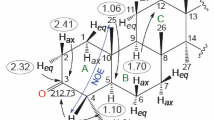
New oxidative and skeletal transformations of olean-9(11),12(13)-dien-30-oic acid that were based on ozonolysis and UV irradiation produced the 9α,11α-epoxide and opened ring C.
Similar content being viewed by others
References
C. S. Graebin, H. Verli, and J. A. Guimaraes, J. Braz. Chem. Soc., 21, 1595 (2010).
G. A. Tolstikov, L. A. Baltina, V. P. Grankina, R. M. Kondratenko, and T. G. Tolstikova, Licorice: Biodiversity, Chemistry, and Use in Medicine [in Russian], Akad. Izd. Geo, Novosibirsk, 2007.
P. Kalaiarasi and K. V. Pugalendi, Eur. J. Pharmacol., 606, 269 (2009).
L. R. Mikhailova, M. V. Khudobko, L. A. Baltina, Jr., L. V. Spirikhin, R. M. Kondratenko, and L. A. Baltina, Chem. Nat. Compd., 45, 393 (2009).
E. Pretsch, P. Buhlmann, and C. Affolter, Structure Determination of Organic Compounds, Springer, Berlin, New York, 2000.
L. R. Mikhailova, L. A. Baltina, R. M. Kondratenko, O. Kunert, L. V. Spirikhin, F. Z. Galin, and G. A. Tolstikov, Chem. Nat. Compd., 42, 553 (2006).
A. J. Gordon and R. A. Ford, A Chemist’s Companion, Wiley-Interscience, New York, 1972.
L. A. Baltina, O. B. Flekhter, Zh. M. Putieva, R. M. Kondratenko, L. V. Krasnova, and G. A. Tolstikov, Khim.-farm. Zh., 30, No. 4, 47 (1996).
Acknowledgment
The work was supported financially by the RFBR (Grant 11-03-00462a) and NSh 7014.2012.3.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiya Prirodnykh Soedinenii, No. 2, March–April, 2014, pp. 265–267.
Rights and permissions
About this article
Cite this article
Budaev, A.S., Mikhailova, L.R., Spirikhin, L.V. et al. Synthesis and NMR Spectra of New C-Modified Glycyrrhetic Acid Derivatives. Chem Nat Compd 50, 302–304 (2014). https://doi.org/10.1007/s10600-014-0937-5
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10600-014-0937-5