a morphologic and molecular review of an evolving concept. Am J Clin Pathol. 2005;124:380–391.
Huang CS, Farraye FA, Yang S, et al. The clinical significance of serrated polyps. Am J Gastroenterol. 2011;106:229–240; quiz 241.
Baxter NN, Goldwasser MA, Paszat LF, et al. Association of colonoscopy and death from colorectal cancer. Ann Intern Med. 2009;150:1–8.
Singh H, Nugent Z, Demers AA, et al. The reduction in colorectal cancer mortality after colonoscopy varies by site of the cancer. Gastroenterology. 2010;139:1128–1137.
Brenner H, Haug U, Arndt V, et al. Low risk of colorectal cancer and advanced adenomas more than 10 years after negative colonoscopy. Gastroenterology. 2010;138:870–876.
Terdiman JP, McQuaid KR. Surveillance guidelines should be updated to recognize the importance of serrated polyps. Gastroenterology. 2010;139:1444–1447.
Hetzel JT, Huang CS, Coukos JA, et al. Variation in the detection of serrated polyps in an average risk colorectal cancer screening cohort. Am J Gastroenterol. 2010;105:2656–2664.
Kahi CJ, Hewett DG, Norton DL, et al. Prevalence and variable detection of proximal colon serrated polyps in screening colonoscopy examinations. Clin Gastroenterol Hepatol. 2011;9:42–46.
Chen SC, Rex DK. Variable detection of nonadenomatous polyps by individual endoscopists at colonoscopy and correlation with adenoma detection. J Clin Gastroenterol. 2008;42:704–707.
Adler A, Aschenbeck J, Yenerim T, et al. Narrow-band versus white-light high definition television endoscopic imaging for screening colonoscopy: a prospective randomized trial. Gastroenterology. 2009;136:410–416.e1; quiz 715.
Aminalai A, Rösch T, Aschenbeck J, et al. Live image processing does not increase adenoma detection rate during colonoscopy: a randomized comparison between FICE and conventional imaging (Berlin colonoscopy project 5, BECOP-5). Am J Gastroenterol. 2010;105:2383–2388.
Rex DK, Helbig CC. High yields of small and flat adenomas with high definition colonoscopes using either white light or narrow band imaging. Gastroenterology. 2007;133:42–47.
Soetikno RM, Kaltenbach T, Rouse RV, et al. Prevalence of nonpolypoid (flat and depressed) colorectal neoplasms in asymptomatic and symptomatic adults. JAMA. 2008;299:1027–1035.
Freedman JS, Harari DY, Bamji ND, et al. The detection of premalignant colon polyps during colonoscopy is stable throughout the workday. Gastrointest Endosc. 2011;73:1197–1206.
Participants in the Paris Workshop. The Paris endoscopic classification of superficial neoplastic lesions: esophagus, stomach, and colon: November 30 to December 1, 2002. Gastrointest Endosc. 2003; 58:S3–S43.
Endoscopic Classification Review Group. Update on the Paris classification of superficial neoplastic lesions in the digestive tract. Endoscopy. 2005;37:570–578.
Kudo S, Hirota S, Nakajima T, et al. Colorectal tumours and pit pattern. J Clin Pathol. 1994;47:880–885.
Gurudu SR, Heigh RI, De Petris G, et al. Sessile serrated adenomas: demographic, endoscopic and pathological characteristics. World J Gastroenterol. 2010;16:3402–3405.
Tadepalli US, Feihel D, Miller KM, et al. A morphologic analysis of sessile serrated polyps observed during routine colonoscopy. Gastrointest Endosc. 2011;74:1360–1368.
Yamada A, Notohara K, Aoyama I, et al. Endoscopic features of sessile serrated adenoma and other serrated colorectal polyps. Hepatogastroenterology. 2011;58:45–51.
Kashida H, Ikehara N, Hamatani S, et al. Endoscopic characteristics of colorectal serrated lesions. Hepatogastroenterology. 2011;58:1163–1167.
Jaramillo E, Tamura S, Mitomi H. Endoscopic appearance of serrated adenomas in the colon. Endoscopy. 2005;37:254–260.
Jaramillo E, Watanabe M, Rubio C, et al. Small colorectal serrated adenomas: endoscopic findings. Endoscopy. 1997;29:1–3.
Snover D, Ahnen D, Burt R, Odze RD. Serrated polyps of the colon and rectum and serrated polyposis. In: Bosman FT, Carneiro F, Hruban RH (eds). WHO Classification of Tumours of the Digestive System, 4th ed. Lyon: IARC; 2010.
Schlemper RJ, Riddell RH, Kato Y, et al. The Vienna classification of gastrointestinal epithelial neoplasia. Gut. 2000;47:251–255.
Lash R, Schuler C, Genta R. Demographic and pathologic evaluation of 2139 patients with sessile serrated adenomas in a one-year period. Am J Gastroenterol. 2008; 103:A191.
Farris AB, Misdraji J, Srivastava A, et al. Sessile serrated adenoma: challenging discrimination from other serrated colonic polyps. Am J Surg Pathol. 2008;32:30–35.
Wong NA, Hunt LP, Novelli MR, et al. Observer agreement in the diagnosis of serrated polyps of the large bowel. Histopathology. 2009;55:63–66.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pereyra, L., Gómez, E.J., González, R. et al. Finding Sessile Serrated Adenomas: Is It Possible to Identify Them During Conventional Colonoscopy?. Dig Dis Sci 59, 3021–3026 (2014). https://doi.org/10.1007/s10620-014-3295-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-014-3295-z