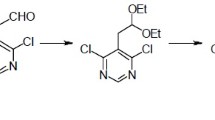
A practical, robust and scalable synthesis of 6-bromo-4-chlorothieno[2,3-d]pyrimidine starting from cheap bulk chemicals has been developed. The method involves four synthetic steps: Gewald reaction, pyrimidone formation, bromination, and chlorination. The process relies on standard laboratory equipment, allowing to obtain the product in an overall yield of 49% without using chromatography for purification of the product and intermediates.





Similar content being viewed by others
Notes
* As an exception, scale in this case is based on amount of product produced in the reaction. If nothing else is indicated, scale is based on the amount of substrate for the reaction.
References
Z. A. Hozien, F. M. Atta, K. M. Hassan, A. A. Abdel-Wahab, and S. A. Ahmed, Synth. Commun., 26, 3733 (1996).
P. J. Hill, A. Abibi, R. Albert, B. Andrews, M. M. Gagnon, N. Gao, T. Grebe, L. I. Hajec, J. Huang, S. Livchak, S. D. Lahiri, D. C. McKinney, J. Thresher, H. Wang, N. Olivier, and E. T. Buurman, J. Med. Chem., 56, 7278 (2013).
S. B. Kanawade, R. B. Toche, and D. P. Rajani, Eur. J. Med. Chem., 64, 314 (2013).
N. Tani, M. Rahnasto-Rilla, C. Wittekindt, K. A. Salminen, A. Ritvanen, R. Ollakka, J. Koskiranta, H. Raunio, and R. O. Juvonen, Eur. J. Med. Chem., 47, 270 (2012).
J. S. De, L.-J. Gao, P. Herdewijn, J. Herman, M. Jang, P. Leyssen, T. Louat, J. Neyts, C. Pannecouque, and B. Vanderhoydonck, WO Pat. Appl. 2011147753.
C.-Y. Leung, A. M. Langille, J. Mancuso, and Y. S. Tsantrizos, Bioorg. Med. Chem., 21, 2229 (2013).
C. Y. Leung, J. Park, J. W. De Schutter, M. Sebag, A. M. Berghuis, and Y. S. Tsantrizos, J. Med. Chem., 56, 7939 (2013).
B. C. Shook, D. Chakravarty, J. K. Barbay, A. Wang, K. Leonard, V. Alford, M. T. Powell, S. Rassnick, R. H. Scannevin, K. Carroll, N. Wallace, J. Crooke, M. Ault, L. Lampron, L. Westover, K. Rhodes, and P. F. Jackson, Bioorg. Med. Chem. Lett., 23, 2688 (2013).
J. K. Barbay, D. Chakravarty, B. C. Shook, and A. Wang, WO Pat. Appl. 2010045006.
C. Tintori, I. Laurenzana, F. La Rocca, F. Falchi, F. Carraro, A. Ruiz, J. A. Este, M. Kissova, E. Crespan, G. Maga, M. Biava, C. Brullo, S. Schenone, and M. Botta, ChemMedChem, 8, 1353 (2013).
M.-Y. Jang, S. De Jonghe, K. Van Belle, T. Louat, M. Waer, and P. Herdewijn, Bioorg. Med. Chem. Lett., 20, 844 (2010).
E. R. Wood, L. M. Shewchuk, B. Ellis, P. Brignola, R. L. Brashear, T. R. Caferro, S. H. Dickerson, H. D. Dickson, K. H. Donaldson, M. Gaul, R. J. Griffin, A. M. Hassell, B. Keith, R. Mullin, K. G. Petrov, M. J. Reno, D. W. Rusnak, S. M. Tadepalli, J. C. Ulrich, C. D. Wagner, D. E. Vanderwall, A. G. Waterson, J. D. Williams, W. L. White, and D. E. Uehling, Proc. Natl. Acad. Sci. USA, 105, 2773 (2008).
Y. Dai, Y. Guo, R. R. Frey, Z. Ji, M. L. Curtin, A. A. Ahmed, D. H. Albert, L. Arnold, S. S. Arries, T. Barlozzari, J. L. Bauch, J. J. Bouska, P. F. Bousquet, G. A. Cunha, K. B. Glaser, J. Guo, J. Li, P. A. Marcotte, K. C. Marsh, M. D. Moskey, L. J. Pease, K. D. Stewart, V. S. Stoll, P. Tapang, N. Wishart, S. K. Davidsen, and M. R. Michaelides, J. Med. Chem., 48, 6066 (2005).
H.-P. Hsieh, S. M. Coumar, T.-A. Hsu, W.-H. Lin, Y.-R. Chen, and Y.-S. Chao, US Pat. Appl. 20100120805.
