Abstract
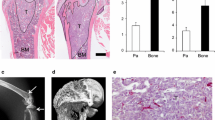
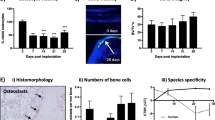
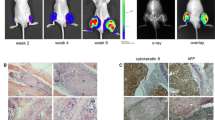
Breast cancer patients with advanced disease exhibit bone metastases, leading to the formation of osteolytic lesions for which the only currently available treatments are palliative. Here, we describe how we refined a mouse model of human breast cancer metastasis into bone, characterized its transcriptome and demonstrated its clinical relevance. Cells were selected from bone metastases caused by MDA-MB-231 cells after several in vivo passages, and engineered to express luciferase. Whole body bioluminescence live imaging indicated that the selected isogenic B02 clone was unique in its ability to form rapidly growing osteolytic bone metastases. B02 cells were detected as early as 10 days after tail vein injection, as opposed to 1 month after cardiac injection in other haematogenous models. Whole transcriptomic analysis identified 114 upregulated and 247 downregulated genes in B02 cells compared to the parental cells, several of which represent novel targets. In addition, there was a 50% overlap between the B02 signature and a recently described signature obtained from human breast cancer bone metastases. Consistent with the plasticity of an aggressive metastatic variant, 10% of the regulated genes are involved in proliferation, migration, invasion and angiogenesis. Strikingly, B02 cells also express osteoblast-specific genes, thus mimicking a process referred to as osteomimicry in the clinic. The B02 cells “human bone metastatic signature”, the expression of bone-specific genes, as well as the live imaging of this convenient model highlight its clinical relevance and usefulness during drug development.





Similar content being viewed by others
References
Green JR (2003) Antitumor effects of bisphosphonates. Cancer 97:840–847
Mundy GR (2002) Metastasis to bone: causes, consequences and therapeutic opportunities. Nat Rev Cancer 2:584–593
Kang Y, Siegel PM, Shu W et al (2003) A multigenic program mediating breast cancer metastasis to bone. Cancer Cell 3:537–549
Minn AJ, Kang Y, Serganova I et al (2005) Distinct organ-specific metastatic potential of individual breast cancer cells and primary tumors. J Clin Invest 115:44–55
Pécheur I, Peyruchaud O, Serre CM et al (2002) Integrin αvβ3 expression confers on tumor cells a greater propensity to metastasize to bone. FASEB J 16:1266–1268
Peyruchaud O, Serre CM, NicAmhlaoibh R et al (2003) Angiostatin inhibits bone metastasis formation in nude mice through a direct anti-osteoclastic activity. J Biol Chem 278:45826–45832
Theilhaber J, Bushnell S, Jackson A et al (2001) Bayesian estimation of fold-changes in the analysis of gene expression: the PFOLD algorithm. J Comput Biol 8:585–614
Roman-Roman S, Garcia T, Jackson A et al (2003) Identification of genes regulated during osteoblastic differentiation by genome-wide expression analysis of mouse calvaria primary osteoblasts in vitro. Bone 32:474–482
Schroeder TM, Jensen ED, Westendorf JJ (2005) Runx2: a master organizer of gene transcription in developing and maturing osteoblasts. Birth Defects Res C Embryo Today 75:213–225
Sharpless NE, DePinho RA (2006) The mighty mouse: genetically engineered mouse models in cancer drug development. Nat Rev Drug Discov 5:741–754
Steeg P (2006) Tumor metastasis: mechanistic insights and clinical challenges. Nat Med 12:895–904
Eccles SA (2005) Targeting key steps in metastatic tumour progression. Curr Opin Genet Dev 15:77–86
Rosol TJ, Tannehill-Gregg SH, LeRoy BE et al (2003) Animal model of bone metastasis. Cancer 97:748–757
Wetterwald A, van der Pluijm G, Que I et al (2002) Optical imaging of cancer metastasis to bone marrow: a mouse model of minimal residual disease. Am J Pathol 160:1143–1153
Kamb A (2005) What’s wrong with our cancer models. Nat Rev Drug Discov 4:161–165
Littlepage LE, Egeblad M, Werb Z (2005) Coevolution of cancer and stromal cellular responses. Cancer cell 7:499–500
Kuperwasser C, Dessain S, Bierbaum BE et al (2005) A mouse model of human breast cancer metastasis to human bone. Cancer Res 65:6130–6138
