Abstract
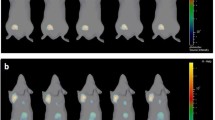
Bioluminescence imaging (BLI) has greatly facilitated the development of animal models of cancer, allowing sensitive detection of luciferase-expressing cancer cells in living mice. Previous efforts characterizing such models have involved small numbers of animals, limiting understanding of their performance features. We employed BLI to serially image the growth and distribution of a prostate cancer cell line, 22Rv1, after intracardiac injection into scid mice (n = 85). This approach models hematogenous dissemination of cancer cells and allows inquiry of the process of metastatic colonization at various organ sites, although accurately injecting cancer cells into the left ventricle remains challenging. Therefore, to predict injection success we measured the ratio of the thoracic bioluminescence signal to the whole body bioluminescence signal (T/WB ratio) immediately following intracardiac injection. A T/WB ratio less than 0.50 predicted the development of tumors outside of the thoracic cavity while a T/WB greater than 0.50 predicted the development of tumors entirely within the thoracic cavity, suggestive of a failed injection. Progressive tumor growth was quantified using BLI. Tumors colonized multiple organ sites including bone, liver, and adrenal glands resembling the spectrum of metastases in autopsy studies of patients with prostate cancer. Tumors growing in bone exhibited mixed osteolytic and osteoblastic features, eliciting a spiculated periosteal response. With the ability to more accurately predict injection success, we can now monitor efficacy of intracardiac injections facilitating the performance of this model.
Similar content being viewed by others
Abbreviations
- BLI:
-
bioluminescence imaging
- TO:
-
thoracic only
- T+:
-
thoracic plus
- ET:
-
extrathoracic
- T/WB:
-
thoracic/whole body
- TI:
-
trypsin inhibitor
- ROI:
-
region of interest
References
Jemal A, Murray T, Ward E et al (2005) Cancer statistics, 2005. CA Cancer J Clin 55:10–30
Scher HI (2003) Prostate carcinoma: defining therapeutic objectives and improving overall outcomes. Cancer 97:758–771
Bubendorf L, Schopfer A, Wagner U et al (2000) Metastatic patterns of prostate cancer: an autopsy study of 1,589 patients. Hum Pathol 31:578–583
Shah RB, Mehra R, Chinnaiyan AM et al (2004) Androgen-independent prostate cancer is a heterogeneous group of diseases: lessons from a rapid autopsy program. Cancer Res 64:9209–9216
Rubin MA, Putzi M, Mucci N et al (2000) Rapid (“warm”) autopsy study for procurement of metastatic prostate cancer. Clin Cancer Res 6:1038–1045
Roudier MP, True LD, Higano CS et al (2003) Phenotypic heterogeneity of end-stage prostate carcinoma metastatic to bone. Hum Pathol 34:646–653
Tantivejkul K, Kalikin LM, Pienta KJ (2004) Dynamic process of prostate cancer metastasis to bone. J Cell Biochem 91:706–717
Edlund M, Sung SY, Chung LW (2004) Modulation of prostate cancer growth in bone microenvironments. J Cell Biochem 91:686–705
Roy-Burman P, Wu H, Powell WC et al (2004) Genetically defined mouse models that mimic natural aspects of human prostate cancer development. Endocr Relat Cancer 11:225–254
Winter SF, Cooper AB, Greenberg NM (2003) Models of metastatic prostate cancer: a transgenic perspective. Prostate Cancer Prostatic Dis 6:204–211
Singh AS, Figg WD (2005) In vivo models of prostate cancer metastasis to bone. J Urol 174:820–826
Stephenson RA (1992) Metastatic model for human prostate cancer using orthotopic implantation in nude mice. J Natl Cancer Inst 84:951–957
