Abstract
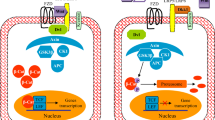
Breast cancer is the second leading cause of cancer death in women and metastasizes to bone in greater than 80 % of advanced-disease patients. Once breast cancer bone metastases are established, the disease is incurable and drives numerous complications that increase morbidity and diminish patients’ quality of life. Many mechanisms have been implicated in bone metastases of breast cancer. The critical role of Wnt signalling pathway inhibition in initiating bone lesions has been demonstrated in a variety of bone diseases and tumours. Overexpression of dickkopf-1 (Dkk1) protein, a negative regulator of the Wnt/β-catenin pathway, has been found in breast cancer cell lines that form osteolytic metastases preferentially and in serum from breast cancer patients with osteolytic bone metastases. Further understanding of the mechanistic role of Dkk1 as a link between primary breast tumours and secondary osteolytic bone metastases may facilitate development of anti-Dkk1 antibody therapeutic tools.


Similar content being viewed by others
References
Ferlay J, Shin HR, Bray F et al (2010) Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int J Cancer 127(12):2893–2917
Kozlow W, Guise TA (2005) Breast cancer metastasis to bone: mechanisms of osteolysis and implications for therapy. J Mammary Gland Biol Neoplasia 10:169–180
Mundy GR (2002) Metastasis to bone: causes, consequences and therapeutic opportunities. Nat Rev Cancer 2:584–593
Lipton A (2005) Management of bone metastases in breast cancer. Cur Treat Options Oncol 6:161–171
Aubin JE, Triffitt J (2002) Mesenchymal stem cells and the osteoblast lineage. In: Principles of bone biology, 2nd edn. Academic Press, New York, pp 59–81
Suda T, Takahashi N, Udagawa N et al (1999) Modulation of osteoclast differentiation and function by the new members of the tumor necrosis factor receptor and ligand families. Endocr Rev 20:345–357
Simonet WS, Lacey DL, Dunstan CR et al (1997) Osteoprotegerin: a novel secreted protein involved in the regulation of bone density. Cell 89:309–319
Boyce BF, Xing L (2008) Functions of RANKL/RANK/OPG in bone modelling and remodelling. Arch Biochem Biophys 473:139–146
Quinn JM, Saleh H (2009) Modulation of osteoclast function in bone by the immune system. Mol Cell Endocrinol 310:40–51
Weitzmann MN (2013) The role of inflammatory cytokines, the RANKL/OPG axis, and the immunoskeletal interface in physiological bone turnover and osteoporosis. Scientifica. doi:10.1155/2013/125705
Fidler IJ (2003) The pathogenesis of cancer metastasis: the ‘seed and soil’ hypothesis revisited. Nat Rev Cancer 3:1–6
Yang J, Mani SA, Donaher JL et al (2004) Twist, a master regulator of morphogenesis, plays an essential role in tumor metastases. Cell 117:927–939
Eckhardt BL, Parker BS, van Laar RK et al (2005) Genomic analysis of a spontaneous model of breast cancer metastasis to bone reveals a role for the extracellular matrix. Mol Cancer Res 3:1–13
Bohn OL, Nasir I, Brufsky A et al (2009) Biomarker profile in breast carcinomas presenting with bone metastasis. Int J Clin Exp Pathol 3:139–146
Ota D, Mimori K, Yokobori T et al (2011) Identification of recurrence-related microRNAs in the bone marrow of breast cancer patients. Int J Oncol 38:955–962
Patel LR, Camacho DF, Shiozawa Y et al (2008) Loss of E-cadherin promotes metastasis via multiple downstream transcriptional pathways. Cancer Res 68:3645–3654
