Abstract
Background
The mechanisms involved in hepatocellular carcinoma (HCC) establishing an immunologically tolerogenic tumor environment remain poorly characterized.
Aims
This study evaluates effector T cell responses and soluble IL-2 receptor alpha chains (sCD25) in relation to HCC stage/survival and characterizes the impact of sCD25 on effectors.
Methods
Effector cell responses with serum from HCC patients and in serum free conditions were assessed by IFN-γ ELISpot, proliferation and ATP production assays at baseline, after depletion of sCD25, and after supplementation with recombinant sCD25. Sera sCD25 were measured by ELISA and any relationship with stage/survival was determined.
Results
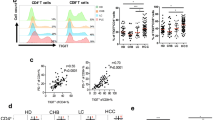
Hepatocellular carcinoma patients had marked global impairment in T cell responses at baseline which correlate with tumor burden and poor outcome. The impairment in immune responses is characterized by low IFN-γ production, cell proliferation, and ATP production. Effector responses are impaired by serum from HCC patients in a dose-dependent manner, implicating soluble factors in the observed immunosuppression. Significant elevations in serum levels of sCD25 are found in patients with HCC, which correlate with tumor burden and a worse survival. T cell reactivity is inversely proportional to serum level of sCD25. Impaired T cell responses improve with sCD25 depletion from HCC serum or IL-2 supplementation suggesting impairment in IL-2 signaling. In contrast, adding increasing doses of sCD25 suppresses effector T cells, which partly involves induction of apoptosis.
Conclusions
These findings show that HCC patients have blunted T cell immunity that is partly related to elevated levels of sCD25, supporting a novel immuno-inhibitory role for this soluble receptor.






Similar content being viewed by others
References
Parkin DM, Bray F, Ferlay J, et al. Estimating the world cancer burden: Globocan 2000. Int J Cancer. 2001;94:153–156.
El-Serag HB, Davila JA, Petersen NJ, McGlynn KA. The continuing increase in the incidence of hepatocellular carcinoma in the United States: an update. Ann Intern Med. 2003;139:817–823.
Fattovich G, Giustina G, Degos F, et al. Morbidity and mortality in compensated cirrhosis type C: a retrospective follow-up study of 384 patients. Gastroenterology. 1997;112:463–472.
Llovet JM, Burroughs A, Bruix J. Hepatocellular carcinoma. Lancet. 2003;362:1907–1917.
Llovet JM, Ricci S, Mazzaferro V, et al. Sorafenib in advanced hepatocellular carcinoma. N Engl J Med. 2008;359:378–390.
Newell P, Villanueva A, Llovet JM. Molecular targeted therapies in hepatocellular carcinoma: from pre-clinical models to clinical trials. J Hepatol. 2008;49:1–5.
Kalos M. Tumor antigen-specific T cells and cancer immunotherapy: current issues and future prospects. Vaccine. 2003;21:781–786.
Wada Y, Nakashima O, Kutami R, Yamamoto O, Kojiro M. Clinicopathological study on hepatocellular carcinoma with lymphocytic infiltration. Hepatology. 1998;27:407–414.
Cao M, Cabrera R, Xu Y, et al. Hepatocellular carcinoma cell supernatants increase expansion and function of CD4+CD25+ regulatory T cells. Lab Invest. 2007;87:582–590.
Ormandy LA, Hillemann T, Wedemeyer H, et al. Increased populations of regulatory T cells in peripheral blood of patients with hepatocellular carcinoma. Cancer Res. 2005;65(6):2457–2464.
Unitt E, Rushbrook SM, Marshall A, et al. Compromised lymphocytes infiltrate hepatocellular carcinoma: the role of T-regulatory cells. Hepatology. 2005;41:722–730.
Fu J, Xu D, Liu Z, et al. Increased regulatory T cells correlate with CD8 T-cell impairment and poor survival in hepatocellular carcinoma patients. Gastroenterology. 2007;132:2328–2339.
Bruix J, Sherman M. Practice guidelines committee, American association for the study of liver diseases. Management of hepatocellular carcinoma. Hepatology. 2005;42:1208–1236.
Nelson DR, Tu Z, Soldevila-Pico C, et al. Long-term interleukin 10 therapy in chronic hepatitis C patients has a proviral and anti-inflammatory effect. Hepatology. 2003;38:859–868.
Cabrera R, Ararat M, Soldevila-Pico C, et al. Using an immune functional assay to differentiate acute cellular rejection from recurrent hepatitis C in liver transplant patients. Liver Transpl. 2009;15:216–222.
Hoechst B, Ormandy LA, Ballmaier M, et al. A new population of myeloid-derived suppressor cells in hepatocellular carcinoma patients induces CD4+CD25+Foxp3+ T cells. Gastroenterology. 2008;135:234–243.
Murakami S. Soluble IL-2 receptor in cancer. Front Biosci. 2004;1(9):3085–3090.
Maier LM, Anderson DE, Severson CA, et al. Soluble IL-2RA levels in multiple sclerosis subjects and the effect of soluble IL-2RA on immune responses. J Immunol. 2009;182:1541–1547.
Chopra RK, Powers DC, Kendig NE, Adler WH, Nagel JE. Soluble interleukin 2 receptors released from mitogen stimulated human peripheral blood lymphocytes bind interleukin 2 and inhibit IL2 dependent cell proliferation. Immunol Invest. 1989;18:961–973.
Rubin LA, Jay G, Nelson DL. The released interleukin 2 receptor binds interleukin 2 efficiently. J Immunol. 1986;137(12):3841–3844.
Levine SJ. Mechanisms of soluble cytokine receptor generation. J Immunol. 2004;173:5343–5348.
Becker C, Fantini MC, Schramm C, et al. TGF-beta suppresses tumor progression in colon cancer by inhibition of IL-6 trans-signaling. Immunity. 2004;21:491–501.
Giannitrapani L, Cervello M, Soresi M, et al. Circulating IL-6 and sIL-6R in patients with hepatocellular carcinoma. Ann N Y Acad Sci. 2002;963:46–52.
Bien E, Balcerska A. Serum soluble IL-2 receptor alpha in human cancer of adults and children: a review. Biomarkers. 2008;13:1–26.
von Bergwelt-Baildon MS, Popov A, Saric T, et al. CD25 and indoleamine 2, 3-dioxygenase are up-regulated by prostaglandin E2 and expressed by tumor-associated dendritic cells in vivo: additional mechanisms of T-cell inhibition. Blood. 2006;108:228–237.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cabrera, R., Ararat, M., Cao, M. et al. Hepatocellular Carcinoma Immunopathogenesis: Clinical Evidence for Global T Cell Defects and an Immunomodulatory Role for Soluble CD25 (sCD25). Dig Dis Sci 55, 484–495 (2010). https://doi.org/10.1007/s10620-009-0955-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-009-0955-5