Abstract
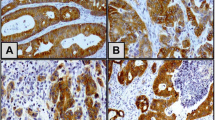
c-Met, the receptor of hepatocyte growth factor is known to be responsible for the motility and mitogenesis of epithelial cells including cancer cells. To investigate the significance of c-Met expression in human colorectal cancer (CRC), total cellular protein, extracted from 130 CRCs were examined by Western blot analysis. The signal was quantitated by ChemiImager™ 4000 Low Light Imaging System. c-Met expression was analyzed as the ratio of tumor to matched normal tissue (T/N) and expressed as fold-increase. The cellular localization of c-Met was assessed by immunohistochemistry. The T/N fold increase of c-Met varied from 0.2 to 10.7 with a mean of 3.41 ± 0.23 (mean ± SE). 69% primary CRC showed overexpression (T/N >2.0) of c-Met. Significantly higher c-Met levels were found in CRC with blood vessel invasion (P = 0.04), and in advanced stage (P = 0.04). No relationship was noted between c-Met expression and age, tumor size, location, differentiation. C-Met immunoreactivity was observed in the membrane and cytoplasm of cancer cells. Positive staining of endothelial cells of blood vessels within normal submucosa and tumor was also evident. C-Met protein is expressed at levels significantly higher than adjacent mucosa in most primary adenocarcinomas of the colon. Our results support an important role for c-Met in human CRC progression and metastasis.
Similar content being viewed by others
Abbreviations
- BVI:
-
blood vessel invasion
- c-Met:
-
receptor of hepatocyte growth factor
- CRC:
-
colorectal cancer
- HGF:
-
hepatocyte growth factor
- LVI:
-
lymphatic vessel invasion
- MMPs:
-
matrix metalloproteinases
- SF:
-
scatter factor
- u-PA:
-
urokinase-type plasminogen activator
References
Jemal A, Murray T, Samuels A et al. Cancer Statistics, 2003. CA Cancer J Clin 2003; 53: 5–26.
Maulik G, Shrikhande A, Kijima T et al. Role of the hepatocyte growth factor receptor, c-Met, in oncogenesis and potential for therapeutic inhibition. Cytokine Growth Factor Rev 2002; 13: 41–59.
To CT, Tsao MS. The roles of hepatocyte growth factor/scatter factor and met receptor in human cancers (Review). Oncol Rep 1998; 5: 1013–24.
Jiang W, Hiscox S, Matsumoto K et al. Hepatocyte growth factor/scatter factor, its molecular, cellular and clinical implications in cancer. Crit Rev Oncol Hematol 1999; 29: 209–48.
Cooper CS, Park M, Blair DG et al. Molecular cloning of a new transforming gene from a chemically transformed human cell line. Nature 1984; 311: 29–33.
Iyer A, Kmiecik TE, Park M et al. Structure, tissue-specific expression, and transforming activity of the mouse met protooncogene. Cell Growth Differ 1990; 1: 87–95.
Giordano S, Ponzetto C, Di Renzo MF et al. Tyrosine kinase receptor indistinguishable from the c-met protein. Nature 1989; 339: 155–6.
Vande Woude GF, Jeffers M, Cortner J et al. Met-HGF/SF: tumorigenesis, invasion and metastasis. Ciba Found Symp 1997; 212: 119–30.
Takayama H, LaRochelle WJ, Sharp R et al. Diverse tumorigenesis associated with aberrant development in mice overexpressing hepatocyte growth factor/scatter factor. Proc Natl Acad Sci USA 1997; 94: 701–6.
Jeffers M, Fiscella M, Webb CP et al. The mutationally activated Met receptor mediates motility and metastasis. Proc Natl Acad Sci USA 1998; 95: 14417–22.
Wang R, Ferrell LD, Faouzi S et al. Activation of the Met receptor by cell attachment induces and sustains hepatocellular carcinomas in transgenic mice. J. Cell Biol. 2001; 153: 1023–34.
Schmidt L, Duh FM, Chen F et al. Germline and somatic mutations in the tyrosine kinase domain of the MET proto-oncogene in papillary renal carcinomas. Nat Genet 1997; 16: 68–73.
Park WS, Dong SM, Kim SY et al. Somatic mutations in the kinase domain of the Met/hepatocyte growth factor receptor gene in childhood hepatocellular carcinomas. Cancer Res 1999; 59: 307–10.
Di Renzo MF, Olivero M, Martone T et al. Somatic mutations of the MET oncogene are selected during metastatic spread of human HNSC carcinomas. Oncogene 2000; 19: 1547–55.
Liu C, Park M, Tsao MS. Overexpression of c-met proto-oncogene but not epidermal growth factor receptor or c-erbB-2 in primary human colorectal carcinomas. Oncogene 1992; 7: 181–5.
Di Renzo MF, Olivero M, Giacomini A et al. Overexpression and amplification of the met/HGF receptor gene during the progression of colorectal cancer. Clin Cancer Res 1995; 1: 147–54.
Hiscox SE, Hallett MB, Puntis MC et al. Expression of the HGF/SF receptor, c-met, and its ligand in human colorectal cancers. Cancer Invest 1997; 15: 513–21.
Di Renzo MF, Narsimhan RP, Olivero M et al. Expression of the Met/HGF receptor in normal and neoplastic human tissues. Oncogene 1991; 6: 1997–2003.
Kiehne K, Herzig KH, Folsch UR. c-met expression in pancreatic cancer and effects of hepatocyte growth factor on pancreatic cancer cell growth. Pancreas 1997; 15: 35–40.
Ramirez R, Hsu D, Patel A et al. Over-expression of hepatocyte growth factor/scatter factor (HGF/SF) and the HGF/SF receptor (cMET) are associated with a high risk of metastasis and recurrence for children and young adults with papillary thyroid carcinoma. Clin Endocrinol (Oxf) 2000; 53: 635–44.
