Abstract
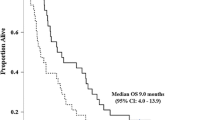
There is no standard chemotherapy option for patients with biliary tract cancers. These patients present fairly ill and can have a rapid progression of disease. We conducted a multi-center, phase-II trial for patients with locally unresectable or metastatic bile duct or gallbladder adenocarcinomas using a modified regimen of gemcitabine and cisplatin to potentially improve tolerability. Patients received a 21-day treatment cycle of gemcitabine at 1,000 mg/m2 and cisplatin at 30 mg/m2 on days 1 and 8. To participate, 33 patients signed informed consent, and 30 patients received at least one dose of chemotherapy. By intention-to-treat analyses, 7 patients (21%) experienced a partial response and another 12 (36%) had stable disease for at least 12 weeks. The median progression-free survival was 6.3 months and median overall survival was 9.7 months. After 1 year, 39% of patients were alive. Most common grade 3–4 toxicities included neutropenia (33%), thrombocytopenia (23%), anemia (20%), nausea (20%), emesis (13%) and fatigue (10%). Of note, 52% of patients withdrew from study treatment, principally due to treatment-related adverse events. We concluded that this modified regimen appeared to have comparable activity to other gemcitabine and cisplatin regimens against advanced bile duct and gallbladder cancers, but there was still moderate toxicity in this patient population.


Similar content being viewed by others
References
Jemal A, Siegel R, Ward E et al (2006) Cancer statistics, 2006. CA Cancer J Clin 56:106–130
Burris HA 3rd, Moore MJ, Andersen J et al (1997) Improvements in survival and clinical benefit with gemcitabine as first-line therapy for patients with advanced pancreas cancer: a randomized trial. J Clin Oncol 15:2403–2413
Eng C, Ramanathan RK, Wong MK et al (2004) A phase II trial of fixed dose rate gemcitabine in patients with advanced biliary tree carcinoma. Am J Clin Oncol 27:565–569
Gebbia V, Giuliani F, Maiello E et al (2001) Treatment of inoperable and/or metastatic biliary tree carcinomas with single-agent gemcitabine or in combination with levofolinic acid and infusional fluorouracil: results of a multicenter phase II study. J Clin Oncol 19:4089–4091
Kubicka S, Rudolph KL, Tietze MK et al (2001) Phase II study of systemic gemcitabine chemotherapy for advanced unresectable hepatobiliary carcinomas. Hepatogastroenterology 48:783–789
Lin MH, Chen JS, Chen HH et al (2003) A phase II trial of gemcitabine in the treatment of advanced bile duct and periampullary carcinomas. Chemotherapy 49:154–158
Okusaka T, Ishii H, Funakoshi A et al (2006) Phase II study of single-agent gemcitabine in patients with advanced biliary tract cancer. Cancer Chemother Pharmacol 57:647–653
Park JS, Oh SY, Kim SH et al (2005) Single-agent gemcitabine in the treatment of advanced biliary tract cancers: a phase II study. Jpn J Clin Oncol 35:68–73
Penz M, Kornek GV, Raderer M et al (2001) Phase II trial of two-weekly gemcitabine in patients with advanced biliary tract cancer. Ann Oncol 12:183–186
Tsavaris N, Kosmas C, Gouveris P et al (2004) Weekly gemcitabine for the treatment of biliary tract and gallbladder cancer. Invest New Drugs 22:193–198
von Delius S, Lersch C, Schulte-Frohlinde E et al (2005) Phase II trial of weekly 24-hour infusion of gemcitabine in patients with advanced gallbladder and biliary tract carcinoma. BMC Cancer 5:61
Alberts SR, Al-Khatib H, Mahoney MR et al (2005) Gemcitabine, 5-fluorouracil, and leucovorin in advanced biliary tract and gallbladder carcinoma: a North Central Cancer Treatment Group phase II trial. Cancer 103:111–118