Onder TT, Gupta PB, Mani SA et al (2005) Breast cancer metastasis to bone: mechanisms of osteolysis and implications for therapy. Mammary Gland Biol Neoplasia 10:169–180
Guise TA, Kozlow WM, Heras-Herzig A et al (2005) Molecular mechanisms of breast cancer metastases to bone. Clin Breast Cancer 5:46–53
Kakonen SM, Mundy GR (2003) Mechanisms of osteolytic bone metastases in breast carcinoma. Cancer 97:834–839
Kozlow W, Guise TA (2005) Breast cancer metastasis to bone: mechanisms of osteolysis and implications for therapy. Mammary Gland Biol Neoplasia 10:169–180
Sethi N, Dai X, Winter CG, Kang Y (2011) Tumor-derived JAGGED1 promotes osteolytic bone metastasis of breast cancer by engaging notch signaling in bone cells. Cancer Cell 19:192–205
Karaplis A, Goltzman D (2000) PTH and PTHrP effects on the skeleton. Rev Endocr Metab Disord 1:331–341
Li J, Karaplis AC, Huang DC et al (2011) PTHrP drives breast tumor initiation, progression, and metastasis in mice and is a potential therapy target. J Clin Investig 121:4655–4669
Bohn OL, Nasir I, Brufsky A et al (2010) Biomarker profile in breast carcinomas presenting with bone metastasis. Int J Clin Exp Pathol 3:139–146
Guise TA, Yin JJ, Taylor SD et al (1996) Evidence for a causal role of parathyroid hormone-related protein in the pathogenesis of human breast cancer-mediated osteolysis. J Clin Investig 98:1544–1549
de la Mata J, Uy HL, Guise TA et al (1995) Interleukin-6 enhances hypercalcemia and bone resorption mediated by parathyroid hormone-related protein in vivo. J Clin Investig 95:2846–2852
Bendre MS, Margulies AG, Walser B et al (2005) Tumor-derived interleukin-8 stimulates osteolysis independent of the receptor activator of nuclear factor-kappaB ligand pathway. Cancer Res 65:11001–11009
Dallas SL, Rosser JL, Mundy GR et al (2002) Proteolysis of latent transforming growth factor-beta (TGF-beta)-binding protein-1 by osteoclasts. A cellular mechanism for release of TGF-beta from bone matrix. J Biol Chem 277:21352–21360
Kang Y, Siegel PM, Shu W et al (2003) A multigenic program mediating breast cancer metastasis to bone. Cancer Cell 3:537–549
Feeley BT, Gamradt SC, Hsu WK et al (2005) Influence of BMPs on the formation of osteoblastic lesions in metastatic prostate cancer. J Bone Miner Res 20:2189–2199
Dai J, Keller J, Zhang J et al (2005) Bone morphogenetic protein-6 promotes osteoblastic prostate cancer bone metastases through a dual mechanism. Cancer Res 65:8274–8285
Gregory LS, Choi W, Burke L et al (2013) Breast cancer cells induce osteolytic bone lesions in vivo through a reduction in osteoblast activity in mice. PLoS One. doi:10.1371/journal.pone.0068103
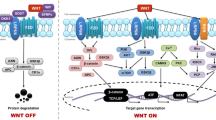
Milat F, Ng KW (2009) Is Wnt signalling the final common pathway leading to bone formation? Mol Cell Endocrinol 310:52–62
Gordon MD, Nusse R (2006) Wnt signaling: multiple pathways, multiple receptors, and multiple transcription factors. J Biol Chem 281:22429–22433
Cadigan KM, Liu YI (2006) Wnt signalling: complexity at the surface. J Cell Sci 119:395–402
Miller JR (2002) The Wnts. Genome Biol 3:3001.1–3001.15
Clevers H (2006) Wnt/β-Catenin signaling in development and disease. Cell 127:469–480
van Amerongen R, Mikels A, Nusse R (2008) Alternative Wnt signaling is initiated by distinct receptors. Sci Signal. doi:10.1126/scisignal.135re9
