Abstract
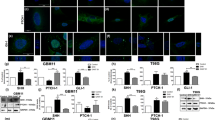
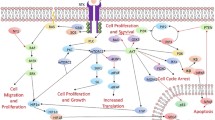
Glioblastoma Multiforme (GBM) is an aggressive form of brain Tumor that has few cures. In this study, we analyze the anti-proliferative effects of a new molecule JQ1 against GBMs induced in Wistar Rats. JQ1 is essentially a Myc inhibitor. c-Myc is also known for altering the biochemistry of a tumor cell. Therefore, the study is intended to analyze certain other oncogenes associated with c-Myc and also the change in cellular biochemistry upon c-Myc inhibition. The quantitative analysis of gene expression gave a co-expressive pattern for all the three genes involved namely; c-Myc, Bcl-2, and Akt. The cellular biochemistry analysis by transmission electron microscopy revealed high glycogen and lipid aggregation in Myc inhibited cells and excessive autophagy. The study demonstrates the role of c-Myc as a central metabolic regulator and Bcl-2 and Akt assisting in extending c-Myc half-life as well as in regulation of autophagy, so as to regulate cell survival on the whole. The study also demonstrates that transient treatment by JQ1 leads to aggressive development of tumor and therefore, accelerating death, emphasizing the importance of dosage fixation, and duration for clinical use in future.






Similar content being viewed by others
Abbreviations
- BET:
-
Bromodomain and extra terminal
- EGFR:
-
Epidermal growth factor receptor
- PKM2:
-
Pyruvate kinase muscle isozyme 2
- IDH:
-
Isocitrate dehydrogenase
- HIF:
-
Hypoxia-inducible factor
- ACL:
-
ATP citrate lyase
- ACC:
-
Acetyl-CoA carboxylase
- FASN:
-
Fatty acid synthase
- GSK:
-
Glycogen synthase kinase
References
Alcantara Llaguno SR, Chen J, Parada LF (2009) Signaling in malignant astrocytomas: role of neural stem cells and its therapeutic implications. Clin Cancer Res 15:7124–7129. doi:10.1158/1078-0432.ccr-09-0433
Bandopadhayay P et al (2013) BET-bromodomain inhibition of MYC-amplified medulloblastoma. Clin Cancer. doi:10.1158/1078-0432.ccr-13-2281
Belkina AC, Blanton WP, Nikolajczyk BS, Denis GV (2013) The double bromodomain protein Brd2 promotes B cell expansion and mitogenesis. J Leukoc Biol. doi:10.1189/jlb.1112588
Cheng Z et al (2013) Inhibition of BET bromodomain targets genetically diverse glioblastoma. Clin Cancer Res 19:1748–1759. doi:10.1158/1078-0432.ccr-12-3066
Cory S, Vaux DL, Strasser A, Harris AW, Adams JM (1999) Insights from Bcl-2 and Myc: malignancy involves abrogation of apoptosis as well as sustained proliferation. Cancer Res 59:1685s–1692s
Cory S, Huang DC, Adams JM (2003) The Bcl-2 family: roles in cell survival and oncogenesis. Oncogene 22:8590–8607. doi:10.1038/sj.onc.1207102
Dang CV (1999) c-Myc target genes involved in cell growth. Apoptosis Metab Mol Cell Biol 19:1–11
Dang CV (2012) Links between metabolism and cancer. Genes Dev 26:877–890. doi:10.1101/gad.189365.112
Darnell JE Jr (2002) Transcription factors as targets for cancer therapy nature reviews. Cancer 2:740–749. doi:10.1038/nrc906
De Salvo M et al (2011) Temozolomide induced c-Myc-mediated apoptosis via Akt signalling in MGMT expressing glioblastoma cells. Int J Radiat Biol 87:518–533. doi:10.3109/09553002.2011.556173
Delmore JE et al (2011) BET bromodomain inhibition as a therapeutic strategy to target c-Myc. Cell 146:904–917. doi:10.1016/j.cell.2011.08.017
Devan S, Janardhanam VA (2011) Effect of naringenin on metabolic markers, lipid profile and expression of GFAP in C6 glioma cells implanted rat’s brain. Ann Neurosci 18:151–155
Evan G, Littlewood T (1998) A matter of life and cell death. Science 281:1317–1322
Fan Y, Dickman KG, Zong WX (2010) Akt and c-Myc differentially activate cellular metabolic programs and prime cells to bioenergetic inhibition. J Biol Chem 285:7324–7333. doi:10.1074/jbc.M109.035584
Fanidi A, Harrington EA, Evan GI (1992) Cooperative interaction between c-myc and bcl-2 proto-oncogenes. Nature 359:554–556. doi:10.1038/359554a0
Favaro E et al (2012) Glucose utilization via glycogen phosphorylase sustains proliferation and prevents premature senescence in cancer cells. Cell Metab 16:751–764. doi:10.1016/j.cmet.2012.10.017
Filippakopoulos P et al (2010) Selective inhibition of BET bromodomains. Nature 468:1067–1073. doi:10.1038/nature09504
Graves JA et al (2012) Mitochondrial structure, function and dynamics are temporally controlled by c-Myc. PLoS One 7:e37699. doi:10.1371/journal.pone.0037699
Grommes C et al (2006) Inhibition of in vivo glioma growth and invasion by peroxisome proliferator-activated receptor γ agonist treatment. Mol Pharmacol 70:1524–1533. doi:10.1124/mol.106.022194
