Abstract
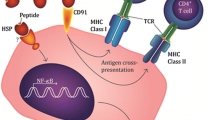
We performed a phase I pilot study to determine if autologous vaccine HSPPC-96 (gp96, Oncophage®) could be purified from completely resected pancreas adenocarcinomas, to determine patient tolerance of vaccine and to explore immune responses and clinical outcomes of these patients. Subjects were vaccinated with 5 μg of autologous HSPPC-96 weekly for 4 doses. Serial ELISPOT assays of T cells for antitumor reactivity were performed. Subjects received neither adjuvant chemotherapy nor radiation. Ten patients received a full course of vaccinations. No dose-limiting toxicities were encountered. Immediate freezing in liquid nitrogen of the tumor specimen resulted in improved vaccine yield. Median overall survival is 2.2 years (Kaplan–Meier estimate). Autologous anti-HSPPC-96 ELISPOT reactivity increased significantly in 1 of 5 patients examined and a second had an increase of unclear significance. Three of 10 treated patients are alive without disease at 2.6, 2.7, and 5.0 years follow-up. There was no observed correlation between immune response and prognosis. This study demonstrates the feasibility of preparing HSPPC-96 from pancreatic adenocarcinomas. Examination of this novel approach using multiple dose levels is 1 approach to further investigate the immunogenicity and clinical utility of HSPPC-96 vaccination in this setting.



Similar content being viewed by others
References
Jemal A, Tiwari RC, Taylor-Murray EW, Ward E, Samuels A, Tiwari RC, Ghafoor A, Feuer EJ, Thun MJ (2005) Cancer statistics, 2005. CA Cancer J Clin 55:10–30
Geer RJ, Brennan MF (1993) Prognostic indicators for survival after resection of pancreatic adenocarcinoma. Am J Surg 165:68–72
Begg CB, Cramer LD, Hoskins WJ, Brennan MF (1998) Impact of hospital volume on operative mortality for major cancer surgery. JAMA 280:1747–1751
Callery MP, Strasberg SM, Doherty GM, Soper J, Norton JA (1997) Staging laparoscopy with laparoscopic ultrasonography: optimizing resectability in hepatobiliary and pancreatic malignancy. J Am Coll Surg 185:33–39
Brooks AD, Mallis MJ, Brennan MF, Conlon KC (2002) The value of laparoscopy in the management of ampullary, duodenal, and distal bile duct tumors. J Gastrointest Surg 6:139–145
Gold EB, Goldin SB (1998) Epidemiology of and risk factors for pancreatic cancer. Surg Oncol Clin North Am 7:67–91
Evans DB, Abbruzzese JL, Willett CG (2001) Cancer of the pancreas. In: DeVita VT, Hellman S, Rosenberg SA (eds) Cancer: Principles and practice of oncology (6th edn). Lippincott Williams & Wilkins, Philadelphia, pp 1126–1161
Conlon KC, Klimstra DS, Brennan MF (1996) Long-term survival after curative resection for pancreatic ductal adenocarcinoma. Clinicopathologic analysis of 5-year survivors. Ann Surg 223:273–279
Kalser MH, Ellenberg SS (1985) Pancreatic cancer. Adjuvant combined radiation and chemotherapy following curative resection. Arch Surg 120:899–903
Klinkenbijl JH, Jeekel J, Sahmoud T, van Pel R, Couvreur ML, Veenhof CH, Arnaud JP, Gonzalez DG, de Wit LT, Hennipman A, Wils J (1999) Adjuvant radiotherapy and 5-fluorouracil after curative resection of cancer of the pancreas and periampullary region: phase III trial of the EORTC gastrointestinal tract cancer cooperative group. Ann Surg 230:776–782
Neoptolemos JP, Dunn JA, Stocken DD, Almond J, Link K, Beger H, Bassi C, Falconi M, Pederzoli P, Dervenis C, Fernandez-Cruz L, Lacaine F, Pap A, Spooner D, Kerr DJ, Friess H, Buchler MW (2001) Adjuvant chemoradiotherapy and chemotherapy in resectable pancreatic cancer: a randomised controlled trial. Lancet 358:1576–1585
Neoptolemos JP, Stocken DD, Friess H, Bassi C, Dunn JA, Hickey H, Beger H, Fernandez-Cruz L, Dervenis C, Lacaine F, Falconi M, Pederzoli P, Pap A, Spooner D, Kerr DJ, Buchler MW (2004) A randomized trial of chemoradiotherapy and chemotherapy after resection of pancreatic cancer. N Engl J Med 350:1200–1210
Srivastava PK (2000) Immunotherapy of human cancer: lessons from mice. Nat Immunol 1:363–366
Srivastava PK, Maki RG (1991) Stress-induced proteins in immune response to cancer. Curr Top Microbiol Immunol 167:109–123
Basu S, Binder RJ, Ramalingam T, Srivastava PK (2001) CD91 is a common receptor for heat shock proteins gp96, hsp90, hsp70, and calreticulin. Immunity 14:303–313
Binder RJ, Srivastava PK (2004) Essential role of CD91 in re-presentation of gp96-chaperoned peptides. Proc Natl Acad Sci U S A 101:6128–6133