Bryja V, Andersson ER, Shambony A et al (2009) The extracellular domain of Lrp5/6 inhibits noncanonical Wnt signaling in vivo. Mol Biol Cell 20:924–936
Bovolenta P, Esteve P, Ruiz JM et al (2008) Beyond Wnt inhibition: new functions of secreted Frizzled-related proteins in development and disease. J Cell Sci 121:737–746
He X, Semenov M, Tamai K et al (2004) LDL receptor-related proteins 5 and 6 in Wnt/β-catenin signaling: arrows point the way. Development 131:1663–1677
Krishnan V, Bryant HU, Macdougald OA (2006) Regulation of bone mass by Wnt signaling. J Clin Investig 116:1202–1209
Yan C, Benjamin A (2009) Wnt pathway, an essential role in bone regeneration. J Cell Biochem 106:353–362
Hill TP, Später D, Taketo MM et al (2005) Canonical Wnt/beta-catenin signaling prevents osteoblasts from differentiating into chondrocytes. Dev Cell 8:727–738
Vaes BLT, Dechering KJ, van Someren EP et al (2005) Microarray analysis reveals expression regulation of Wnt antagonists in differentiating osteoblasts. Bone 36:803–811
Kato M, Patel MS, Levasseur R et al (2002) Cbfa1-independent decrease in osteoblast proliferation, osteopenia, and persistent embryonic eye vascularization in mice deficient in Lrp5, a Wnt coreceptor. J Cell Biol 157:303–314
Glass DA 2nd, Bialek P, Ahn JD et al (2005) Canonical Wnt signaling in differentiated osteoblasts controls osteoclast differentiation. Dev Cell 8:751–764
Gunn WG, Conley A, Deininger L et al (2006) A crosstalk between myeloma cells and marrow stromal cells stimulates production of DKK1 and interleukin-6: a potential role in the development of lytic bone disease and tumor progression in multiple myeloma. Stem Cells 24:986–991
Hall CL, Daignault SD, Shah RB et al (2008) Dickkopf-1 expression increases early in prostate cancer development and decreases during progression from primary tumor to metastasis. Prostate 68:1396–1404
Barth AL, Näthke IS, Nelson WJ (1997) Cadherins, catenins and APC protein: interplay between cytoskeletal complexes and signaling pathways. Curr Opin Cell Biol 9:683–690
Fuchs E (2009) The tortoise and the hair: slow-cycling cells in the stem cell race. Cell 137:811–819
Brennan KR, Brown AMC (2004) Wnt proteins in mammary development and cancer. J Mammary Gland Biol Neoplasia 9:119–131
Hatsell S, Rowlands T, Hiremath M et al (2003) Beta-catenin and Tcfs in mammary development and cancer. J Mammary Gland Biol Neoplasia 8:145–158
Rijsewijk F, Schuermann M, Wagenaar E et al (1987) The Drosophila homolog of the mouse mammary oncogene int-1 is identical to the segment polarity gene wingless. Cell 14:649–657
Prosperi JR, Khramtsov AI, Khramtsova GF et al (2011) Apc mutation enhances PyMT-induced mammary tumorigenesis. PLOS One. doi:10.1371/journal.pone.0029339
Klopocki E, Kristiansen G, Wild PJ et al (2004) Loss of SFRP1 is associated with breast cancer progression and poor prognosis in early stage tumors. Int J Oncol 25:641–649
Ai L, Tao Q, Zhong S et al (2006) Inactivation of Wnt inhibitory factor-1 (WIF1) expression by epigenetic silencing is a common event in breast cancer. Carcinogenesis 27:1341–1348
Zardawi SJ, O’Toole SA, Sutherland RL et al (2009) Dysregulation of Hedgehog, Wnt and Notch signalling pathways in breast cancer. Histol Histopathol 24:385–398
Ryo A, Nakamura M, Wulf G et al (2001) Pin1 regulates turnover and subcellular localization of beta-catenin by inhibiting its interaction with APC. Nat Cell Biol 3:793–801
