Abstract
Background
Current treatments for the inflammatory bowel diseases, encompassing Crohn’s disease and ulcerative colitis, are variably effective. Emu oil, extracted from emu fat, predominantly comprises fatty acids, with purported claims of anti-inflammatory properties.
Aim
We evaluated emu oil for its potential to ameliorate dextran sulphate sodium (DSS)-induced colitis in rats.
Methods
Male Sprague–Dawley Rats were allocated to treatment groups (n = 8). Groups 1 and 2 consumed water and were gavaged (1 ml) daily with water (group 1) or emu oil (group 2) from days 0 to 10. Groups 3–6 ingested 2% DSS in the drinking water from days 5 to 10 and were gavaged from days 0 to 10 with water (group 3), 0.5 ml emu oil (group 4) or 1 ml emu oil (group 5). Group 6 received 1 ml emu oil after commencing DSS treatment (days 6–10). Disease activity index, metabolic parameters, 13C-sucrose breath test, and histological colonic damage severity and crypt depth were assessed.
Results
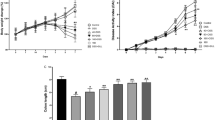
Emu oil in DSS-treated rats reduced colonic damage severity compared to DSS-controls (up to threefold; P < 0.001). In DSS-treated rats, crypts in the proximal colon were lengthened by 0.5 ml emu oil (373 ± 18 μm), compared with DSS-controls (302 ± 8 μm); whilst in the distal colon (DSS control: 271 ± 17 μm), crypt depth was greater following 0.5 ml emu oil (352 ± 22 μm) and 1 ml emu oil (341 ± 9 μm) and also when emu oil was administered post-DSS commencement (Group 6: 409 ± 16 μm; P < 0.05). Emu oil did not significantly affect other parameters of colonic architecture.
Conclusions
Emu oil improved tissue damage associated with colitis, suggesting its potential as a unique formulation to augment conventional treatment approaches for IBD.






Similar content being viewed by others
References
Hendrickson BA, Gokhale R, Cho JH. Clinical aspects and pathophysiology of inflammatory bowel disease. Clin Microbiol Rev. 2002;15:79–94.
Fiocchi C. Inflammatory bowel disease: etiology and pathogenesis. Gastroenterology. 1998;115:182–205.
Boismenu R, Chen Y. Insights from mouse models of colitis. J Leukoc Biol. 2000;67:267–278.
Podolsky DK. Inflammatory bowel disease. N Engl J Med. 2002;347:417–429.
Camuesco D, Galvez J, Nieto A, et al. Dietary olive oil supplemented with fish oil, rich in EPA and DHA (n-3) polyunsaturated fatty acids, attenuates colonic inflammation in rats with dss-induced colitis. J Nutr. 2005;135:687–694.
Tenikoff D, Murphy KJ, Le M, Howe PR, Howarth GS. Lyprinol (stabilised lipid extract of New Zealand green-lipped mussel): a potential preventative treatment modality for inflammatory bowel disease. J Gastroenterol. 2005;40:361–365.
Whitehouse MW, Turner AG, Davis CK, Roberts MS. Emu oil(s): a source of non-toxic transdermal anti-inflammatory agents in aboriginal medicine. Inflammopharmacol. 1998;6:1–8.
Snowden JM, Whitehouse MW. Anti-inflammatory activity of emu oils in rats. Inflammopharmacol. 1997;5:127–132.
Beckerbauer LM, Thiel-Cooper R, Ahn DU, Sell JL, Parrish FC Jr, Beitz DC. Influence of two dietary fats on the composition of emu oil and meat. Poult Sci. 2001;80:187–194.
Brown MA, Craig-Schmidt MC, Smith PC. Fatty acid composition of emu. Inform (Silver Spring, Md). 1995;6:470.
Howarth GS, Lindsay RJ, Butler RN, Geier MS. Can emu oil ameliorate inflammatory disorders affecting the gastrointestinal system? Aust J Exp Agric. 2008;48:1276–1279.
Yoganathan S, Nicolosi R, Wilson T, et al. Antagonism of croton oil inflammation by topical emu oil in cd-1 mice. Lipids. 2003;38:603–607.
James MJ, Gibson RA, Cleland LG. Dietary polyunsaturated fatty acids and inflammatory mediator production. Am J Clin Nutr. 2000;71:343S–348S.
Calder PC. N-3 polyunsaturated fatty acids, inflammation, and inflammatory diseases. Am J Clin Nutr. 2006;83:1505S–1519S.
Lopez A, Sims DE, Ablett RF, et al. Effect of emu oil on auricular inflammation induced with croton oil in mice. Am J Vet Res. 1999;60:1558–1561.
Qiu XW, Wang JH, Fang XW, Gong ZY, Li ZQ, Yi ZH. Anti-inflammatory activity and healing-promoting effects of topical application of emu oil on wound in scalded rats. Di Yi Jun Yi Da Xue Xue Bao. 2005;25:407–410.
