Abstract
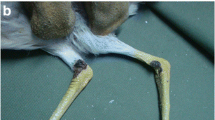
We cultured avian pox (Avipoxvirus spp.) from lesions collected on Hawai‘i, Maui, Moloka‘i, and ‘Oahu in the Hawaiian Islands from 15 native or non-native birds representing three avian orders. Phylogenetic analysis of a 538 bp fragment of the gene encoding the virus 4b core polypeptide revealed two distinct variant clusters, with sequences from chickens (fowlpox) forming a third distinct basal cluster. Pox isolates from one of these two clusters appear closely related to canarypox and other passerine pox viruses, while the second appears more specific to Hawai‘i. There was no evidence that birds were infected simultaneously with multiple pox virus variants based on evaluation of multiples clones from four individuals. No obvious temporal or geographic associations were observed and strict host specificity was not apparent among the 4b-defined field isolates. We amplified a 116 bp 4b core protein gene fragment from an ‘Elepaio (Chasiempis sandwichensis) collected in 1900 on Hawai‘i Island that clustered closely with the second of the two variants, suggesting that this variant has been in Hawai‘i for at least 100 years. The high variation detected between the three 4b clusters provides evidence for multiple, likely independent introductions, and does not support the hypothesis of infection of native species through introduction of infected fowl. Preliminary experimental infections in native Hawai‘i ‘Amakihi (Hemignathus virens) suggest that the 4b-defined variants may be biologically distinct, with one variant appearing more virulent. These pox viruses may interact with avian malaria (Plasmodium relictum), another introduced pathogen in Hawaiian forest bird populations, through modulation of host immune responses.



Similar content being viewed by others
References
Afonso CL, Tulmen ER, Lu Z, Zsak L, Kutish GF, Rock DL (2000) The genome of the fowlpox virus. J Virol 74:3815–3831
Atkinson CT, Woods KL, Dusek RJ, Sileo LS, Iko WM (1995) Wildlife disease and conservation in Hawai‘i: Pathogenicity of avian malaria (Plasmodium relictum) in experimentally infected Iiwi (Vestiaria coccinea). Parasitology 111(Suppl.):59–69
Atkinson CT, Dusek RJ, Woods KL, Iko WM (2000) Pathogenicity of avian malaria in experimentally-infected Hawai‘i ‘Amakihi. J Wildlife Diseases 36:197–204
Atkinson CT, Dusek RJ, Lease JK, Samuel MD (2005) Prevalence of pox-like lesions and malaria in forest bird communities on leeward Mauna Loa Volcano, Hawai‘i. Condor 107:537–546
Babkin IV, Shchelkunov SN (2006) Time scale of poxvirus evolution. Molecular Biology 40(1):16–19 (4)
Binns MM, Boursnell MEG, Tomley FM, Campbell J (1989) Analysis of the fowlpoxvirus gene encoding the 4b core polypeptide and demonstration that it possesses efficient promoter sequences. Virology 170:288–291
Hardy DE (1960) Insects of Hawai‘i. Diptera: Nematocera-Brachycera. University of Hawai‘i Press. Honolulu, HI. 10
Henshaw HW (1902) Birds of the Hawaiian Islands, being a complete list of the birds of the Hawaiian possessions with notes on their habits. TG Thrum, Honolulu, Hawaii
Jarvi SI, Farias MEM (2006) Molecular sexing and sources of CHD1-Z/W sequence variation in Hawaiian birds. Mol Ecol Notes 6(4):1003–1005
Kato SE, Strahl AL, Moussatche N, Condit RC (2004) Temperature-sensitive mutants in the vaccinia virus 4b virion structural protein assemble malformed, transcriptionally inactive intracellular mature virions. Virology 330(1):127–146
Kim T, Tripathy DN (2006) Evaluation of pathogenicity of avian poxvirus isolates from endangered Hawaiian wild birds in chickens. Avian Diseases 50(2):288–291
Kotwal GJ (1997) Microorganisms and their interaction with the immune system. J Leukoc Biol 62:415–429
Kumar S, Tamura K, Jakobsen I, Nei M (2000) MEGA: Molecular Evolutionary Genetics Analysis, Version 2.1. Arizona State University, Tempe, AZ
Lee LH, Lee KH (1997) Application of the polymerase chain reaction for the diagnosis of fowl poxvirus infection. J Virol Methods 63:113–119