Tuck A, Park M, Sterns E et al. Coexpression of hepatocyte growth factor and receptor (Met) in human breast carcinoma. Am J Pathol 1996; 148: 225–32.
Beviglia L, Matsumoto K, Lin CS et al. Expression of the c-Met/HGF receptor in human breast carcinoma: correlation with tumor progression. Int J Cancer 1997; 74: 301–9.
Huntsman D, Resau JH, Klineberg E et al. Comparison of c-met expression in ovarian epithelial tumors and normal epithelia of the female reproductive tract by quantitative laser scan microscopy. Am J Pathol 1999; 155: 343–8.
Wagatsuma S, Konno R, Sato S et al. Tumor angiogenesis, hepatocyte growth factor, and c-Met expression in endometrial carcinoma. Cancer 1998; 82: 520–30.
Humphrey PA, Zhu X, Zarnegar R et al. Hepatocyte growth factor and its receptor (c-MET) in prostatic carcinoma. Am J Pathol 1995; 147: 386–96.
Hu YC, Lam KY, Law S et al. Profiling of differentially expressed cancer-related genes in esophageal squamous cell carcinoma (ESCC) using human cancer cDNA arrays: Overexpression of oncogene MET correlates with tumor differentiation in ESCC. Clin Cancer Res 2001; 7: 3519–25.
Koochekpour S, Jeffers M, Rulong S et al. Met and hepatocyte growth factor/scatter factor expression in human gliomas. Cancer Res 1997; 57: 5391–8.
Trusolino L, Pugliese L, Comoglio PM. Interactions between scatter factors and their receptors: hints for therapeutic applications. Faseb J 1998; 12: 1267–80.
Fazekas K, Csuka O, Koves I et al. Experimental and clinicopathologic studies on the function of the HGF. Clin Exp Metast 2000; 18: 639–49.
Otte JM, Schmitz F, Kiehne K et al. Functional expression of HGF and its receptor in human colorectal cancer. Digestion 2000; 61: 237–46.
Takeuchi H, Bilchik A, Saha S et al. c-MET expression level in primary colon cancer: A predictor of tumor invasion and lymph node metastases. Clin Cancer Res 2003; 9: 1480–8.
Herynk MH, Stoeltzing O, Reinmuth N et al. Down-regulation of c-Met inhibits growth in the liver of human colorectal carcinoma cells. Cancer Res 2003; 63: 2990–6.
Herynk M, Tsan R, Radinsky R et al. Activation of c-Met in colorectal carcinoma cells leads to constitutive association of tyrosine-phosphorylated beta-catenin. Clin Exp Metast 2003; 20: 291–300.
Rubin JS, Bottaro DP, Aaronson SA. Hepatocyte growth factor/scatter factor and its receptor, the c-met proto-oncogene product. Biochim Biophys Acta 1993; 1155: 357–71.
Jeffers M, Schmidt L, Nakaigawa N et al. Activating mutations for the Met tyrosine kinase receptor in human cancer. PNAS 1997; 94: 11445–50.
Ghoussoub RA, Dillon DA, D’Aquila T et al. Expression of c-met is a strong independent prognostic factor in breast carcinoma. Cancer 1998; 82: 1513–20.
Di Renzo MF, Olivero M, Katsaros D et al. Overexpression of the Met/HGF receptor in ovarian cancer. Int J Cancer 1994; 58: 658–62.
Inoue K, Karashima T, Chikazawa M et al. Overexpression of c-met proto-oncogene associated with chromophilic renal cell carcinoma with papillary growth. Virchows Arch 1998; 433: 511–5.
Blumberg D, Paty PB, Picon AI et al. Stage I rectal cancer: Identification of high-risk patients. J Am Coll Surg 1998; 186: 574–9; discussion 579-80.
Minsky BD, Mies C, Recht A et al. Resectable adenocarcinoma of the rectosigmoid and rectum. II. The influence of blood vessel invasion. Cancer 1988; 61: 1417–24.
Besser D, Bardelli A, Didichenko S et al. Regulation of the urokinase-type plasminogen activator gene by the oncogene Tpr-Met involves GRB2. Oncogene 1997; 14: 705–11.
Kermorgant S, Aparicio T, Dessirier V et al. Hepatocyte growth factor induces colonic cancer cell invasiveness via enhanced motility and protease overproduction. Evidence for PI3 kinase and PKC involvement. Carcinogenesis 2001; 22: 1035–42.
Silvagno F, Follenzi A, Arese M et al. In vivo activation of Met tyrosine kinase by heterodimeric hepatocyte growth factor molecule promotes angiogenesis. Arterioscler Thromb Vasc Biol. 1995; 15: 1857–65.
Schmidt NO, Westphal M, Hagel C et al. Levels of vascular endothelial growth factor, hepatocyte growth factor/scatter factor and basic fibroblast growth factor in human gliomas and their relation to angiogenesis. Int J Cancer 1999; 84: 10–18.
Dong G, Chen Z, Li Z-Y et al. Hepatocyte growth factor/scatter factor-induced activation of MEK and PI3K signal pathways contributes to expression of proangiogenic cytokines interleukin-8 and vascular en-dothelial growth factor in head and neck squamous cell carcinoma. Cancer Res 2001; 61: 5911–8.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zeng, Z., Weiser, M.R., D’Alessio, M. et al. Immunoblot analysis of c-Met expression in human colorectal cancer: Overexpression is associated with advanced stage cancer. Clin Exp Metastasis 21, 409–417 (2004). https://doi.org/10.1007/s10585-005-1617-4
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s10585-005-1617-4