Dolled-Filhart M, McCabe A, Giltnane J et al (2006) Quantitative in situ analysis of beta-catenin expression in breast cancer shows decreased expression is associated with poor outcome. Cancer Res 66:5487–5494
Wong SC, Lo SF, Lee KC et al (2002) Expression of frizzled-related protein and Wnt-signalling molecules in invasive human breast tumours. Br J Cancer 87:1281–1286
Chen Y, Shi HY, Stock SR et al (2011) Regulation of breast cancer-induced bone lesions by β-catenin protein signaling. J Biol Chem 286:42575–42584
Johnson RW, Merkel AR, Page JM et al (2014) PTHrP Wnt signaling induces gene expression of factors associated with bone destruction in lung and breast cancer. Clin Exp Metastasis 31:945–959
Johnson RW, Mai PN, Susan SP et al (2010) TGF-β promotion of Gli2-induced expression of parathyroid hormone-related protein, an important osteolytic factor in bone metastasis, is independent of canonical hedgehog signaling. Cancer Res 71:822–831
Thiery JP, Acloque H, Huang RY et al (2009) Epithelial-mesenchymal transitions in development and disease. Cell 139:871–890
McDermott SP, MS Wicha (2010) Targeting breast cancer stem cells. Mol Oncol 4:404–419
Mani SA, Guo W, Mai-Jing L et al (2008) The epithelial-mesenchymal transition generates cells wit properties of stem cells. Cell 133:704–715
Scheel C, Eaton EN, Li SH et al (2011) Paracrine and autocrine signals induce and maintain mesenchymal and stem cell states in the breast. Cell 145:926–940
Lamb R, Ablett MP, Spence K et al (2013) Wnt pathway activity in breast cancer sub-types and stem-like cells. PLoS One 8(7):e67811
Pedersen KB, Nesland JM, Fodstad O et al (2002) Expression of S100A4. E-cadherin, alpha- and beta-catenin in breast cancer biopsies. Br J Cancer 87:1281–1286
Gillett CE, Miles DW, Ryder K et al (2001) Retention of the expression of E-cadherin and catenins is associated with shorter survival in grade III ductal carcinoma of the breast. J Pathol 193:433–441
Bukholm IK, Nesland JM, Karesen R et al (1998) E-cadherin and alpha-, beta-, and gamma-catenin protein expression in relation to metastasis in human breast carcinoma. J Pathol 185:262–266
Zorn AM (2001) Wnt signalling: antagonistic Dickkopfs. Curr Biol 11:592–595
Glinka A, Wu W, Delius H et al (1998) Dickkopf-1 is a member of a new family of secreted proteins and functions in head induction. Nature 391:357–362
Mao J, Wu W, Li Y et al (2001) LDL-receptor-related protein 6 is a receptor for Dickkopf proteins. Nature 411:321–325
Mao B, Niehrs C (2003) Kremen2 modulates Dickkopf2 activity during Wnt/LRP6 signalling. Gene 302:179–183
Mao B, Wu W, Davidson G et al (2002) Kremen proteins are Dickkopf receptors that regulate Wnt/β-catenin signalling. Nature 417:664–667
Wu W, Glinka A, Delius H et al (2000) Mutual antagonism between dickkopf1 and dickkopf2 regulates Wnt/β-catenin signalling. Curr Biol 10:1611–1614
Brott BK, Sokol SY (2002) Regulation of Wnt/LRP signaling by distinct domains of Dickkopf proteins. Mol Cell Biol 22:6100–6110
Boyden LM, Mao J, Belsky J et al (2002) High bone density due to a mutation in LDL-receptor-related protein 5. N Engl J Med 346:1513–1521
Gong Y, Slee RB, Fukai N et al (2001) LDL receptor-related protein 5 (LRP5) affects bone accrual and eye development. Cell 107:513–523
Morvan F, Boulukos K, Clement-Lacroix P et al (2006) Deletion of a single allele of the Dkk1 gene leads to an increase in bone formation and bone mass. J Bone Miner Res 21:934–945
