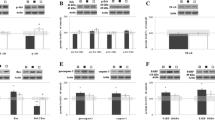
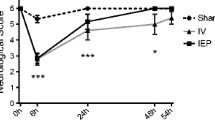
Abstract
Sustained activation of pro-apoptotic signaling due to a sudden and prolonged disturbance of cerebral blood circulation governs the neurodegenerative processes in prefrontal cortex (PFC) of rats whose common carotid arteries are permanently occluded. The adequate neuroprotective therapy should minimize the activation of toxicity pathways and increase the activity of endogenous protective mechanisms. Several neuroprotectants have been proposed, including progesterone (P4). However, the underlying mechanism of its action in PFC following permanent bilateral occlusion of common carotid arteries is not completely investigated. We, thus herein, tested the impact of post-ischemic P4 treatment (1.7 mg/kg for seven consecutive days) on previously reported aberrant neuronal morphology and amount of DNA fragmentation, as well as the expression of progesterone receptors along with the key elements of Akt/Erk/eNOS signal transduction pathway (Bax, Bcl-2, cytochrome C, caspase 3, PARP, and the level of nitric oxide). The obtained results indicate that potential amelioration of histological changes in PFC might be associated with the absence of activation of Bax/caspase 3 signaling cascade and the decline of DNA fragmentation. The study also provides the evidence that P4 treatment in repeated regiment of administration might be effective in neuronal protection against ischemic insult due to re-establishment of the compromised action of Akt/Erk/eNOS-mediated signaling pathway and the upregulation of progesterone receptors.



Similar content being viewed by others
References
Allen RS, Sayeed I, Oumarbaeva Y et al (2016) Progesterone treatment shows greater protection in brain vs. retina in a rat model of middle cerebral artery occlusion: progesterone receptor levels may play an important role. Restor Neurol Neurosci 34:947–963. https://doi.org/10.3233/RNN-160672
Andrabi SS, Parvez S, Tabassum H (2017) Neurosteroids and ischemic stroke: progesterone a promising agent in reducing the brain injury in ischemic stroke. J Environ Pathol Toxicol Oncol 36:191–205. https://doi.org/10.1615/JEnvironPatholToxicolOncol.2017017156
Boyadjieva NI, Andreeva-Gateva P (2011) Effects of kainic acid on glutatione and nitrite in rat hippocampus. J IMAB 17(1):149–151. https://doi.org/10.5272/jimab.2011171.149
Brinton RD, Thompson RF, Foy MR et al (2008) Progesterone receptors: form and function in brain. Front Neuroendocrinol 29:313–339. https://doi.org/10.1016/j.yfrne.2008.02.001
Cai W, Zhu Y, Furuya K et al (2008) Two different molecular mechanisms underlying progesterone neuroprotection against ischemic brain damage. Neuropharmacology 55:127–138. https://doi.org/10.1016/j.neuropharm.2008.04.023
Cargnello M, Roux PP (2011) Activation and function of the MAPKs and their substrates, the MAPK-activated protein kinases. Microbiol Mol Biol Rev 75:50–83. https://doi.org/10.1128/MMBR.00031-10
Casas S, García S, Cabrera R et al (2011) Progesterone prevents depression-like behavior in a model of Parkinson’s disease induced by 6-hydroxydopamine in male rats. Pharmacol Biochem Behav 99:614–618. https://doi.org/10.1016/j.pbb.2011.06.012
Cechetti F, Pagnussat AS, Worm PV et al (2012) Chronic brain hypoperfusion causes early glial activation and neuronal death, and subsequent long-term memory impairment. Brain Res Bull 87:109–116. https://doi.org/10.1016/j.brainresbull.2011.10.006