Lindsay RJ, Geier MS, Yazbeck R, Butler RN, Howarth GS. Orally administered emu oil decreases acute inflammation and alters selected small intestinal parameters in a rat model of mucositis. Br J Nutr. 2010;104:513–519.
Geier MS, Butler RN, Giffard PM, Howarth GS. Lactobacillus fermentum BR11, a potential new probiotic, alleviates symptoms of colitis induced by dextran sulfate sodium (DSS) in rats. Int J Food Microbiol. 2007;114:267–274.
Tomas FM, Knowles SE, Owens PC, et al. Effects of full-length and truncated insulin-like growth factor-i on nitrogen balance and muscle protein metabolism in nitrogen-restricted rats. J Endocrinol. 1991;128:97–105.
Portolesi R, Powell BC, Gibson RA. Competition between 24:5n–3 and ala for delta 6 desaturase may limit the accumulation of DHA in HepG2 cell membranes. J Lipid Res. 2007;48:1592–1598.
Murthy SN, Cooper HS, Shim H, Shah RS, Ibrahim SA, Sedergran DJ. Treatment of dextran sulfate sodium-induced murine colitis by intracolonic cyclosporin. Dig Dis Sci. 1993;38:1722–1734.
Howarth GS, Xian CJ, Read LC. Predisposition to colonic dysplasia is unaffected by continuous administration of insulin-like growth factor-i for twenty weeks in a rat model of chronic inflammatory bowel disease. Growth Factors. 2000;18:119–133.
Pelton NC, Tivey D, Howarth GS, Davidson GP, Butler RN. A novel breath test for the non-invasive assessment of small intestinal mucosal injury following methotrexate administration in the rat. Scand J Gastroenterol. 2004;39:1015–1016.
Tooley KL, Howarth GS, Butler RN. Mucositis and non-invasive markers of small intestinal function. Cancer Biol Ther. 2009;8:753–758.
Yazbeck R, Howarth GS, Geier MS, Demuth HU, Abbott CA. Inhibiting dipeptidyl peptidase activity partially ameliorates colitis in mice. Front Biosci. 2008;13:6850–6858.
Vowinkel T, Kalogeris T, Mori M, Krieglstein CF, Granger DN. Impact of dextran sulfate sodium load on the severity of inflammation in experimental colitis. Dig Dis Sci. 2004;49:556–564.
Gaudio E, Taddei G, Vetuschi A, et al. Dextran sulfate sodium (DSS) colitis in rats: Clinical, structural, and ultrastructural aspects. Dig Dis Sci. 1999;44:1458–1475.
Drozdowski L, Thomson AB. Intestinal mucosal adaptation. World J Gastroenterol. 2006;12:4614–4627.
Deschner EE, Maskens AP. Significance of the labeling index and labeling distribution as kinetic parameters in colorectal mucosa of cancer patients and DMH treated animals. Cancer. 1982;50:1136–1141.
Roediger WE. The colonic epithelium in ulcerative colitis: an energy-deficiency disease? Lancet. 1980;2:712–715.
Breuer RI, Buto SK, Christ ML, et al. Rectal irrigation with short-chain fatty acids for distal ulcerative colitis. Preliminary report. Dig Dis Sci. 1991;36:185–187.
Harig JM, Soergel KH, Komorowski RA, Wood CM. Treatment of diversion colitis with short-chain-fatty acid irrigation. N Engl J Med. 1989;320:23–28.
Camoglio L, Te Velde AA, Tigges AJ, Das PK, Van Deventer SJ. Altered expression of interferon-gamma and interleukin-4 in inflammatory bowel disease. Inflamm Bowel Dis. 1998;4:285–290.
Egger B, Bajaj-Elliott M, MacDonald TT, Inglin R, Eysselein VE, Buchler MW. Characterisation of acute murine dextran sodium sulphate colitis: Cytokine profile and dose dependency. Digestion. 2000;62:240–248.
Acknowledgments
The authors would like to thank Kerry Lymn for a substantial contribution towards planning and conducting the animal trials, Esther Burt for Isotope Ratio Mass Spectrometry analyses and Chris Gregory and Geoff Lean from Emu Tracks Pty Ltd, Marleston, South Australia for their technical advice. S.M.A. conducted all animal trials and data analyses and prepared the manuscript. R.J.L. assisted with animal trials and data analyses. R. N. B. and A.G.C. assisted with analysis of data. G. S. H. contributed to the experimental design, analysis of data and manuscript preparation. Suzanne Mashtoub Abimosleh was granted The Queen Elizabeth Hospital Research Foundation Scholarship. Professor Gordon Howarth is supported by the Sally Birch Cancer Council Australia Research Fellowship in Cancer Control. This project was funded in part by Rural Industries Research and Development Corporation. No other conflicts of interest or financial agreements are in place.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Abimosleh, S.M., Lindsay, R.J., Butler, R.N. et al. Emu Oil Increases Colonic Crypt Depth in a Rat Model of Ulcerative Colitis. Dig Dis Sci 57, 887–896 (2012). https://doi.org/10.1007/s10620-011-1979-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-011-1979-1