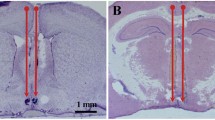
Abstract
We examined the role of the estrogen receptors alpha (ERα) and beta (ERβ) in of the preoptic-anterior hypothalamic area (POA-AHA) in the regulation of ovulation in rats. The number of ERα- and ERβ-immunoreactive (-ir) cells was determined at 09:00, 13:00, and 17:00 h of each stage of the estrous cycle in intact rats. Additionally, the effects of blocking ERα and ERβ on ovulation rate at 09:00 h on diestrus-2 or proestrus day through the microinjection of methyl-piperidino-pyrazole (MPP) or cyclofenil in either side of POA-AHA were evaluated. The number of ERα-ir and ERβ-ir cells in POA-AHA varied in each phase of estrous cycle. Either MPP or cyclofenil in the right side of POA-AHA on diestrus-2 day reduced the ovulation rate, while at proestrus day it was decreased in rats treated in either side with MPP, and in those treated with cyclofenil in the left side. MPP or cyclofenil produced a decrease in the surge of luteinizing hormone levels (LH) and an increase in progesterone and follicle stimulating hormone (FSH). Replacement with synthetic luteinizing hormone-releasing hormone in non-ovulating rats treated with MPP or cyclofenil restored ovulation. These results suggest that activation of estrogen receptors on the morning of diestrus-2 and proestrus day asymmetrically regulates ovulation and appropriately regulates the secretion of FSH and progesterone in the morning and afternoon of proestrus day. This ensures that both, the preovulatory secretion of LH and ovulation, occur at the right time.





Similar content being viewed by others
Abbreviations
- POA-AHA:
-
Preoptic and hypothalamic area
- LH:
-
Luteinizing hormone
- FSH:
-
Follicle stimulating hormone
- GnRH:
-
Gonadotropin-releasing hormone
- P4 :
-
Progesterone
- E2 :
-
E2
- ERα:
-
Estrogen receptors alpha
- ERβ:
-
Beta
- -ir:
-
Immunoreactive cells
- MPP:
-
Methyl-piperidino-pyrazole
- SERM:
-
Selective estrogen receptor modulator
- ELISA:
-
Enzyme-linked immunoassay
- RIA:
-
Radioimmunoassay
- LHRH-Gly-OH:
-
Synthetic LH-Releasing Hormone
- GABA:
-
Gamma aminobutyric acid
- PRL:
-
Prolactin
- PVN:
-
Paraventricular nucleus
- MPA:
-
Medial preoptic area
- MPOL:
-
Lateral part of medial preoptic nucleus
- VMPO:
-
Ventromedial preoptic nucleus
- PE:
-
Periventricular nucleus
- 3V:
-
Third ventricle
- AVPe:
-
Anteroventral periventricular nucleus
- ox:
-
Optic chiasm
References
Akema T, Kimura F (1991) 2-Hydroxysaclofen, a potent GABAB receptor antagonist, stimulates luteinizing hormone secretion in female rats. Brain Res 546(1):143–145
Arteaga-Lopez PR, Dominguez R, Cerbon MA, Mendoza-Rodriguez CA, Cruz ME (2003) Differential mRNA expression of alpha and beta estrogen receptor isoforms and GnRH in the left and right side of the preoptic and anterior hypothalamic area during the estrous cycle of the rat. Endocrine 21(3):251–260. https://doi.org/10.1385/ENDO:21:3:251
Blake CA (1974) Differentiation between the “critical period,” the “activation period” and the “potential activation period” for neurohumoral stimulation of LH release in proestrous rats. Endocrinology 95:572–578
Borras M, Hardy L, Lempereur F, el Khissiin AH, Legros N, Gol-Winkler R, Leclercq G (1994) Estradiol-induced down-regulation of estrogen receptor. Effect of various modulators of protein synthesis and expression. J Steroid Biochem Mol Biol 48(4):325–336
Bowman SP, Leake A, Morris ID (1982) Biological activity and steroid receptor interactions of cyclofenil with the oestrogen target tissues of the brain, pituitary gland and uterus of the rat. J Reprod Fertil 65(2):355–366
Bronson FH (1981) The regulation of luteinizing hormone secretion by estrogen: relationships among negative feedback, surge potential, and male stimulation in juvenile, peripubertal, and adult female mice. Endocrinology 108(2):506–516. https://doi.org/10.1210/endo-108-2-506
