Abstract
Background
CA19-9 is a tumor marker for pancreatic cancer, cholangiocarcinoma, and other malignancies. However, its sensitivity and specificity is suboptimal in clinical practice, which we hypothesized limits its clinical utility.
Aims
To evaluate the clinical utility and limitations of CA19-9 as a tumor marker.
Methods
We performed a retrospective review of CA19-9 levels (U/ml) in 483 consecutive patients between 2006 and 2008 at two university hospitals. We abstracted clinical, radiographic, and pathological data and final diagnoses. Descriptive and non-parametric analyses were performed.
Results
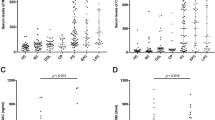
Patients presenting with jaundice had the highest CA19-9 (420) compared to other complaints (<20) (p < 0.01). The indications with the highest CA19-9 had evidence of biliary obstruction (71), liver mass (54), and pancreatic head mass (27) compared to other indications (<15) (p < 0.01). The diagnoses with the highest CA19-9 (p < 0.01) were cholangiocarcinoma (476), pancreatic cancer (161), and choledocholithiasis (138). Using a receiver operator curve to evaluate CA19-9, the area under the curve was 0.7 when evaluating all patients for pancreatic cancer or cholangiocarcinoma or patients with pancreatic head mass for pancreatic cancer.
Conclusions
This study found that for pancreatic cancer and cholangiocarcinoma, CA19-9 had poor clinical utility as a tumor marker and did not change patient management. Elevations in CA19-9 were associated with biliary obstruction based on clinical history, laboratory data, and diagnoses.

Similar content being viewed by others
References
Koprowski H, Steplewski Z, Mitchell K, et al. Colorectal carcinoma antigen detected by hybridoma antibodies. Somatic Cell Genet. 1979;5:957–972.
Itkowitz SH, Kim YS. New carbohydrate tumor markers. Gastroenterology. 1986;90:491–494.
Tempero MA, Uchida E, Takasaki H, et al. Relationship of carbohydrate antigen and Lewis antigens in pancreatic cancer. Cancer Res. 1987;47:5501–5503.
Goonetilleke KS, Siriwardena AK. Systematic review of carbohydrate antigen (CA19-9) as a biochemical marker in the diagnosis of pancreatic cancer. Eur J Surg Oncol. 2007;33:266–270.
Steinberg W. The clinical utility of the CA19-9 tumor-associated antigen. Am J Gastroenterol. 1990;85:350–355.
Andriulli A, Gindro T, Plantino P, et al. Prospective evaluation of the diagnostic efficacy of CA19-9 assay as a marker for gastrointestinal cancers. Digestion. 1986;33:26–33.
Uygur-Bayramicli O, Dabak R, Orbay E, et al. Type 2 diabetes mellitus and CA19-9 levels. World J Gastroenterol. 2007;13:5537–5539.
Mertz HR, Sechopoulos P, Delbeke D, et al. EUS, PET, and CT scanning for evaluation of pancreatic adenocarcinoma. Gastrointest Endosc. 2000;52:367–371.
Sreenarasimhaiah J. Efficacy of endoscopic ultrasound in characterizing mass lesions in chronic pancreatitis. J Clin Gastroenterol. 2008;42:81–85.
Mishra G, Conway JD. Endoscopic ultrasound in the evaluation of radiologic abnormalities of the liver and biliary tree. Curr Gastroenterol Rep. 2009;11:150–154.
Mann DV, Edwards R, Ho S, et al. Elevated tumour marker CA19-9: clinical interpretation and influence of obstructive jaundice. Eur J Surg Oncol. 2000;26:474–479.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Singh, S., Tang, Sj., Sreenarasimhaiah, J. et al. The Clinical Utility and Limitations of Serum Carbohydrate Antigen (CA19-9) as a Diagnostic Tool for Pancreatic Cancer and Cholangiocarcinoma. Dig Dis Sci 56, 2491–2496 (2011). https://doi.org/10.1007/s10620-011-1709-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-011-1709-8