Chang C-M, Su Y-F, Chang C-Z et al (2014) Progesterone attenuates experimental subarachnoid hemorrhage-induced vasospasm by upregulation of endothelial nitric oxide synthase via Akt signaling pathway. Biomed Res Int 2014:1–6. https://doi.org/10.1155/2014/207616
Chen C-A, Druhan LJ, Varadharaj S et al (2008) Phosphorylation of endothelial nitric-oxide synthase regulates superoxide generation from the enzyme. J Biol Chem 283:27038–27047. https://doi.org/10.1074/jbc.M802269200
Chen Z, Mou R, Feng D et al (2017) The role of nitric oxide in stroke. Med Gas Res 7:194. https://doi.org/10.4103/2045-9912.215750
Clark LR, Berman SE, Rivera-Rivera LA et al (2017) Macrovascular and microvascular cerebral blood flow in adults at risk for Alzheimer’s disease. Alzheimer’s Dementia Diagn, Assess Dis Monit 7:48–55. https://doi.org/10.1016/j.dadm.2017.01.002
De Jong GI, Farkas E, Stienstra CM et al (1999) Cerebral hypoperfusion yields capillary damage in the hippocampal CA1 area that correlates with spatial memory impairment. Neuroscience 91:203–210. https://doi.org/10.1016/S0306-4522(98)00659-9
de la Torre JC (2002a) Alzheimer disease as a vascular disorder: nosological evidence. Stroke 33:1152–1162. https://doi.org/10.1161/01.STR.0000014421.15948.67
de la Torre JC (2002b) Alzheimer’s disease: how does it start? J Alzheimer’s Dis 4:497–512. https://doi.org/10.3233/JAD-2002-4606
Deutsch ER, Espinoza TR, Atif F et al (2013) Progesterone’s role in neuroprotection, a review of the evidence. Brain Res 1530:82–105. https://doi.org/10.1016/j.brainres.2013.07.014
Djebaili M, Hoffman SW, Stein DG (2004) Allopregnanolone and progesterone decrease cell death and cognitive deficits after a contusion of the rat pre-frontal cortex. Neuroscience 123:349–359
Djebaili M, Guo Q, Pettus EH et al (2005) The Neurosteroids progesterone and allopregnanolone reduce cell death, gliosis, and functional deficits after traumatic brain injury in rats. J Neurotrauma 22:106–118. https://doi.org/10.1089/neu.2005.22.106
Drakulić D, Veličković N, Stanojlović M et al (2013) Low-dose dexamethasone treatment promotes the pro-survival signalling pathway in the adult rat prefrontal cortex. J Neuroendocrinol 25:605–616. https://doi.org/10.1111/jne.12037
Duckles SP, Miller VM (2010) Hormonal modulation of endothelial NO production. Pflügers Archiv 459:841–851. https://doi.org/10.1007/s00424-010-0797-1
Farkas E, Luiten PGM, Bari F (2007) Permanent, bilateral common carotid artery occlusion in the rat: a model for chronic cerebral hypoperfusion-related neurodegenerative diseases. Brain Res Rev 54:162–180. https://doi.org/10.1016/j.brainresrev.2007.01.003
Forstermann U, Sessa WC (2012) Nitric oxide synthases: regulation and function. Eur Heart J 33:829–837. https://doi.org/10.1093/eurheartj/ehr304
Friguls B, Petegnief V, Justicia C et al (2002) Activation of ERK and Akt signalling in focal cerebral ischemia: modulation by TGF-α and involvement of NMDA receptor. Neurobiol Dis 11:443–456. https://doi.org/10.1006/nbdi.2002.0553
Geddes RI, Sribnick EA, Sayeed I, Stein DG (2014) Progesterone treatment shows benefit in a pediatric model of moderate to severe bilateral brain injury. PLoS ONE 9:e87252. https://doi.org/10.1371/journal.pone.0087252
Ghoumari AM, Ibanez C, El-Etr M et al (2003) Progesterone and its metabolites increase myelin basic protein expression in organotypic slice cultures of rat cerebellum. J Neurochem 86:848–859
Giangrande PH, McDonnell DP (1999) The A and B isoforms of the human progesterone receptor: two functionally different transcription factors encoded by a single gene. Recent Prog Horm Res 54:291–313