Fu X, Herrera H, Hoffman RM (1992) Orthotopic growth and metastasis of human prostate carcinoma in nude mice after transplantation of histologically intact tissue. Int J␣Cancer 52:987–990
Gingrich JR, Barrios RJ, Morton RA et al (1996) Metastatic prostate cancer in a transgenic mouse. Cancer Res 56:4096–4102
Wang S, Gao J, Lei Q et al (2003) Prostate-specific deletion of the murine Pten tumor suppressor gene leads to metastatic prostate cancer. Cancer Cell 4:209–221
Nemeth JA, Harb JF, Barroso U Jr et al (1999) Severe combined immunodeficient-hu model of human prostate cancer metastasis to human bone. Cancer Res 59:1987–1993
Corey E, Quinn JE, Bladou F et al (2002) Establishment and characterization of osseous prostate cancer models: intra-tibial injection of human prostate cancer cells. Prostate 52:20–33
Lee Y, Schwarz E, Davies M et al (2003) Differences in the cytokine profiles associated with prostate cancer cell induced osteoblastic and osteolytic lesions in bone. J Orthop Res 21:62–72
Wu T, Sikes RA, Cui Q et al (1998) Establishing human prostate cancer cell xenografts in bone: induction of osteoblastic reaction by prostate-specific antigen-presenting tumors in athymic and scid/bg mice using LNCaP and␣lineage-derived metastatic sublines. Int J Cancer 77:887–894
Henry MD, Silva MD, Wen S et al (2005) Spiculated periosteal response induced by intraosseous injection of 22Rv1 prostate cancer cells resembles subset of bone metastases in prostate cancer patients. Prostate 65:347–354
Blomme EA, Dougherty KM, Pienta KJ et al (1999) Skeletal metastasis of prostate adenocarcinoma in rats: morphometric analysis and role of parathyroid hormone-related protein. Prostate 39:187–197
Keller ET, Brown J (2004) Prostate cancer bone metastases promote both osteolytic and osteoblastic activity. J Cell Biochem 91:718–729
Schneider A, Kalikin LM, Mattos AC et al (2005) Bone turnover mediates preferential localization of prostate cancer in the skeleton. Endocrinology 146:1727–1736
Chishima T, Miyagi Y, Wang X et al (1997) Cancer invasion and micrometastasis visualized in live tissue by green fluorescent protein expression. Cancer Res 57:2042–2047
Contag CH, Spilman SD, Contag PR et al (1997) Visualizing gene expression in living mammals using a bioluminescent reporter. Photochem Photobiol 66:523–531
Choy G, O’Connor S, Diehn FE et al (2003) Comparison of non-invasive fluorescent and bioluminescent small animal optical imaging. Biotechniques 35:1022–1026, 1028–1030
Ray P, De A, Min JJ et al (2004) Imaging tri-fusion multimodality reporter gene expression in living subjects. Cancer Res 64:1323–1330
Troy T, Jekic-McMullen D, Sambucetti L et al (2004) Quantitative comparison of the sensitivity of detection of fluorescent and bioluminescent reporters in animal models. Mol Imaging 3:9–23
Kalikin LM, Schneider A, Thakur MA et al (2003) In vivo visualization of metastatic prostate cancer and quantitation of disease progression in immunocompromised mice. Cancer Biol Ther 2:656–660
Rosol TJ, Tannehill-Gregg SH, LeRoy BE et al (2003) Animal models of bone metastasis. Cancer 97:748–757
Scatena CD, Hepner MA, Oei YA et al (2004) Imaging of bioluminescent LNCaP-luc-M6 tumors: a new animal model for the study of metastatic human prostate cancer. Prostate 59:292–303
Jenkins DE, Yu SF, Hornig YS et al (2003) In vivo monitoring of tumor relapse and metastasis using bioluminescent PC-3M-luc-C6 cells in murine models of human prostate cancer. Clin Exp Metastasis 20:745–756
Rehemtulla A, Stegman LD, Cardozo SJ et al (2000) Rapid and quantitative assessment of cancer treatment response using in vivo bioluminescence imaging. Neoplasia 2:491–495