van’t Veer LJ, Dai H, van de Vijver MJ et al (2002) Gene expression profiling predicts clinical outcome of breast cancer. Nature 415:530–536
Bourguignon LY, Zhu H, Shao L et al (2000) Ankyrin-Tiam1 interaction promotes Rac1 signaling and metastatic breast tumor cell invasion and migration. J Cell Biol 150:177–191
van der Pluijm G, Sijmons B, Vloedgraven H et al (2001) Urokinase-receptor/integrin complexes are functionally involved in adhesion and progression of human breast cancer in vivo. Am J Pathol 159:971–982
Matsuura N, Puzon-McLaughlin W, Irie A et al (1996) Induction of experimental bone metastasis in mice by transfection of integrin alpha 4 beta 1 into tumor cells. Am J Pathol 148:55–61
Yoneda T, Sasaki A, Dunstan C et al (1997) Inhibition of osteolytic bone metastasis of breast cancer by combined treatment with the bisphosphonate ibandronate and tissue inhibitor of the matrix metalloproteinase-2. J Clin Invest 99:2509–2517
Yoneda T, Hiraga T (2005) Crosstalk between cancer cells and bone microenvironment in bone metastasis. BBRC 328:679–687
Hla T, Lee MJ, Ancellin N et al (2000) Sphingosine-1-phosphate signaling via the EDG-1 family of G-protein-coupled receptors. Ann N Y Acad Sci 905:16–24
Boucharaba A, Serre MC, Gres S et al (2004) Platelet-derived lysophosphatidic acid supports the progession of osteolytic bone metastases in breast cancer. J Clin Invest 114:1714–1725
Li A, Dubey S, Varney ML et al (2003) IL-8 directly enhanced endothelial cell survival, proliferation, and matrix metalloproteinases production and regulated angiogenesis. J Immunol 170:3369–3376
Bendre MS, Marguiles AG, Wasler B et al (2005) Tumor-derived interleukin-8 stimulates osteolysis independent of the receptor activator of nuclear factor-kappaB ligand pathway. Cancer Res 65:11001–11009
Kim SJ, Uehara H, Yazici S et al (2005) Modulation of bone microenvironment with zoledronate enhances the therapeutic effects of STI571 and paclitaxel against experimental bone metastasis of human prostate cancer. Cancer Res 65:3707–3715
Minn AJ, Gupta GP, Siegel PM et al (2005) Genes that mediate breast cancer metastasis to lung. Nature 28:518–524
Segal E, Friedman N, Koller D et al (2004) A module map showing conditional activity of expression modules in cancer. Nat Genet 36:1090–1098
Jones DH, Nakashima T, Sanchez OH et al (2006) Regulation of cancer cell migration and bone metastasis by RANKL. Nature 440:692–696
Berg RW, Leung E, Gough S et al (1999) Cloning and characterization of a novel beta integrin-related cDNA coding for the protein TIED (“ten beta integrin EGF-like repeat domains”) that maps to chromosome band 13q33: A divergent stand-alone integrin stalk structure. Genomics 56:169–178
Gettner SN, Kenyon C, Reichardt LF (1995) Characterization of beta pat-3 heterodimers, a family of essential integrin receptors in C. elegans. J Cell Biol 129:1127–1141
Hynes RO (2002) Integrins: bidirectional, allosteric signaling machines. Cell 110:673–687
Tucker GC (2003) Alpha v integrin inhibitors and cancer therapy. Curr Opin Investig Drugs 4:722–731
Bellahcene A, Bachelier R, Detry C et al (2006) Transcriptome analysis reveals an osteoblast-like phenotype for human osteotropic breast cancer cells. Breast Cancer Res Treat 101:135–148
Koeneman KS, Yeung F, Chung LW (1999) Osteomimetic properties of prostate cancer cells: a hypothesis supporting the predilection of prostate cancer metastasis and growth in the bone environment. Prostate 39:246–261
Pratap J, Javed A, Languino LR et al (2005) The Runx2 osteogenic transcription factor regulates matrix metalloproteinase 9 in bone metastatic cancer cells and controls cell invasion. Mol Cell Biol 25:8581–8591
Acknowledgment
This work was partially supported by a grant from the European Community contract No. LSHCCT2004-506049. www.metabre.org
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Garcia, T., Jackson, A., Bachelier, R. et al. A convenient clinically relevant model of human breast cancer bone metastasis. Clin Exp Metastasis 25, 33–42 (2008). https://doi.org/10.1007/s10585-007-9099-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10585-007-9099-1