Janetzki S, Palla D, Rosenhauer V, Lochs H, Lewis JJ, Srivastava PK (2000) Immunization of cancer patients with autologous cancer-derived heat shock protein gp96 preparations: a pilot study. Int J Cancer 88:232–238
Belli F, Testori A, Rivoltini L, Maio M, Andreola G, Sertoli MR, Gallino G, Piris A, Cattelan A, Lazzari I, Carrabba M, Scita G, Santantonio C, Pilla L, Tragni G, Lombardo C, Arienti F, Marchiano A, Queirolo P, Bertolini F, Cova A, Lamaj E, Ascani L, Camerini R, Corsi M, Cascinelli N, Lewis JJ, Srivastava P, Parmiani G (2002) Vaccination of metastatic melanoma patients with autologous tumor-derived heat shock protein gp96-peptide complexes: clinical and immunologic findings. J Clin Oncol 20:4169–4180
Mazzaferro V, Coppa J, Carrabba MG, Rivoltini L, Schiavo M, Regalia E, Mariani L, Camerini T, Marchiano A, Andreola S, Camerini R, Corsi M, Lewis JJ, Srivastava PK, Parmiani G (2003) Vaccination with autologous tumor-derived heat-shock protein gp96 after liver resection for metastatic colorectal cancer. Clin Cancer Res 9:3235–3245
Caudill MM, Li Z (2001) HSPPC-96: a personalised cancer vaccine. Expert Opin Biol Ther 1:539–547
Younes A, Fayad LE, Pro B, McLaughlin P, Hagemeister FB, Mansfield P, Clayman G, Medeiros LJ, Lewis J, Srivastava P (2003) Safety and efficacy of heat shock protein-peptide 96 complex (HSPPC-96) in low-grade lymphoma. Proc Am Soc Clin Oncol 22:570 [Abstr 2294]
Srivastava PK, Jaikaria NS (2001) Methods of purification of heat shock protein-peptide complexes for use as vaccines against cancers and infectious diseases. Methods Mol Biol 156:175–186
Heike M, Blachere NE, Wolfel T, Meyer zum Buschenfelde KH, Storkel S, Srivastava PK (1994) Membranes activate tumor- and virus-specific precursor cytotoxic T lymphocytes in vivo and stimulate tumor-specific T lymphocytes in vitro: implications for vaccination. J Immunother Emphasis Tumor Immunol 15:165–174
Kaplan EL, Meier P (1958) Nonparametric estimation from incomplete observations. J Am Stat Assoc 53:457–481
Jaffee EM, Hruban RH, Biedrzycki B, Laheru D, Schepers K, Sauter PR, Goemann M, Coleman J, Grochow L, Donehower RC, Lillemoe KD, O'Reilly S, Abrams RA, Pardoll DM, Cameron JL, Yeo CJ (2001) Novel allogeneic granulocyte-macrophage colony-stimulating factor-secreting tumor vaccine for pancreatic cancer: a phase I trial of safety and immune activation. J Clin Oncol 19:145–156
Gjertsen MK, Buanes T, Rosseland AR, Bakka A, Gladhaug I, Soreide O, Eriksen JA, Moller M, Baksaas I, Lothe RA, Saeterdal I, Gaudernack G (2001) Intradermal ras peptide vaccination with granulocyte-macrophage colony-stimulating factor as adjuvant: Clinical and immunological responses in patients with pancreatic adenocarcinoma. Int J Cancer 92:441–450
Goydos JS, Elder E, Whiteside TL, Finn OJ, Lotze MT (1996) A phase I trial of a synthetic mucin peptide vaccine. Induction of specific immune reactivity in patients with adenocarcinoma. J Surg Res 63:298–304
Marshall J (2003) Carcinoembryonic antigen-based vaccines. Semin Oncol 30(3 Suppl 8):30–36
Schuetz T, Marshall J, Kaufman HL, Safran H, Panicali D (2004) Two phase I studies of prime-boost vaccinations with vaccinia-fowlpox vaccines expressing CEA, MUC-1, and TRICOM costimulatory molecules (B7.1/ICAM-1/LFA-3) in patients with advanced pancreatic cancer. J Clin Oncol 22(Suppl 14S):2564
Acknowledgments
The authors are grateful to all patients agreeing to enroll on this study. We wish to thank Jennifer Yamada and Cate Hirst for their assistance with data management over the several years of this study, and to H. Kelley for her assistance with the biostatistical analysis. Portions of this study were presented in abstract form at the American Society for Clinical Oncology meeting in 1999 (abstract 1687), and European Cancer Conference 12 in 2003 (ECCO, abstract 48). P.K.S. is supported by an NIH grant (CA 44786) and by a research agreement with Antigenics, Inc., in which he has significant financial interest. P.L. is supported in part by NIH grant CA 33049.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Maki, R.G., Livingston, P.O., Lewis, J.J. et al. A Phase I Pilot Study of Autologous Heat Shock Protein Vaccine HSPPC-96 in Patients With Resected Pancreatic Adenocarcinoma. Dig Dis Sci 52, 1964–1972 (2007). https://doi.org/10.1007/s10620-006-9205-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-006-9205-2