Andre T, Tournigand C, Rosmorduc O et al (2004) Gemcitabine combined with oxaliplatin (GEMOX) in advanced biliary tract adenocarcinoma: a GERCOR study. Ann Oncol 15:1339–1343
Bhargava P, Jani CR, Savarese DM et al (2003) Gemcitabine and irinotecan in locally advanced or metastatic biliary cancer: preliminary report. Oncology (Williston Park) 17:23–26
Cho JY, Paik YH, Chang YS et al (2005) Capecitabine combined with gemcitabine (CapGem) as first-line treatment in patients with advanced/metastatic biliary tract carcinoma. Cancer 104:2753–2758
Doval DC, Sekhon JS, Gupta SK et al (2004) A phase II study of gemcitabine and cisplatin in chemotherapy-naive, unresectable gall bladder cancer. Br J Cancer 90:1516–1520
Hsu C, Shen YC, Yang CH et al (2004) Weekly gemcitabine plus 24-h infusion of high-dose 5-fluorouracil/leucovorin for locally advanced or metastatic carcinoma of the biliary tract. Br J Cancer 90:1715–1719
Kim ST, Park JO, Lee J et al (2006) A phase II study of gemcitabine and cisplatin in advanced biliary tract cancer. Cancer 106:1339–1346
Knox JJ, Hedley D, Oza A et al (2005) Combining gemcitabine and capecitabine in patients with advanced biliary cancer: a phase II trial. J Clin Oncol 23:2332–2338
Knox JJ, Hedley D, Oza A et al (2004) Gemcitabine concurrent with continuous infusional 5-fluorouracil in advanced biliary cancers: a review of the Princess Margaret Hospital experience. Ann Oncol 15:770–774
Kornek GV, Schuell B, Laengle F et al (2004) Mitomycin C in combination with capecitabine or biweekly high-dose gemcitabine in patients with advanced biliary tract cancer: a randomised phase II trial. Ann Oncol 15:478–483
Kuhn R, Hribaschek A, Eichelmann K et al (2002) Outpatient therapy with gemcitabine and docetaxel for gallbladder, biliary, and cholangio-carcinomas. Invest New Drugs 20:351–356
Thongprasert S, Napapan S, Charoentum C et al (2005) Phase II study of gemcitabine and cisplatin as first-line chemotherapy in inoperable biliary tract carcinoma. Ann Oncol 16:279–281
Valle W, Wasan H, Johnson P et al (2006) Gemcitabine, alone or in combination with cisplatin, in patients with advanced or metastatic cholangiocarcinoma (CC) and other biliary tract tumors: a multicenter, randomized, phase II (the UK ABC-01) study, Gastrointestinal Cancer Symposium, pp Abstr 98
Bergman AM, Ruiz van Haperen VW, Veerman G et al (1996) Synergistic interaction between cisplatin and gemcitabine in vitro. Clin Cancer Res 2:521–530
Peters GJ, Bergman AM, Ruiz van Haperen VW et al (1995) Interaction between cisplatin and gemcitabine in vitro and in vivo. Semin Oncol 22:72–79
Therasse P, Arbuck SG, Eisenhauer EA et al (2000) New guidelines to evaluate the response to treatment in solid tumors. European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. J Natl Cancer Inst 92:205–216
Rocha Lima CM, Flores AM (2006) Gemcitabine doublets in advanced pancreatic cancer: should we move on? J Clin Oncol 24:327–329
Heinemann V, Labianca R, Hinke A et al (2006) Superiority of gemcitabine plus platinum analog compared to gemcitabine alone in advanced pancreatic cancer: pooled analysis of two randomized trials, the GERCOR/GISCAD Intergroup Study and a German Multicenter Study, Gastrointestinal Cancer Symposium, pp Abstr 96
Acknowledgment
Supported by Lilly Pharmaceuticals.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Meyerhardt, J.A., Zhu, A.X., Stuart, K. et al. Phase-II Study of Gemcitabine and Cisplatin in Patients with Metastatic Biliary and Gallbladder Cancer. Dig Dis Sci 53, 564–570 (2008). https://doi.org/10.1007/s10620-007-9885-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-007-9885-2