Abstract
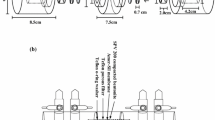
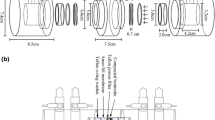
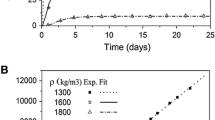
The main aim of this study was to investigate diffusion of technetium 99Tc under different conditions. Because technetium represents one of the most dangerous fission products due to its very long halftime and high mobility in aerobic conditions diffusion experiments of technetium (as 99TcO −4 anion) in Czech bentonite from Rokle locality have been carried out. For performance and evaluation of experiments the through-diffusion method was chosen and apparent (Da) and effective (De) diffusion coefficients were evaluated. The effects of particle mesh-size, dry bulk density and aerobic or anaerobic conditions on diffusion were studied. In the presence of oxygen, technetium occurs in oxidation state VII, as an anion, soluble and mobile in the environment. However, under reducing conditions it occurs in a lower oxidation states, mainly as insoluble oxides or hydroxides. Aerobic experiments were carried out under laboratory conditions and anaerobic experiments were performed in a nitrogen atmosphere in a glove box, to simulate the real underground conditions.
Similar content being viewed by others
References
Vinšová H. et al.: J. Radioanal. Nucl. Chem. (2004) 407.
Riebe B., Bors J., and Dultz St.: J. Contam Hydrol. (2001) 255.
Molera M. et al.: Appl. Clay Sci. (2003) 69.
Kozaki T. et al.: J. Nucl.Mater. (1999) 265.
Kozaki T. et al.: Eng. Geol. (2005) 246.
Sawatski N. G. and Oscarson D. W.: Water, Air and Soil Pollution (1991) 449.
Szántó Zs. et al.: J. Radioanal. Nucl. Chem. (2002) 133.
Vopálka D., Filipská H. and Vokál A.: Mat. Res. Soc. Symp. Proc. (2005) (accepted).
Kuroda Y. et al.: Materials Research Society Scientific Basis for Nuclear Waste Management (1997) 909.
Chen F., Burns P.C. and Ewing R.C.: J. Nucl. Mater. 278 (2000) 225.
Jansson M. and Eriksen T.E.: J Contam. Hydrol. (2004) 183.
Wang X. and Tao Z.: J. Radioanal. Nucl. Chem. (2004) 3
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Večerník, P., Jedináková-Křížová, V. Diffusion of 99-technetium in compacted bentonite under aerobic and anaerobic conditions. Czech J Phys 56, D665–D672 (2006). https://doi.org/10.1007/s10582-006-1081-7
Issue Date:
DOI: https://doi.org/10.1007/s10582-006-1081-7