Hu S et al. (2013) MYC/BCL2 protein coexpression contributes to the inferior survival of activated B-cell subtype of diffuse large B-cell lymphoma and demonstrates high-risk gene expression signatures: a report from The International DLBCL Rituximab-CHOP Consortium Program Blood 121:4021-4031; quiz 4250 doi:10.1182/blood-2012-10-460063
Jahani-Asl A, Bonni A (2013) iNOS: a potential therapeutic target for malignant glioma. Curr Mol Med 13:1241–1249
Kihara-Negishi F et al (1998) Down-regulation of c-myc and bcl-2 gene expression in PU.1-induced apoptosis in murine erythroleukemia cells international journal of cancer. J Int cancer 76:523–530
Kim J, Lee JH, Iyer VR (2008) Global identification of Myc target genes reveals its direct role in mitochondrial biogenesis and its E-box usage in vivo. PloS one 3:e1798. doi:10.1371/journal.pone.0001798
Morrish F, Isern N, Sadilek M, Jeffrey M, Hockenbery DM (2009) c-Myc activates multiple metabolic networks to generate substrates for cell-cycle entry. Oncogene 28:2485–2491. doi:10.1038/onc.2009.112
Morrish F et al (2010) Myc-dependent mitochondrial generation of acetyl-CoA contributes to fatty acid biosynthesis and histone acetylation during cell cycle entry. J Biol Chem 285:36267–36274. doi:10.1074/jbc.M110.141606
Murat A et al (2008) Stem cell-related “self-renewal” signature and high epidermal growth factor receptor expression associated with resistance to concomitant chemoradiotherapy in glioblastoma. J Clin Oncol 26:3015–3024. doi:10.1200/jco.2007.15.7164
Ohgaki H, Kleihues P (2009) Genetic alterations and signaling pathways in the evolution of gliomas. Cancer Sci 100:2235–2241. doi:10.1111/j.1349-7006.2009.01308.x
Pescador N et al (2010) Hypoxia promotes glycogen accumulation through hypoxia inducible factor (HIF)-mediated induction of glycogen synthase 1. PLoS ONE 5:e9644. doi:10.1371/journal.pone.0009644
Preusser M, de Ribaupierre S, Wöhrer A, Erridge SC, Hegi M, Weller M, Stupp R (2011) Current concepts and management of glioblastoma. Ann Neurol 70:9–21. doi:10.1002/ana.22425
Rabinowitz JD, White E (2010) Autophagy and metabolism. Science 330:1344–1348. doi:10.1126/science.1193497
Sears R, Nuckolls F, Haura E, Taya Y, Tamai K, Nevins JR (2000) Multiple ras-dependent phosphorylation pathways regulate Myc protein stability. Genes Dev 14:2501–2514. doi:10.1101/gad.836800
Singh R, Cuervo AM (2011) Autophagy in the cellular energetic balance. Cell Metab 13:495–504. doi:10.1016/j.cmet.2011.04.004
Singh SK et al (2004) Identification of human brain tumour initiating cells. Nature 432:396–401. doi:10.1038/nature03128
Tang X et al (2013) BET bromodomain proteins mediate downstream signaling events following growth factor stimulation in human lung fibroblasts and are involved in bleomycin-induced pulmonary fibrosis. Mol Pharmacol 83:283–293. doi:10.1124/mol.112.081661
Venneti S, Thompson CB (2013) Metabolic modulation of epigenetics in gliomas. Brain Pathol 23:217–221. doi:10.1111/bpa.12022
Wu X, Qi J, Bradner JE, Xiao G, Chen LF (2013) Bromodomain and extraterminal (BET) protein inhibition suppresses human T cell leukemia virus 1 (HTLV-1) tax protein-mediated tumorigenesis by inhibiting nuclear factor kappab (NF-kappaB) signaling. J Biol Chem 288:36094–36105. doi:10.1074/jbc.M113.485029
Zeller KI et al (2006) Global mapping of c-Myc binding sites and target gene networks in human B cells. Proc Natl Acad Sci 103:17834–17839. doi:10.1073/pnas.0604129103
Zhang F, Du G (2012) Dysregulated lipid metabolism in cancer. World j Biol Chem 3:167–174. doi:10.4331/wjbc.v3.i8.167
Zhao S et al (2009) Glioma-derived mutations in IDH1 dominantly inhibit IDH1 catalytic activity and induce HIF-1alpha. Science 324:261–265. doi:10.1126/science.1170944
Zirath H et al (2013) MYC inhibition induces metabolic changes leading to accumulation of lipid droplets in tumor cells. Proc Natl Acad Sci U S A 110:10258–10263. doi:10.1073/pnas.1222404110
Acknowledgments
The study was supported by University Grants Commission, New Delhi, India. We thank the Sophisticated Analytical Instrument Facility (SAIF)-Department of Anatomy, AIIMS, New Delhi, India for providing microscopes in Electron Microscopy. We thank Dr. Aparna Rajagopalan for her critical reading and editing. We also thank University of Madras for all the infrastructure support in carrying out the experiments.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rajagopalan, V., Vaidyanathan, M., Janardhanam, V.A. et al. Pre-Clinical Analysis of Changes in Intra-cellular Biochemistry of Glioblastoma Multiforme (GBM) Cells Due to c-Myc Silencing. Cell Mol Neurobiol 34, 1059–1069 (2014). https://doi.org/10.1007/s10571-014-0083-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-014-0083-4