Abstract
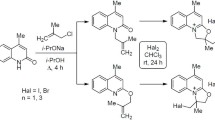
The bromination of 3-allyl-4-hydroxy-2-oxo-1,2-dihydroquinoline by molecular bromine is accompanied by the closure of a five membered furan ring and gives the corresponding 2-bromomethyl-3,9-dihydro-2H-furo[2,3-b]quinolin-4-one.
Similar content being viewed by others
References
I. V. Ukrainets, L. V. Sidorenko, E. N. Svechnikova, and O. V. Shishkin, Khim. Geterotsikl. Soedin., 1503 (2007). [Chem. Heterocycl. Comp., 43, 1275 (2007)].
I. V. Ukrainets, L. V. Sidorenko, O. V. Gorokhova, S. V. Shishkina, and A. V. Turov, Khim. Geterotsikl. Soedin., 736 (2007). [Chem. Heterocycl. Comp., 43, 617 (2007)].
I. V. Ukrainets, N. L. Bereznyakova, V. A. Parshikov, and A. V. Turov, Khim. Geterotsikl. Soedin., 1496 (2007). [Chem. Heterocycl. Comp., 43, 1269 (2007)].
I. V. Ukrainets, N. L. Bereznyakova, O. V. Gorokhova, A. V. Turov, and S. V. Shishkina, Khim. Geterotsikl. Soedin., 1180 (2007). [Chem. Heterocycl. Comp., 43, 1001 (2007)].
I. V. Ukrainets, N. L. Bereznyakova, A. V. Turov, and S. V. Slobodzyan, Khim. Geterotsikl. Soedin., 1365 (2007). [Chem. Heterocycl. Comp., 43, 1159 (2007)].
I. V. Ukrainets, S. G. Taran, O. A. Evtifeeva, O. V. Gorokhova, N. I. Filimonova, and A. V. Turov, Khim. Geterotsikl. Soedin., 204 (1995). [Chem. Heterocycl. Comp., 31, 176 (1995)].
I. V. Ukrainets, L. V. Sidorenko, O. V. Gorokhova, E. V. Mospanova, and O. V. Shishkin, Khim. Geterotsikl. Soedin., 718 (2006). [Chem. Heterocycl. Comp., 42, 631 (2006)].
I. V. Ukrainets, A. A. Tkach, L. V. Sidorenko, and O. V. Gorokhova, Khim. Geterotsikl. Soedin., 1508 (2006). [Chem. Heterocycl. Comp., 42, 1301 (2006)].
Yu. V. Zefirov, Kristallografiya, 42, 936 (1997).
H.-B. Burgi and J. D. Dunitz, Structure Correlation, Vol. 2, VCH, Weinheim (1994), p. 741.
L. Jurd, M. Benson, and R. Y. Wong, Aust. J. Chem., 36, 759 (1983).
I. V. Ukrainets, S. G. Taran, O. L. Kamenetskaya, O. V. Gorokhova, L. V. Sidorenko, and A. V. Turov, Khim. Geterotsikl. Soedin., 1532 (2000). [Chem. Heterocycl. Comp., 36, 1319 (2000)].
P. B. Terentiev and A. P. Stankevicius, Mass-spectrometric Analysis of Biologically Active Compounds [in Russian], Mosklas, Vilnius (1987), p. 255.
I. V. Ukrainets, S. G. Taran, O. A. Evtifeeva, O. V. Gorokhova, P. A. Bezugly, A. V. Turov, L. N. Voronina, and N. I. Filimonova, Khim. Geterotsikl. Soedin., 673 (1994). [Chem. Heterocycl. Comp., 30, 591 (1994)].
G. M. Sheldrick, SHELXTL PLUS. PC Version. A System of Computer Programs for the Determination of Crystal Structure from X-ray Diffraction Data. Rev. 5.1 (1998).
Author information
Authors and Affiliations
Corresponding author
Additional information
__________
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 11, pp. 1677–1686, November, 2007.
Rights and permissions
About this article
Cite this article
Ukrainets, I.V., Bereznyakova, N.L., Gorokhova, O.V. et al. 4-Hydroxy-2-quinolones 131. Bromination of 3-allyl-4-hydroxy-2-oxo-1,2-dihydroquinoline. Chem Heterocycl Comp 43, 1426–1433 (2007). https://doi.org/10.1007/s10593-007-0220-9
Received:
Issue Date:
DOI: https://doi.org/10.1007/s10593-007-0220-9