Calogero AE, Palumbo MA, Bosboom AM, Burrello N, Ferrara E, Palumbo G, Petraglia F, D’Agata R (1998) The neuroactive steroid allopregnanolone suppresses hypothalamic gonadotropin-releasing hormone release through a mechanism mediated by the gamma-aminobutyric acidA receptor. J Endocrinol 158(1):121–125
Caraty A, Locatelli A, Martin GB (1989) Biphasic response in the secretion of gonadotrophin-releasing hormone in ovariectomized ewes injected with oestradiol. J Endocrinol 123(3):375–382
Chaban VV, Lakhter AJ, Micevych P (2004) A membrane estrogen receptor mediates intracellular calcium release in astrocytes. Endocrinology 145(8):3788–3795. https://doi.org/10.1210/en.2004-0149
Constantin S, Iremonger KJ, Herbison AE (2013) In vivo recordings of GnRH neuron firing reveal heterogeneity and dependence upon GABAA receptor signaling. J Neurosci 33(22):9394–9401. https://doi.org/10.1523/jneurosci.0533-13.2013
Couse JF, Yates MM, Walker VR, Korach KS (2003) Characterization of the hypothalamic-pituitary-gonadal axis in estrogen receptor (ER) Null mice reveals hypergonadism and endocrine sex reversal in females lacking ERalpha but not ERbeta. Mol Endocrinol 17(6):1039–1053. https://doi.org/10.1210/me.2002-0398
Cruz ME, Jaramillo LP, Dominguez R (1989) Asymmetric ovulatory response induced by a unilateral implant of atropine in the anterior hypothalamus of the cyclic rat. J Endocrinol 123(3):437–439
Cruz ME, Arteaga P, Delgadillo H, Ma S, Dominguez R (1997) Differences on the acetylcholine concentration and binding and affinity parameters of the muscarinic receptors in the preoptic anterior hypothalamic area during the oestrous cycle of the rat. Med Sci Res 25:3
Daane TA, Parlow AF (1971) Periovulatory patterns of rat serum follicle stimulating hormone and luteinizing hormone during the normal estrous cycle: effects of pentobarbital. Endocrinology 88(3):653–663. https://doi.org/10.1210/endo-88-3-653
Domínguez R, Smith ER (1974) Barbiturate blockade of ovulation on days other than proestrus in the rat. Neuroendocrinology 14:212–223
Domínguez A, Damián-Matsumura P, Timossi C, Cruz ME, Dominguez R (1998) Characterization of monoamine neural activity in the preoptic anterior hypothalamic area and medial basal hypothalamus in rats during the day of pro-oestrus and its relation to gonadotrophin and sexual steroid hormone plasma levels. Med Sci Res 26(4):275–278
Donahue JE, Stopa EG, Chorsky RL, King JC, Schipper HM, Tobet SA, Blaustein JD, Reichlin S (2000) Cells containing immunoreactive estrogen receptor-alpha in the human basal forebrain. Brain Res 856(1–2):142–151
Dorling AA, Todman MG, Korach KS, Herbison AE (2003) Critical role for estrogen receptor alpha in negative feedback regulation of gonadotropin-releasing hormone mRNA expression in the female mouse. Neuroendocrinology 78(4):204–209. https://doi.org/10.1159/000073703
Espinosa-Valdez A, Flores A, Arrieta-Cruz I, Cardenas M, Chavira R, Dominguez R, Cruz ME (2016) The participation of the muscarinic receptors in the preoptic-anterior hypothalamic areas in the regulation of ovulation depends on the ovary. Reprod Biol Endocrinol 14(1):75. https://doi.org/10.1186/s12958-016-0208-3
Fortune JE, Vincent SE (1986) Prolactin modulates steroidogenesis by rat granulosa cells: i Effects on progesterone. Biol Reprod 35(1):84–91
Freeman ME (2006) Chapter 43 - Neuroendocrine Control of the Ovarian Cycle of the Rat. In: Wassarman JDNMPWPRGCMdKSRM (ed) Knobil and Neill’s Physiology of Reproduction (Third Edition). Academic Press, St Louis, pp 2327-2388. doi:http://dx.doi.org/10.1016/B978-012515400-0/50048-8