Gibson CL, Murphy SP (2004) Progesterone enhances functional recovery after middle cerebral artery occlusion in male mice. J Cereb Blood Flow Metab 24:805–813. https://doi.org/10.1097/01.WCB.0000125365.83980.00
Guennoun R, Meffre D, Labombarda F et al (2008) The membrane-associated progesterone-binding protein 25-Dx: expression, cellular localization and up-regulation after brain and spinal cord injuries. Brain Res Rev 57:493–505. https://doi.org/10.1016/j.brainresrev.2007.05.009
Guerra-Araiza C, Amorim MAR, Pinto-Almazán R et al (2009) Regulation of the phosphoinositide-3 kinase and mitogen-activated protein kinase signaling pathways by progesterone and its reduced metabolites in the rat brain. J Neurosci Res 87:470–481. https://doi.org/10.1002/jnr.21848
Hanna RN, Daly SCJ, Pang Y et al (2010) Characterization and expression of the nuclear progestin receptor in zebrafish gonads and brain1. Biol Reprod 82:112–122. https://doi.org/10.1095/biolreprod.109.078527
Hossain M, Qadri SM, Liu L (2012) Inhibition of nitric oxide synthesis enhances leukocyte rolling and adhesion in human microvasculature. J Inflamm 9:28. https://doi.org/10.1186/1476-9255-9-28
Huang Z, Huang PL, Ma J et al (1996) Enlarged infarcts in endothelial nitric oxide synthase knockout mice are attenuated by nitro-L-arginine. J Cereb Blood Flow Metab 16:981–987. https://doi.org/10.1097/00004647-199609000-00023
Hussain R, El-Etr M, Gaci O et al (2011) Progesterone and nestorone facilitate axon remyelination: a role for progesterone receptors. Endocrinology 152:3820–3831. https://doi.org/10.1210/en.2011-1219
Ishrat T, Sayeed I, Atif F et al (2012) Progesterone is neuroprotective against ischemic brain injury through its effects on the phosphoinositide 3-kinase/protein kinase B signaling pathway. Neuroscience 210:442–450. https://doi.org/10.1016/j.neuroscience.2012.03.008
Ito Y, Ohkubo T, Asano Y et al (2010) Nitric oxide production during cerebral ischemia and reperfusion in eNOS- and nNOS-knockout mice. Curr Neurovasc Res 7:23–31
Kaur P, Jodhka PK, Underwood WA et al (2007) Progesterone increases brain-derived neuroptrophic factor expression and protects against glutamate toxicity in a mitogen-activated protein kinase- and phosphoinositide-3 kinase-dependent manner in cerebral cortical explants. J Neurosci Res 85:2441–2449. https://doi.org/10.1002/jnr.21370
Khorram O, Han G (2009) Influence of progesterone on endometrial nitric oxide synthase expression. Fertil Steril 91:2157–2162. https://doi.org/10.1016/j.fertnstert.2008.05.019
Kim YM, Kim TH, Seol DW et al (1998) Nitric oxide suppression of apoptosis occurs in association with an inhibition of Bcl-2 cleavage and cytochrome C release. J Biol Chem 273:31437–31441
Kim YM, Bombeck CA, Billiar TR (1999) Nitric oxide as a bifunctional regulator of apoptosis. Circ Res 84:253–256
Kuhlencordt PJ, Rosel E, Gerszten RE et al (2004) Role of endothelial nitric oxide synthase in endothelial activation: insights from eNOS knockout endothelial cells. Am J Physiol-Cell Physiol 286:C1195–C1202. https://doi.org/10.1152/ajpcell.00546.2002
Lange CA (2008) Integration of progesterone receptor action with rapid signaling events in breast cancer models. J Steroid Biochem Mol Biol 108:203–212. https://doi.org/10.1016/j.jsbmb.2007.09.019
Lei B, Wang H, Jeong S et al (2016) Progesterone improves neurobehavioral outcome in models of intracerebral hemorrhage. Neuroendocrinology 103:665–677. https://doi.org/10.1159/000442204