Lefkowitz EJ, Wang C, Upton C (2006) Poxviruses: past, present and future. Virus Res 117(1):105–118
McLysaght A, Baldi PF, Gaut BS (2003) Extensive gene gain associated with adaptive evolution of poxviruses. Proc. Nat Acad Sci 100(26):15655–15660
Moss B (1978) In: Nayak DP (ed) Molecular biology of animals. Dekker, New York
Moss B (2001) Poxviridae: the viruses and their Replication. In: Fields BN, Knipe DM, Howley PM, Griffin DE (eds) Fields virology. Lippincott Williams & Wilkins, Philadelphia, pp 2849–2884
Perkins RCL (1903) Vertebrata (Aves). In: Sharp D (eds) Fauna Hawaiiensis, vol 1, Part IV (University Press, Cambridge, UK, pp 368–465
Saitou N, Nei M (1987) The neighbor-joining method. A new method for reconstructing phylogenetic trees. Mol Biol Evol 4:406–425
Smith SA, Kotwal GJ (2002) Immune response to poxvirus infections in various animals. Crit Rev Microbiol 28(3):149–185
Thiel T, Whiteman NK, Tirape A, Baquero MI, Cedeno V, Walsh T, Uzcategui GJ, Parker PG (2005) Characterization of canarypox-like viruses infecting endemic birds in the Galapagos Islands. J Wildlife Diseases 41(2):342–353
Thomas MB, Watson EL, Valverde-Garcia P (2003) Mixed infections and insect-pathogen interactions. Ecol Lett 6:183–188
Tripathy DN (1993) Avipox viruses. In: McFerran JB, McNulty MS (eds) Virus Infection of birds. Elsevier Science Pub, New York, NY, pp 5–15
Tripathy DN, Schnitzlein WM, Morris PJ, Jannsen DL, Zuba JK, Massey G, Atkinson CT (2000) Characterization of poxviruses from forest birds in Hawai‘i. J Wildlife Diseases 36(2):225–230
VanderWerf EA (2001) Distribution and potential impacts of avian poxlike lesions in ‘Elepaio at Hakalau Forest National Wildlife Refuge In: Scott JM, Conant S, van Riper III C (eds) Evolution, ecology, conservation and management of Hawaiian birds: a vanishing avifauna vol 22. Studies in Avian Biology, pp 247–253
van Riper III C, van Riper SG, Goff ML, Laird M (1986) The epizootiology and ecological significance of malaria in Hawaiian land birds. Ecol Monogr 56:327–344
van Riper III C, van Riper SG, Hansen WR (2002) Epizootiology and effect of avian pox on Hawaiian forest birds. The Auk 119(4):929–942
Warner RE (1968) The role of introduced diseases in the extinction of the endemic Hawaiian avifauna. Condor 70:101–120
Woodworth BL, Atkinson CT, LaPointe DA, Hart PJ, Spiegel CS, Tweed EJ, Hennemann C, LeBrun J, Denette T, DeMotts R, Kozar KL, Triglia D, Lease D, Gregor A, Smith T, Duffy D (2005) Host population persistence in the face of introduced vector-borne diseases: Hawai‘i ‘Amakihi and avian malaria. Proceedings of the National Academy of Sciences 102:1531–1536
Acknowledgements
This work was supported in part by National Science Foundation Grant DEB 0083944 and the USGS Wildlife and Invasive Species Programs. We thank Carla Kishinami and the Vertebrate Zoology Section at the Bernice Pauahi Bishop Museum, Honolulu, Hawaii for cooperation and assistance in collecting pox lesions from ‘Elepaio specimens in their collection, Wallace Hansen at the USGS-National Wildlife Health Center for supplying embryonic Muscovy Duck fibroblasts and advice for pox virus cultivation, Alan Lieberman at the Keauhou Bird Conservation Center for pox virus lesions from captive Palila and ‘Alalā, Eric VanderWerf, Fern Duvall, Sam Aruch, Julie Lease, Donna Ball, Eric Tweed, Paul Banko, and Colleen Cole for collecting pox lesions from other species and Leayne Patch for assistance with aviary experiments. Any use of trade, product, or firm names in this publication is for descriptive purposes only and does not imply endorsement by the U.S. Government. Mahalo i ka po‘e o Hawai‘i no ko lākou kōkua a me ko lākou aloha. Thank you very much to the people of Hawai‘i for their help and aloha.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jarvi, S.I., Triglia, D., Giannoulis, A. et al. Diversity, origins and virulence of Avipoxviruses in Hawaiian Forest Birds. Conserv Genet 9, 339–348 (2008). https://doi.org/10.1007/s10592-007-9346-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10592-007-9346-7