Green S, Walter P, Kumar V, Krust A, Bornert JM, Argos P, Chambon P (1986) Human oestrogen receptor cDNA: sequence, expression and homology to v-erb-A. Nature 320(6058):134–139. https://doi.org/10.1038/320134a0
Handa RJ, Weiser MJ (2014) Gonadal steroid hormones and the hypothalamo-pituitary-adrenal axis. Front Neuroendocrinol 35(2):197–220. https://doi.org/10.1016/j.yfrne.2013.11.001
Herbison AE (1998) Multimodal influence of estrogen upon gonadotropin-releasing hormone neurons. Endocr Rev 19(3):302–330. https://doi.org/10.1210/edrv.19.3.0332
Herbison AE (2006) Chapter 28 - Physiology of the Gonadotropin-Releasing Hormone Neuronal Network. In: Wassarman JDNMPWPRGCMdKSRM (ed) Knobil and Neill’s Physiology of Reproduction (Third Edition). Academic Press, St Louis, pp 1415-VII. doi:http://dx.doi.org/10.1016/B978-012515400-0/50033-6
Herbison AE (2015) Physiology of the adult gonadotropin-releasing hormone neuronal network. In: Zeleznik AJ, Plant TM (eds) Knobil and neill’s physiology of reproduction. Academic Press, San Diego, pp 399–467
Herbison AE, Heavens RP, Dye S, Dyer RG (1991) Acute action of oestrogen on medial preoptic gamma-aminobutyric Acid neurons: correlation with oestrogen negative feedback on luteinizing hormone secretion. J Neuroendocrinol 3(1):101–106. https://doi.org/10.1111/j.1365-2826.1991.tb00246.x
Hewitt SC, Korach KS (2002) Estrogen receptors: structure, mechanisms and function. Rev Endocr Metab Disord 3(3):193–200
Hrabovszky E, Shughrue PJ, Merchenthaler I, Hajszan T, Carpenter CD, Liposits Z, Petersen SL (2000) Detection of estrogen receptor-beta messenger ribonucleic acid and 125I-estrogen binding sites in luteinizing hormone-releasing hormone neurons of the rat brain. Endocrinology 141(9):3506–3509. https://doi.org/10.1210/endo.141.9.7788
Hrabovszky E, Steinhauser A, Barabas K, Shughrue PJ, Petersen SL, Merchenthaler I, Liposits Z (2001) Estrogen receptor-beta immunoreactivity in luteinizing hormone-releasing hormone neurons of the rat brain. Endocrinology 142(7):3261–3264. https://doi.org/10.1210/endo.142.7.8176
Humphrey RR, Dermody WC, Brink HO, Bousley FG, Schottin NH, Sakowski R, Vaitkus JW, Veloso HT, Reel JR (1973) Induction of luteinizing hormone (LH) release and ovulation in rats, hamsters, and rabbits by synthetic luteinizing hormone-releasing factor (LRF). Endocrinology 92(5):1515–1526. https://doi.org/10.1210/endo-92-5-1515
Jarry H, Perschl A, Wuttke W (1988) Further evidence that preoptic anterior hypothalamic GABAergic neurons are part of the GnRH pulse and surge generator. Acta Endocrinol 118(4):573–579
Kaiser UB, Jakubowiak A, Steinberger A, Chin WW (1997) Differential effects of gonadotropin-releasing hormone (GnRH) pulse frequency on gonadotropin subunit and GnRH receptor messenger ribonucleic acid levels in vitro. Endocrinology 138(3):1224–1231. https://doi.org/10.1210/endo.138.3.4968
Kallo I, Butler JA, Barkovics-Kallo M, Goubillon ML, Coen CW (2001) Oestrogen receptor beta-immunoreactivity in gonadotropin releasing hormone-expressing neurones: regulation by oestrogen. J Neuroendocrinol 13(9):741–748
Kuiper GG, Enmark E, Pelto-Huikko M, Nilsson S, Gustafsson JA (1996) Cloning of a novel receptor expressed in rat prostate and ovary. Proc Natl Acad Sci USA 93(12):5925–5930
Legan SJ, Tsai HW (2003) Oestrogen receptor-alpha and -beta immunoreactivity in gonadotropin-releasing hormone neurones after ovariectomy and chronic exposure to oestradiol. J Neuroendocrinol 15(12):1164–1170
Legan SJ, Coon GA, Karsch FJ (1975) Role of estrogen as initiator of daily LH surges in the ovariectomized rat. Endocrinology 96(1):50–56. https://doi.org/10.1210/endo-96-1-50