Liu M, Kelley MH, Herson PS, Hurn PD (2010) Neuroprotection of sex steroids. Minerva Endocrinol 35:127–143
Liu A, Margaill I, Zhang S et al (2012) Progesterone receptors: a key for neuroprotection in experimental stroke. Endocrinology 153:3747–3757. https://doi.org/10.1210/en.2012-1138
Mani S (2008) Progestin receptor subtypes in the brain: the known and the unknown. Endocrinology 149:2750–2756. https://doi.org/10.1210/en.2008-0097
Meffre D, Pianos A, Liere P et al (2007) Steroid profiling in brain and plasma of male and pseudopregnant female rats after traumatic brain injury: analysis by gas chromatography/mass spectrometry. Endocrinology 148:2505–2517. https://doi.org/10.1210/en.2006-1678
Mehla J, iere S, Stuart E et al (2017) Gradual cerebral hypoperfusion impair fear conditioning and object recognition learning and memory in mice: potential roles of neurodegeneration and cholinergic dysfunction. bioRxiv. https://doi.org/10.1101/177121
Montes P, Vigueras-Villaseñor RM, Rojas-Castañeda JC et al (2019) Progesterone treatment in rats after severe global cerebral ischemia promotes hippocampal dentate gyrus neurogenesis and functional recovery. Neurol Res 41:429–436. https://doi.org/10.1080/01616412.2019.1576356
Moore C, Sanz-Rosa D, Emerson M (2011) Distinct role and location of the endothelial isoform of nitric oxide synthase in regulating platelet aggregation in males and females in vivo. Eur J Pharmacol 651:152–158. https://doi.org/10.1016/j.ejphar.2010.11.011
Moro MA, Cárdenas A, Hurtado O et al (2004) Role of nitric oxide after brain ischaemia. Cell Calcium 36:265–275. https://doi.org/10.1016/j.ceca.2004.02.011
Nabah YNA, Mateo T, Cerdá-NicoláS M et al (2005) L-NAME induces direct arteriolar leukocyte adhesion, which is mainly mediated by angiotensin-II. Microcirculation 12:443–453. https://doi.org/10.1080/10739680590960962
Nelson AR, Sweeney MD, Sagare AP, Zlokovic BV (2016) Neurovascular dysfunction and neurodegeneration in dementia and Alzheimer’s disease. Biochim et Biophys Acta 1862:887–900. https://doi.org/10.1016/j.bbadis.2015.12.016
Pang Y, Dong J, Thomas P (2015) Progesterone increases nitric oxide synthesis in human vascular endothelial cells through activation of membrane progesterone receptor-α. Am J Physiol-Endocrinol Metab 308:E899–E911. https://doi.org/10.1152/ajpendo.00527.2014
Patel A, Moalem A, Cheng H et al (2017) Chronic cerebral hypoperfusion induced by bilateral carotid artery stenosis causes selective recognition impairment in adult mice. Neurol Res 39:910–917. https://doi.org/10.1080/01616412.2017.1355423
Perez-Alvarez MJ, Wandosell F (2016) Stroke and neuroinflamation: role of sexual hormones. Curr Pharm Des 22:1334–1349. https://doi.org/10.2174/138161282210160304112834
Perez-Alvarez MJ, Mateos L, Alonso A, Wandosell F (2015) Estradiol and progesterone administration after pMCAO stimulates the neurological recovery and reduces the detrimental effect of ischemia mainly in hippocampus. Mol Neurobiol 52:1690–1703. https://doi.org/10.1007/s12035-014-8963-7
Rius-Pérez S, Tormos AM, Pérez S, Taléns-Visconti R (2018) Vascular pathology: cause or effect in alzheimer disease? Neurología (English Edition) 33:112–120. https://doi.org/10.1016/j.nrleng.2015.07.008
Salazar M, Lerma-Ortiz A, Hooks GM et al (2016) Progestin-mediated activation of MAPK and AKT in nuclear progesterone receptor negative breast epithelial cells: the role of membrane progesterone receptors. Gene 591:6–13. https://doi.org/10.1016/j.gene.2016.06.044