Li J, Sarosi I, Cattley RC et al (2006) Dkk1-mediated inhibition of Wnt signaling in bone results in osteopenia. Bone 39:754–766
Heiland GR, Zwerinal K, Baum W et al (2010) Neutralisation of Dkk-1 protects from systemic bone loss during inflammation and reduces sclerostin expression. Ann Rheum Dis 69:2152–2159
Weng LH, Wang CJ, Ko JY et al (2010) Control of Dkk-1 ameliorates chondrocyte apoptosis, cartilage destruction, and subchondral bone deterioration in osteoarthritic knees. Arthritis Rheum 62:1393–1402
Christodoulides C, Scarda A, Granzotto M et al (2006) WNT10B mutations in human obesity. Diabetologia 49:678–684
Cheng SL, Shao JS, Cai J et al (2008) Msx2 exerts bone anabolism via canonical Wnt signaling. J Biol Chem 283:20505–20522
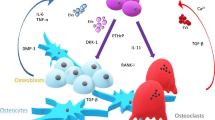
Diarra D, Stolina M, Polzer K et al (2007) Dickkopf-1 is a master regulator of joint remodeling. Nat Med 13:156–163
Liu Y, Tang W, Xie L et al (2014) Prognostic significance of dickkopf-1 overexpression in solid tumors: a meta-analysis. Tumour Biol 35:3145–3154
Zhou XL, Qin XR, Zhang XD et al (2010) Downregulation of Dickkopf-1 is responsible for high proliferation of breast cancer cells via losing control of Wnt/beta-catenin signaling. Acta Pharmacol Sin 31:202–210
Kim HY, Park JH, Won HY et al (2015) CBX7 inhibits breast tumorigenicity through DKK-1-mediated suppression of the Wnt/β-catenin pathway. FASEB J 29:300–313
Mikheey AM, Mikheeva SA, Maxwell JP et al (2008) Dickkopf-1 mediated tumor suppression in human breast carcinoma cells. Breast Cancer Res Treat 112:263–273
Agur Z, Kirnasovsky OU, Vasserman G et al (2011) Dickkopf1 regulates fate decision and drives breast cancer stem cells to differentiation: an experimentally supported mathematical model. PLoS One 6(9):e24225
Sato N, Yamabuki T, Takano A et al (2010) Wnt inhibitor Dickkopf-1 as a target for passive cancer immunotherapy. Cancer Res 70:5326–5336
Forget MA, Turcotte S, Beauseigle D et al (2007) The Wnt pathway regulator DKK1 is preferentially expressed in hormone-resistant breast tumours and in some common cancer types. Br J Cancer 96:646–653
Xu WH, Liu ZB, Yang C et al (2012) Expression of dickkopf-1 and beta-catenin related to the prognosis of breast cancer patients with triple negative phenotype. PLoS One. doi:10.1371/journal.pone.0037624
Zhou SJ, Zhou SR, Yang XQ et al (2014) Serum Dickkopf-1 expression level positively correlates with a poor prognosis in breast cancer. Diagn Pathol 9:161
Hideshima T, Mitsiades C, Tonon G et al (2007) Understanding multiple myloma pathogenesis in the bone marrow to identify new therapeutics targets. Nat Rev Cancer 7:585–595
Qiang YW, Chen Y, Stephens O et al (2008) Myeloma derived Dickkopf-1 disrupts Wnt-regulated osteoprotegerin and RANKL production by osteoblasts: a potential mechanism underlying osteolytic bone lesions in multiple myeloma. Blood 112:196–207
Voorzanger-Rousselot N, Journe F, Doriath V et al (2009) Assessment of circulating Dickkopf-1 with a new two-site immunoassay in healthy subjects and women with breast cancer and bone metastases. Calcif Tissue Int 84:348–354
Yoneda T, Williams PJ, Hiraga T et al (2001) A bone-seeking clone exhibits different biological properties from the MDA-MB-231 parental human breast cancer cells and a brain-seeking clone in vivo and in vitro. J Bone Miner Res 16:1486–1495
