Abstract
Background
Several cross-sectional studies have shown an association between pre-S mutation and hepatocellular carcinoma (HCC).
Aims
We aim to verify whether pre-S mutation represents a risk for HCC development in a longitudinal way.
Methods
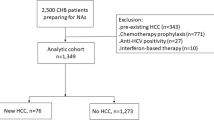
A total of 195 patients with chronic HBV infection [age: 43.7 ± 10.8 years, males: 141 (72.3 %), genotype C: 195 (100 %), hepatitis B e antigen (HBeAg) positive: 109 (55.9 %), cirrhosis: 79 (40.5 %), and pre-S mutation positive: 44 (22.6 %)] were followed up for a median of 7.2 years (range 1.0–7.8 years).
Results
HCC developed in 24 patients during follow-up. The 1-, 3-, and 5-year cumulative incidences of HCC were 0.5, 4.9, and 10.4 %, respectively. Patients with pre-S mutation had significantly higher 5-year cumulative incidences of HCC than those without (26.5 vs. 5.7 %, p < 0.001) and showed higher hazard ratio for HCC [3.04 (95 % CI 1.24–7.42), p = 0.015, adjusted for age, gender, HBeAg, cirrhosis and baseline HBV DNA level]. Notably, in patients aged ≥50 years, the 5-year cumulative incidences of HCC in patients with pre-S mutation were considerably high (58.3 %), compared to those without (16.1 %, p < 0.001).
Conclusions
Patients with pre-S mutations had higher incidence of HCC during follow-up, especially in aged patients. Patients with pre-S mutations, especially older ones, may require careful attention to HCC development.


Similar content being viewed by others
Abbreviations
- HBV:
-
Hepatitis B virus
- HBsAg:
-
Hepatitis B surface antigen
- HCC:
-
Hepatocellular carcinoma
- PCR:
-
Polymerase chain reaction
- AFP:
-
Alpha-fetoprotein
- ROC:
-
Receiver operator curve
- OR:
-
Odd ratios
- ER:
-
Endoplasmic reticulum
References
Tiollais P, Pourcel C, Dejean A. The hepatitis B virus. Nature. 1985;317:489–495.
Berthelot P, Neurath R, Courouce AM, et al. Hepatitis B vaccines with pre-S gene product. Lancet. 1986;1:1150.
Neurath AR, Adamowicz P, Kent SB, et al. Characterization of monoclonal antibodies specific for the pre-S2 region of the hepatitis B virus envelope protein. Mol Immunol. 1986;23:991–997.
Milich DR, McNamara MK, McLachlan A, Thornton GB, Chisari FV. Distinct H-2-linked regulation of T-cell responses to the pre-S and S regions of the same hepatitis B surface antigen polypeptide allows circumvention of nonresponsiveness to the S region. Proc Natl Acad Sci USA. 1985;82:8168–8172.
Milich DR, Thornton GB, Neurath AR, et al. Enhanced immunogenicity of the pre-S region of hepatitis B surface antigen. Science. 1985;228:1195–1199.
Hunt CM, McGill JM, Allen MI, Condreay LD. Clinical relevance of hepatitis B viral mutations. Hepatology. 2000;31:1037–1044.
Kao JH. Role of viral factors in the natural course and therapy of chronic hepatitis B. Hepatol Int. 2007;1:415–430.
Yamamoto K, Horikita M, Tsuda F, et al. Naturally occurring escape mutants of hepatitis B virus with various mutations in the S gene in carriers seropositive for antibody to hepatitis B surface antigen. J Virol. 1994;68:2671–2676.
Choi MS, Kim DY, Lee DH, et al. Clinical significance of pre-S mutations in patients with genotype C hepatitis B virus infection. J Viral Hepat. 2007;14:161–168.
Cao Z, Bai X, Guo X, Jin Y, Qian G, Tu H. High prevalence of hepatitis B virus pre-S mutation and its association with hepatocellular carcinoma in Qidong, China. Arch Virol. 2008;153:1807–1812.
Wang HC, Huang W, Lai MD, Su IJ. Hepatitis B virus pre-S mutants, endoplasmic reticulum stress and hepatocarcinogenesis. Cancer Sci. 2006;97:683–688.
Chen CF, Lee WC, Yang HI, et al. Changes in serum levels of HBV DNA and alanine aminotransferase determine risk for hepatocellular carcinoma. Gastroenterology. 2011;141:1240–1248.