C.-H. Wu, M. S. Coumar, C.-Y. Chu, W.-H. Lin, Y.-R. Chen, C.-T. Chen, H.-Y. Shiao, S. Rafi, S.-Y. Wang, H. Hsu, C.-H. Chen, C.-Y. Chang, T.-Y. Chang, T.-W. Lien, M.-Y. Fang, K.-C. Yeh, C.-P. Chen, T.-K. Yeh, S.-H. Hsieh, J. T.-A. Hsu, C.-C. Liao, Y.-S. Chao, and H.-P. Hsieh, J. Med. Chem., 53, 7316 (2010).
A. Zhao, X. Gao, Y. Wang, J. Ai, Y. Wang, Y. Chen, M. Geng, and A. Zhang, Bioorg. Med. Chem., 19, 3906 (2011).
J. Zhang, CN Pat. Appl. 102872018.
T. P. Heffron, B. Q. Wei, A. Olivero, S. T. Staben, V. Tsui, S. Do, J. Dotson, A. J. Folkes, P. Goldsmith, R. Goldsmith, J. Gunzner, J. Lesnick, C. Lewis, S. Mathieu, J. Nonomiya, S. Shuttleworth, D. P. Sutherlin, N. C. Wan, S. Wang, C. Wiesmann, and B.-Y. Zhu, J. Med. Chem., 54, 7815 (2011).
S. Bugge, S. J. Kaspersen, S. Larsen, U. Nonstad, G. Bjørkøy, E. Sundby, and B. H. Hoff, Eur. J. Med. Chem., 75, 354 (2014).
J. K. Barbay, K. Leonard, D. Chakravarty, B. C. Shook, and A. Wang, WO Pat. Appl. 2010045013.
T. Sakamoto, Y. Kondo, R. Watanabe, and H. Yamanaka, Chem. Pharm. Bull., 34, 2719 (1986).
T. Sakamoto, Y. Kondo, and H. Yamanaka, Chem. Pharm. Bull., 30, 2417 (1982).
M. Robba, J. M. Lecomte, and M. Cugnon de Sevricourt, Bull. Soc. Chim. Fr., 587 (1975).
V. P. Arya, Indian J. Chem., 10, 1141 (1972).
R. W. Sabnis, Sulfur Rep., 16, 1 (1994).
R. W. Sabnis, D. W. Rangnekar, and N. D. Sonawane, J. Heterocycl. Chem., 36, 333 (1999).
J. Grembecka, S. He, A. Shi, T. Purohit, A. G. Muntean, R. J. Sorenson, H. D. Showalter, M. J. Murai, A. M. Belcher, T. Hartley, J. L. Hess, and T. Cierpicki, Nat. Chem. Biol., 8, 277 (2012).
P. Herdewijn, J. S. De, L.-J. Gao, M.-Y. Jang, B. Vanderhoydonck, M. J. A. Waer, Y. Lin, J. F. Herman, and T. A. M. Louat, WO Pat. Appl. 2010103130.
S. Bugge, S. J. Kaspersen, E. Sundby, and B. H. Hoff, Tetrahedron, 68, 9226 (2012).
K. Wang, D. Kim, and A. Domling, J. Comb. Chem., 12, 111 (2010).
G. Hallas and A. D. Towns, Dyes Pigm., 33, 319 (1997).
F. D. Therkelsen, M. Rottländer, N. Thorup, and E. B. Pedersen, Org. Lett., 6, 1991 (2004).
J. Herman and T. Louat, WO Pat. Appl. 2012035423.
S. Hesse, E. Perspicace, and G. Kirsch, Tetrahedron Lett., 48, 5261 (2007).
T. R. Caferro, S. D. Chamberlain, K. H. Donaldson, P. A. Harris, M. D. Gaul, D. E. Uehling, and D. E. Vanderwall, WO Pat. Appl. 2003053446.
M. Robba, J. M. Lecomte, and M. Cugnon de Sevricourt, Bull. Soc. Chim. Fr., 592 (1975).
T. Park, M. F. Mayer, S. Nakashima, and S. C. Zimmerman, Synlett, 1435 (2005).
K. Gewald, Chem. Ber., 98, 3571 (1965).
Y. Huang, S. Wolf, M. Bista, L. Meireles, C. Camacho, T. A. Holak, and A. Domling, Chem. Biol. Drug Des., 76, 116 (2010).
J. F. Atherall, T. L. Hough, S. D. Lindell, M. J. O'Mahony, E. A. Saville-Stones, and J. H. Parsons, WO Pat. Appl. 9849899.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Khimiya Geterotsiklicheskikh Soedinenii, No. 8, pp. 1275-1285, August, 2014.
Rights and permissions
About this article
Cite this article
Bugge, S., Skjønsfjell, E.M., Willumsen, F.B. et al. Improved and Scalable Preparation of 6-Bromo-4-Chlorothieno[2,3-d]Pyrimidine. Chem Heterocycl Comp 50, 1177–1187 (2014). https://doi.org/10.1007/s10593-014-1579-z
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-014-1579-z