Shear DA, Galani R, Hoffman SW, Stein DG (2002) Progesterone protects against necrotic damage and behavioral abnormalities caused by traumatic brain injury. Exp Neurol 178:59–67. https://doi.org/10.1006/exnr.2002.8020
Si D, Yang P, Jiang R et al (2014) Improved cognitive outcome after progesterone administration is associated with protecting hippocampal neurons from secondary damage studied in vitro and in vivo. Behav Brain Res 264:135–142. https://doi.org/10.1016/j.bbr.2014.01.049
Singh M (2001) Ovarian Hormones elicit phosphorylation of Akt and extracellular-signal regulated kinase in explants of the cerebral cortex. Endocrine 14:407–416. https://doi.org/10.1385/ENDO:14:3:407
Singh M, Su C, Ng S (2013) Non-genomic mechanisms of progesterone action in the brain. Front Neurosci. https://doi.org/10.3389/fnins.2013.00159
Smith K, Saraswati M, Koehler R, Robertson C (2013) 182: the effect of progesterone on neurologic outcome in a rat model of pediatric traumatic brain injury. Crit Care Med 41:A40. https://doi.org/10.1097/01.ccm.0000439331.43555.fb
Stanojlovic M, Gusevac I, Grkovic I et al (2014a) Time-related sex differences in cerebral hypoperfusion-induced brain injury. Arch Biol Sci 66:1673–1680. https://doi.org/10.2298/ABS1404673S
Stanojlović M, Horvat A, Guševac I et al (2014b) Time course of cerebral hypoperfusion-induced neurodegenerative changes in the cortex of male and female rats. Folia Biol (Praha) 60:123–132
Stanojlović M, Guševac I, Grković I et al (2015a) Effects of chronic cerebral hypoperfusion and low-dose progesterone treatment on apoptotic processes, expression and subcellular localization of key elements within Akt and Erk signaling pathways in rat hippocampus. Neuroscience 311:308–321. https://doi.org/10.1016/j.neuroscience.2015.10.040
Stanojlović M, Zlatković J, Guševac I et al (2015b) Repeated low-dose 17β-estradiol treatment prevents activation of apoptotic signaling both in the synaptosomal and cellular fraction in rat prefrontal cortex following cerebral ischemia. Neurochem Int 83–84:1–8. https://doi.org/10.1016/j.neuint.2015.03.002
Stein DG (2008) Progesterone exerts neuroprotective effects after brain injury. Brain Res Rev 57:386–397. https://doi.org/10.1016/j.brainresrev.2007.06.012
Tang H, Zhang Q, Yang L et al (2014) GPR30 mediates estrogen rapid signaling and neuroprotection. Mol Cell Endocrinol 387:52–58. https://doi.org/10.1016/j.mce.2014.01.024
Terada K, Kaziro Y, Satoh T (2000) Analysis of Ras-dependent signals that prevent caspase-3 activation and apoptosis induced by cytokine deprivation in hematopoietic cells. Biochem Biophys Res Commun 267:449–455. https://doi.org/10.1006/bbrc.1999.1955
Vegeto E, Shahbaz MM, Wen DX et al (1993) Human progesterone receptor A form is a cell- and promoter-specific repressor of human progesterone receptor B function. Mol Endocrinol 7:1244–1255. https://doi.org/10.1210/mend.7.10.8264658
Villapol S, Byrnes KR, Symes AJ (2014) Temporal dynamics of cerebral blood flow, cortical damage, apoptosis, astrocyte–vasculature interaction and astrogliosis in the pericontusional region after traumatic brain injury. Front Neurol. https://doi.org/10.3389/fneur.2014.00082
Wagner AK, McCullough EH, Niyonkuru C et al (2011) Acute serum hormone levels: characterization and prognosis after severe traumatic brain injury. J Neurotrauma 28:871–888. https://doi.org/10.1089/neu.2010.1586
Wali B, Ishrat T, Stein DG, Sayeed I (2016) Progesterone improves long-term functional and histological outcomes after permanent stroke in older rats. Behav Brain Res 305:46–56. https://doi.org/10.1016/j.bbr.2016.02.024