Voorzanger-Rousselot N, Goehrig D, Journe F et al (2007) Increased Dickkopf-1 expression in breast cancer bone metastases. Br J Cancer 97:964–970
Bu G, Lu W, Liu CC et al (2008) Breast cancer-derived Dickkopf1 inhibits osteoblast differentiation and osteoprotegerin expression: implication for breast cancer osteolytic bone metastases. Int J Cancer 123:1034–1042
Niida A, Hiroko T, Kasai M et al (2004) DKK1, a negative regulator of Wnt signaling, is a target of the β-catenin/TCF pathway. Oncogene 23:8520–8526
Menezes ME, Devine DJ, Shevde LA et al (2012) Dickkopf1: a tumor suppressor or metastasis promoter? Int J Cancer 130:1477–1483
Ellies DL, Viviano B, McCarthy J et al (2006) Bone density ligand. Sclerostin directly interacts with LRP5 but not LRP5G171V to modulate Wnt activity. Bone Miner 21:1738–1749
Kawai M, Mödder UI, Khosia S et al (2011) Emerging therapeutic opportunities for skeletal restoration. Nat Rev Drug Discov 10:141–156
Mendoza-Villanueva D, Zeef L, Shore P (2011) Metastatic breast cancer cells inhibit osteoblast differentiation through the Runx2-CBFβ-dependent expression of the Wnt antagonist, sclerostin. Breast Cancer Res 13:R106
van Bezooijen DL, Roelen BAJ, Visser A et al (2004) Sclerostin is an osteocyte-expressed negative regulator of bone formation, but not a classical BMP antagonist. J Exp Med 6:805–814
Rachner TD, Hadji P, Hofbauer LC (2012) Novel therapies in benign and malignant bone diseases. Pharmacol Ther 134:338–344
Pinzone JJ, Hall BM, Thudi NK et al (2009) The role of Dikkopf-1 in bone development, homeostasis, and disease. Blood 113:517–525
Rachner TD, Göbel A, Benad-Mehner P et al (2014) Dickkopf-1 as a mediator and novel target in malignant bone disease. Cancer Lett 346:172–177
Iyer SP, Beck JT, Ak Stewart et al (2014) A Phase IB multicentre dose-determination study of BHQ880 in combination with anti-myeloma therapy and zoledronic acid in patients with relapsed or refractory multiple myeloma and prior skeletal-related events. Br J Haematol 167:366–375
Rachner TD, Göbel A, Thiele S (2014) Dickkopf-1 is regulated by the mevalonate pathway in breast cancer. Breast Cancer Res. doi:10.1186/bcr3616
Kyvernitakis I, Rachner TD, Urbschat A et al (2014) Effect of aromatase inhibition on serum levels of sclerostin and dickkopf-1, bone turnover markers and bone mineral density in women with breast cancer. J Cancer Res Clin Oncol 140:1671–1680
Coleman RE, Guise TA, Lipton A et al (2008) Advancing treatment for metastatic bone cancer: consensus recommendations from the second Cambridge Conference. Clin Cancer Res 14:6387–6395
Funding
All authors declare that they have received no funding concerning this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Erich-Franz Solomayer holds a consultancy position at Novartis and Amgen and received compensation from Novartis, Amgen and Roche. Mariz Kasoha, Ingolf Juhasz-Boess, Daniel Herr and Jasmin Teresa Ney declare that they have no conflict of interests.
Ethical approval
No ethical approval was required.
Rights and permissions
About this article
Cite this article
Mariz, K., Ingolf, JB., Daniel, H. et al. The Wnt inhibitor dickkopf-1: a link between breast cancer and bone metastases. Clin Exp Metastasis 32, 857–866 (2015). https://doi.org/10.1007/s10585-015-9750-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10585-015-9750-1