Chen CH, Hung CH, Lee CM, et al. Pre-S deletion and complex mutations of hepatitis B virus related to advanced liver disease in HBeAg-negative patients. Gastroenterology. 2007;133:1466–1474.
Park JW. Practice guideline for diagnosis and treatment of hepatocellular carcinoma. Korean J Hepatol. 2004;10:88–98.
Qu LS, Liu TT, Jin F, et al. Combined pre-S deletion and core promoter mutations related to hepatocellular carcinoma: a nested case-control study in China. Hepatol Res. 2011;41:54–63.
Yeung P, Wong DK, Lai CL, Fung J, Seto WK, Yuen MF. Association of hepatitis B virus pre-S deletions with the development of hepatocellular carcinoma in chronic hepatitis B. J Infect Dis. 2011;203:646–654.
Chen CH, Changchien CS, Lee CM, et al. Combined mutations in pre-s/surface and core promoter/precore regions of hepatitis B virus increase the risk of hepatocellular carcinoma: a case-control study. J Infect Dis. 2008;198:1634–1642.
Chen BF, Liu CJ, Jow GM, Chen PJ, Kao JH, Chen DS. High prevalence and mapping of pre-S deletion in hepatitis B virus carriers with progressive liver diseases. Gastroenterology. 2006;130:1153–1168.
Lin CL, Liu CH, Chen W, et al. Association of pre-S deletion mutant of hepatitis B virus with risk of hepatocellular carcinoma. J Gastroenterol Hepatol. 2007;22:1098–1103.
Abe K, Thung SN, Wu HC, et al. Pre-S2 deletion mutants of hepatitis B virus could have an important role in hepatocarcinogenesis in Asian children. Cancer Sci. 2009;100:2249–2254.
Hildt E, Munz B, Saher G, Reifenberg K, Hofschneider PH. The PreS2 activator MHBs(t) of hepatitis B virus activates c-raf-1/Erk2 signaling in transgenic mice. EMBO J. 2002;21:525–535.
Hung JH, Su IJ, Lei HY, et al. Endoplasmic reticulum stress stimulates the expression of cyclooxygenase-2 through activation of NF-kappaB and pp 38 mitogen-activated protein kinase. J Biol Chem. 2004;279:46384–46392.
Wang HC, Chang WT, Chang WW, et al. Hepatitis B virus pre-S2 mutant upregulates cyclin A expression and induces nodular proliferation of hepatocytes. Hepatology. 2005;41:761–770.
Tai PC, Suk FM, Gerlich WH, Neurath AR, Shih C. Hypermodification and immune escape of an internally deleted middle-envelope (M) protein of frequent and predominant hepatitis B virus variants. Virology. 2002;292:44–58.
Wang HC, Wu HC, Chen CF, Fausto N, Lei HY, Su IJ. Different types of ground glass hepatocytes in chronic hepatitis B virus infection contain specific pre-S mutants that may induce endoplasmic reticulum stress. Am J Pathol. 2003;163:2441–2449.
Hsieh YH, Su IJ, Wang HC, et al. Pre-S mutant surface antigens in chronic hepatitis B virus infection induce oxidative stress and DNA damage. Carcinogenesis. 2004;25:2023–2032.
Benz CC, Yau C. Ageing, oxidative stress and cancer: paradigms in parallax. Nat Rev Cancer. 2008;8:875–879.
Bruix J, Sherman M. Management of hepatocellular carcinoma: an update. Hepatology. 2011;53:1020–1022.
Kao JH, Chen PJ, Lai MY, Chen DS. Basal core promoter mutations of hepatitis B virus increase the risk of hepatocellular carcinoma in hepatitis B carriers. Gastroenterology. 2003;124:327–334.
Huy TT, Ushijima H, Win KM, et al. High prevalence of hepatitis B virus pre-s mutant in countries where it is endemic and its relationship with genotype and chronicity. J Clin Microbiol. 2003;41:5449–5455.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sinn, D.H., Choi, M.S., Gwak, GY. et al. Pre-S Mutation Is a Significant Risk Factor for Hepatocellular Carcinoma Development: A Long-Term Retrospective Cohort Study. Dig Dis Sci 58, 751–758 (2013). https://doi.org/10.1007/s10620-012-2408-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-012-2408-9