Lindzey J, Jayes FL, Yates MM, Couse JF, Korach KS (2006) The bi-modal effects of estradiol on gonadotropin synthesis and secretion in female mice are dependent on estrogen receptor-alpha. J Endocrinol 191(1):309–317. https://doi.org/10.1677/joe.1.06965
Liu X, Herbison AE (2011) Estrous cycle- and sex-dependent changes in pre- and postsynaptic GABAB control of GnRH neuron excitability. Endocrinology 152(12):4856–4864. https://doi.org/10.1210/en.2011-1369
Liu X, Porteous R, Herbison AE (2017) Dynamics of GnRH neuron ionotropic GABA and glutamate synaptic receptors are unchanged during estrogen positive and negative feedback in female mice. eNeuro. https://doi.org/10.1523/eneuro.0259-17.2017
Lopez-Ramirez YL, Lopez-Ramirez K, Arrieta-Cruz I, Flores A, Mendoza-Garces L, Librado-Osorio RA, Gutierrez-Juarez R, Dominguez R, Cruz ME (2017) Muscarinic receptors types 1 and 2 in the preoptic-anterior hypothalamic areas regulate ovulation unequally in the rat oestrous cycle. Int J Endocrinol 2017:4357080. https://doi.org/10.1155/2017/4357080
Mendoza-Garces L, Mendoza-Rodriguez CA, Jimenez-Trejo F, Picazo O, Rodriguez MC, Cerbon M (2011) Differential expression of estrogen receptors in two hippocampal regions during the estrous cycle of the rat. Anat Rec 294(11):1913–1919. https://doi.org/10.1002/ar.21247
Mikhael S, Punjala-Patel A, Gavrilova-Jordan L (2019) Hypothalamic-pituitary-ovarian axis disorders impacting female fertility. Biomedicines 7(1):5
Moenter SM, Caraty A, Karsch FJ (1990) The estradiol-induced surge of gonadotropin-releasing hormone in the ewe. Endocrinology 127(3):1375–1384. https://doi.org/10.1210/endo-127-3-1375
Moran JL, Dominguez R (1995) Effects of the unilateral implant of haloperidol at the preoptic-anterior hypothalamic area, on ovulation. Endocrine 3(6):391–393. https://doi.org/10.1007/bf02935642
Mosselman S, Polman J, Dijkema R (1996) ER beta: identification and characterization of a novel human estrogen receptor. FEBS Lett 392(1):49–53
Muthyala RS, Sheng S, Carlson KE, Katzenellenbogen BS, Katzenellenbogen JA (2003) Bridged bicyclic cores containing a 1,1-diarylethylene motif are high-affinity subtype-selective ligands for the estrogen receptor. J Med Chem 46(9):1589–1602. https://doi.org/10.1021/jm0204800
Nencioni T, Miragoli A, Bertaglia MG, Parini J (1982) Plasma FSH, LH and prolactin levels in postmenopausal women undergoing cyclofenil treatment. Acta Obstet Gynecol Scand 61(6):487–490
Paxinos G, Watson C (2005) The rat brain in stereotaxic coordinates, 5th edn. Elsevier Academic Press, Amsterdam
Seo JW, Comninos JS, Chi DY, Kim DW, Carlson KE, Katzenellenbogen JA (2006) Fluorine-substituted cyclofenil derivatives as estrogen receptor ligands: synthesis and structure-affinity relationship study of potential positron emission tomography agents for imaging estrogen receptors in breast cancer. J Med Chem 49(8):2496–2511. https://doi.org/10.1021/jm0512037
Sharifi N, Reuss AE, Wray S (2002) Prenatal LHRH neurons in nasal explant cultures express estrogen receptor beta transcript. Endocrinology 143(7):2503–2507. https://doi.org/10.1210/endo.143.7.8897
Shivers BD, Harlan RE, Morrell JI, Pfaff DW (1983) Absence of oestradiol concentration in cell nuclei of LHRH-immunoreactive neurones. Nature 304(5924):345–347
Shughrue PJ, Lane MV, Merchenthaler I (1997) Comparative distribution of estrogen receptor-α and -β mRNA in the rat central nervous system. J Comp Neurol 388:507–525
Shughrue PJ, Scrimo PJ, Merchenthaler I (1998) Evidence for the colocalization of estrogen receptor-beta mRNA and estrogen receptor-alpha immunoreactivity in neurons of the rat forebrain. Endocrinology 139(12):5267–5270
Simerly RB, Carr AM, Zee MC, Lorang D (1996) Ovarian steroid regulation of estrogen and progesterone receptor messenger ribonucleic acid in the anteroventral periventricular nucleus of the rat. J Neuroendocrinology 8(1):45–56