Jenkins DE, Oei Y, Hornig YS et al (2003) Bioluminescent imaging (BLI) to improve and refine traditional murine models of tumor growth and metastasis. Clin Exp Metastasis 20:733–744
Sramkoski RM, Pretlow TG 2nd, Giaconia JM et al (1999) A new human prostate carcinoma cell line, 22Rv1. In Vitro Cell Dev Biol Anim 35:403–409
Arguello F, Baggs RB, Frantz CN (1988) A murine model of experimental metastasis to bone and bone marrow. Cancer Res 48:6876–6881
Lehr JE, Pienta KJ (1998) Preferential adhesion of prostate cancer cells to a human bone marrow endothelial cell line. J Natl Cancer Inst 90:118–123
Simpson MA, Reiland J, Burger SR et al (2001) Hyaluronan synthase elevation in metastatic prostate carcinoma cells correlates with hyaluronan surface retention, a prerequisite for rapid adhesion to bone marrow endothelial cells. J Biol Chem 276:17949–17957
Dimitroff CJ, Lechpammer M, Long-Woodward D et al (2004) Rolling of human bone-metastatic prostate tumor cells on human bone marrow endothelium under shear flow is mediated by E-selectin. Cancer Res 64:5261–5269
Glinsky VV, Glinsky GV, Rittenhouse-Olson K et al (2001) The role of Thomsen–Friedenreich antigen in adhesion of human breast and prostate cancer cells to the endothelium. Cancer Res 61:4851–4857
Paget S (1889) The distribution of secondary growths in cancer of the breast. Lancet 1:99–101
Batson OV (1995) The function of the vertebral veins and their role in the spread of metastases. 1940. Clin Orthop Relat Res 312:4–9
Angelucci A, Gravina GL, Rucci N et al (2004) Evaluation of metastatic potential in prostate carcinoma: an in vivo model. Int J Oncol 25:1713–1720
Pettaway CA, Pathak S, Greene G et al (1996) Selection of highly metastatic variants of different human prostatic carcinomas using orthotopic implantation in nude mice. Clin Cancer Res 2:1627–1636
Byers HR, Etoh T, Lee KW et al (1993) Organ-specific metastases in immunodeficient mice injected with human melanoma cells: a quantitative pathological analysis. Melanoma Res 3:247–253
Fodstad O, Kjonniksen I, Aamdal S et al (1988) Extrapulmonary, tissue-specific metastasis formation in nude mice injected with FEMX-I human melanoma cells. Cancer Res 48:4382–4388
Chirgwin JM, Mohammad KS, Guise TA (2004) Tumor-bone cellular interactions in skeletal metastases. J Musculoskelet Neuronal Interact 4:308–318
Bloom RA, Libson E, Husband JE et al (1987) The periosteal sunburst reaction to bone metastases. A literature review and report of 20 additional cases. Skeletal Radiol 16:629–634
Hove B, Gyldensted C (1990) Spiculated vertebral metastases from prostatic carcinoma. Report of first two cases. Neuroradiology 32:337–339
Lehrer HZ, Maxfield WS, Nice CM (1970) The periosteal “sunburst” pattern in metastatic bone tumors. Am J Roentgenol Radium Ther Nucl Med 108:154–161
Wyche LD, de Santos LA (1978) Spiculated periosteal reaction in metastatic disease resembling osteosarcoma. Orthopedics 1:215–221
Acknowledgements
We would like to thank members of the Henry laboratory for comments on the manuscript; Edward Solin and the University of Iowa College of Medicine Core Pathology lab for assistance with histology and Jim Olson in the University of Iowa College of Medicine Department of Radiology for digitizing X-ray images.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Drake, J.M., Gabriel, C.L. & Henry, M.D. Assessing Tumor Growth and Distribution in a Model of Prostate Cancer Metastasis using Bioluminescence Imaging. Clin Exp Metastasis 22, 674–684 (2005). https://doi.org/10.1007/s10585-006-9011-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10585-006-9011-4