Wang Z, Fan J, Wang J et al (2016) Chronic cerebral hypoperfusion induces long-lasting cognitive deficits accompanied by long-term hippocampal silent synapses increase in rats. Behav Brain Res 301:243–252. https://doi.org/10.1016/j.bbr.2015.12.047
Watts L, Sprague S, Fletcher L et al (2014) Does progesterone show neuroprotective effects on traumatic brain injury through increasing phosphorylation of Akt in the hippocampus? Neural Regener Res 9:1891. https://doi.org/10.4103/1673-5374.145355
Wei G, Dawson VL, Zweier JL (1999) Role of neuronal and endothelial nitric oxide synthase in nitric oxide generation in the brain following cerebral ischemia. Biochim Biophys Acta 1455:23–34
Yao X-L, Liu J, Lee E et al (2005) Progesterone differentially regulates pro- and anti-apoptotic gene expression in cerebral cortex following traumatic brain injury in rats. J Neurotrauma 22:656–668. https://doi.org/10.1089/neu.2005.22.656
Yousuf S, Atif F, Sayeed I et al (2014) Progesterone in transient ischemic stroke: a dose–response study. Psychopharmacology 231:3313–3323. https://doi.org/10.1007/s00213-014-3556-8
Yousuf S, Atif F, Sayeed I et al (2015) Long-term behavioral deficits and recovery after transient ischemia in middle-aged rats: effects of behavioral testing. Restor Neurol Neurosci 33:251–261. https://doi.org/10.3233/RNN-140450
Yousuf S, Brat DJ, Shu H-K et al (2017) Progesterone improves neurocognitive outcomes following therapeutic cranial irradiation in mice. Horm Behav 96:21–30. https://doi.org/10.1016/j.yhbeh.2017.08.004
Zaric M, Drakulic D, Stojanovic IG et al (2018) Regional-specific effects of cerebral ischemia/reperfusion and dehydroepiandrosterone on synaptic NMDAR/PSD-95 complex in male Wistar rats. Brain Res 1688:73–80. https://doi.org/10.1016/j.brainres.2018.03.023
Zhao Y, Zhang M, Liu H, Wang J (2015) Progesterone is neuroprotective by inhibiting cerebral edema after ischemia. Neural Regener Res 10:1076. https://doi.org/10.4103/1673-5374.160097
Acknowledgements
We thank Milorad Dragić for providing his unselfish technical support with Nissl staining and image analyses. We show gratitude to our retired colleague dr Anica Horvat who provided the original funding, knowledge, and expertise that greatly assisted the research.
Funding
This work was funded by the Ministry of Education, Science and Technological Development of the Republic of Serbia, grants 173044 and 41014.
Author information
Authors and Affiliations
Contributions
MS and IGS performed experiments and analyzed data. MZ assisted during biochemical measurements. IGS, MZ, JM, NM, and IG edited the manuscript. DD designed and supervised experimental setup, participated in the interpretation of obtained results, and wrote the manuscript. All authors approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical Approval
All applicable international, national, and institutional guidelines for the care and use of animals were followed. This article does not contain any studies with human participants performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Stanojlović, M., Guševac Stojanović, I., Zarić, M. et al. Progesterone Protects Prefrontal Cortex in Rat Model of Permanent Bilateral Common Carotid Occlusion via Progesterone Receptors and Akt/Erk/eNOS. Cell Mol Neurobiol 40, 829–843 (2020). https://doi.org/10.1007/s10571-019-00777-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-019-00777-2