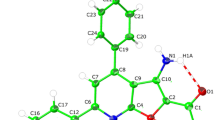
The new fluorophores of the 3H-imidazo[4,5-e][2,1]benzoxazoles series were synthesized by the regioselective nitration of 3-alkyl-8-phenyl-3H-imidazo[4,5-e][2,1]benzoxazoles. The latter compounds were obtained from the reaction of 1-alkyl-5-nitro-1Hbenzimidazoles with benzyl cyanide in basic MeOH solution. The structures of synthesized compounds were established using spectral (UV-vis, IR, 1H NMR, 13C NMR, and NOESY) and analytical data. Furthermore, it was found that these fluorophores underwent thermal rearrangement to new 5H-imidazo[4,5-f][2,1,3]benzoxadiazole 3-oxides in AcOH in moderate yields. The fluorescence properties and antibacterial activities of new compounds against Gram-positive and Gram-negative bacterial species were also studied.




Similar content being viewed by others
References
Kumar, A.; Kumar, R. A. Int. Res. J. Pharm. 2011, 2, 11.
Patel, N. B.; Shaikh, F. M. Sci. Pharm. 2010, 78, 753.
Bala, S.; Kamboj, S.; Kumar, A. J. Pharm. Res. 2010, 3, 2993.
Fridman, N.; Kaftory, M.; Speiser, S. Sens. Actuators, B 2007, 126, 107.
Karolak-Wojciechowska, J.; Mrozek, A.; Czylkowski, R.; Tekiner-Gulbas, B.; Akı-Sener, E.; Yalcin, I. J. Mol. Struct. 2007, 839, 125.
Pan, W. L.; Tan, H. B.; Chen, Y.; Mu, D. H.; Liu, H. B.; Wan, Y. Q.; Song, H. C. Dyes Pigm. 2008, 76, 17.
Um, S. I. Dyes Pigm. 2007, 75, 185.
Szarfman, A.; Tonning, J.; Levine, J.; Doraiswamy, P. Pharmacotherapy 2006, 26, 748.
Loudon, J. D.; Tennant, G. Quart. Rev. 1964, 18, 389.
Rahimizadeh, M.; Pordel, M.; Bakavoli, M.; Bakhtiarpoor, Z.; Orafaie, A. Monatsh. Chem. 2009, 140, 633.
Bakavoli, M.; Bagherzadeh, G.; Vaseghifar, M.; Shiri, A.; Pordel, M.; Mashreghi, M.; Pordeli, P.; Araghi, M. Eur. J. Med. Chem. 2010, 45, 647.
Pordel, M.; Abdollahi, A.; Razavi, B. Russ. J. Bioorg. Chem. 2013, 39, 211.
Pordel, M.; Beyramabadi, S. A.; Mohammadinejad, A. Dyes Pigm. 2014, 102, 46.
Baf, M. M. F.; Pordel, M.; Daghigh, L. R. Tetrahedron Lett. 2014, 55, 6925.
Alikhani, E.; Pordel, M.; Daghigh, L. R. Spectrochim. Acta, Part A 2015, 136, 1484.
Davis, R. B.; Pizzini, L. C. J. Org. Chem. 1960, 25, 1884.
Boulton, A. J.; Brown, R. C. J. Org. Chem. 1970, 35, 1662.
Sztaricskai, F.; Pinter, G.; Roth, E.; Herczegh, P.; Kardos, S.; Rozgonyi, F.; Boda, Z. J. Antibiot. 2007, 60, 529.
Joux, F.; Lebaron, P. Microbes Infect. 2000, 2, 1523.
Preston, P. N. The Chemistry of Heterocyclic Compounds, Benzimidazoles and Cogeneric Tricyclic Compounds; John Wiley & Sons Interscience, pt. 1, Vol. 40, p. 87.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Khimiya Geterotsiklicheskikh Soedinenii, 2015, 51(10), 918–922
Rights and permissions
About this article
Cite this article
Rezazadeh, M., Pordel, M., Davoodnia, A. et al. New fluorescent 3H-imidazo[4,5-e][2,1]benzoxazoles: synthesis, spectroscopic characterization, and antibacterial activity. Chem Heterocycl Comp 51, 918–922 (2015). https://doi.org/10.1007/s10593-015-1796-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-015-1796-0