Skynner MJ, Sim JA, Herbison AE (1999) Detection of estrogen receptor α and β messenger ribonucleic acids in adult gonadotropin-releasing hormone neurons. Endocrinology 140:5195–5201
Sullivan SD, Moenter SM (2003) Neurosteroids alter gamma-aminobutyric acid postsynaptic currents in gonadotropin-releasing hormone neurons: a possible mechanism for direct steroidal control. Endocrinology 144(10):4366–4375. https://doi.org/10.1210/en.2003-0634
Suzuki S, Handa RJ (2005) Estrogen receptor-beta, but not estrogen receptor-alpha, is expressed in prolactin neurons of the female rat paraventricular and supraoptic nuclei: comparison with other neuropeptides. J Comp Neurol 484(1):28–42. https://doi.org/10.1002/cne.20457
Taubert HD, Kessler R, Busch G, Werner HJ (1970) The effect of clomiphene and cyclofenil upon pituitary LH and hypothalamic LH-releasing-factor content in the female rat. Experientia 26(1):97–98
White R, Lees JA, Needham M, Ham J, Parker M (1987) Structural organization and expression of the mouse estrogen receptor. Mol Endocrinol 1(10):735–744. https://doi.org/10.1210/mend-1-10-735
Xia L, Van Vugt D, Alston EJ, Luckhaus J, Ferin M (1992) A surge of gonadotropin-releasing hormone accompanies the estradiol-induced gonadotropin surge in the rhesus monkey. Endocrinology 131(6):2812–2820. https://doi.org/10.1210/endo.131.6.1446619
Yamaji T, Dierschke DJ, Hotchkiss J, Bhattacharya AN, Surve AH, Knobil E (1971) Estrogen induction of LH release in the rhesus monkey. Endocrinology 89(4):1034–1041. https://doi.org/10.1210/endo-89-4-1034
Zhang C, Bosch MA, Ronnekleiv OK, Kelly MJ (2009) Gamma-aminobutyric acid B receptor mediated inhibition of gonadotropin-releasing hormone neurons is suppressed by kisspeptin-G protein-coupled receptor 54 signaling. Endocrinology 150(5):2388–2394. https://doi.org/10.1210/en.2008-1313
Acknowledgements
We thank to Facultad de Estudios Superiores, Zaragoza from UNAM at Mexico City by the access to the facilities used to perform this study and Michael Martínez-Mayo for his technical assistance.
Funding
This study was funded by UNAM-DGAPA-PAPIIT (grant number: IN214508-3 and IN-21005319), and CONACYT (grant number: 81898). Author Cruz ME has received research grants from UNAM-DGAPA-PAPIIT and CONACYT.
Author information
Authors and Affiliations
Contributions
MEC designed the experiments; MEC, IAC, RGJ, and RDC wrote the manuscript. IAC, MGL, and RLO performed the immunohistochemical analysis; RCh and CM quantified the concentrations of hormones; MEC, AF, IAC, RGJ, and RDC participated in the analysis and discussion of the results. All authors read and approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
All authors declares that they have no conflict of interest.
Ethical Approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. All experiments were performed in strict accordance with the Mexican Law of Animal Treatment and Protection Guidelines and the specifications of the Mexican Official Standard NOM-062-ZOO-1999. The Institutional Committee of the Facultad de Estudios Superiores Zaragoza, Universidad Nacional Autónoma de México approved the experimental protocols (FES/DEPUCI/236/14). All efforts were made to minimize the number of animals used and their suffering.
Research Involving Human and Animal Participants
This article does not contain any studies with human participants performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Arrieta-Cruz, I., Librado-Osorio, R., Flores, A. et al. Estrogen Receptors Alpha and Beta in POA-AHA Region Regulate Asymmetrically Ovulation. Cell Mol Neurobiol 39, 1139–1149 (2019). https://doi.org/10.1007/s10571-019-00708-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-019-00708-1