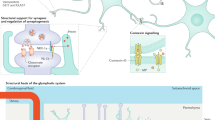
Abstract
Pathological pain has been subjected to intense research to shed light on the underlying mechanisms of key symptoms, such as allodynia and hyperalgesia. The main focus has by and large concerned plasticity of spinal cord neurons and the primary afferent nerves relaying peripheral information to the spinal cord. Animal pain models display an increased presence of reactive astrocytes in the spinal cord, but in contrast to neurons, little is known about how they contribute to abnormal pain sensation. However, astrocytes are now beginning to receive greater attention, and as new information is emerging, it appears that astrocytes undertake critical roles in manifesting pathological pain. Through the secretion of diffusible transmitters, such as interleukins, ATP, and NO, astrocytes may augment primary afferent neuronal signaling or sensitize second order neurons in the spinal cord. In addition, astrocytes might lead to altered pain perception by a direct modulation of synaptic transmission between neurons in the nociceptive pathway or through the creation of astrocytic networks capable of transducing signals for extended distances across and along the spinal cord. Future research in astrocyte activation and signaling may therefore reveal novel drug targets for managing pathological pain.




Similar content being viewed by others
References
Ahmed Z, Lewis CA, Faber DS (1990) Glutamate stimulates release of Ca2+ from internal stores in astroglia. Brain Res 516:165–169. doi:10.1016/0006-8993(90)90914-W
Anderson CM, Bergher JP, Swanson RA (2004) ATP-induced ATP release from astrocytes. J Neurochem 88:246–256
Araque A, Parpura V, Sanzgiri RP, Haydon PG (1999) Tripartite synapses: glia, the unacknowledged partner. Trends Neurosci 22:208–215. doi:10.1016/S0166-2236(98)01349-6
Araque A, Carmignoto G, Haydon PG (2001) Dynamic signaling between astrocytes and neurons. Annu Rev Physiol 63:795–813. doi:10.1146/annurev.physiol.63.1.795
Baron R, Wasner G (2001) Complex regional pain syndromes. Curr Pain Headache Rep 5:114–123. doi:10.1007/s11916-001-0079-x
Beattie EC, Stellwagen D, Morishita W, Bresnahan JC, Ha BK, Von Zastrow M, Beattie MS, Malenka RC (2002) Control of synaptic strength by glial TNFalpha. Science 295:2282–2285. doi:10.1126/science.1067859
Beaujouan JC, Daguet de Montety MC, Torrens Y, Saffroy M, Dietl M, Glowinski J (1990) Marked regional heterogeneity of 125I-Bolton Hunter substance P binding and substance P-induced activation of phospholipase C in astrocyte cultures from the embryonic or newborn rat. J Neurochem 54:669–675. doi:10.1111/j.1471-4159.1990.tb01923.x
Benveniste EN, Sparacio SM, Norris JG, Grenett HE, Fuller GM (1990) Induction and regulation of interleukin-6 gene expression in rat astrocytes. J Neuroimmunol 30:201–212. doi:10.1016/0165-5728(90)90104-U
Bogoyevitch MA, Kobe B (2006) Uses for JNK: the many and varied substrates of the c-Jun N- terminal kinases. Microbiol Mol Biol Rev 70:1061–1095. doi:10.1128/MMBR.00025-06
Boitano S, Dirksen ER, Sanderson MJ (1992) Intercellular propagation of calcium waves mediated by inositol trisphosphate. Science 258:292–295. doi:10.1126/science.1411526
Chen CC, Akopian AN, Sivilotti L, Colquhoun D, Burnstock G, Wood JN (1995) A P2X purinoceptor expressed by a subset of sensory neurons. Nature 377:428–431. doi:10.1038/377428a0
Ching DW, McClintock A, Beswick F (2003) Successful treatment with low-dose thalidomide in a patient with both Behcet’s disease and complex regional pain syndrome type I: case report. J Clin Rheumatol 9:96–98. doi:10.1097/01.RHU.0000062620.10630.21
Cholewinski AJ, Hanley MR, Wilkin GP (1988) A phosphoinositide-linked peptide response in astrocytes: evidence for regional heterogeneity. Neurochem Res 13:389–394. doi:10.1007/BF00972490
Copray JC, Mantingh I, Brouwer N, Biber K, Kust BM, Liem RS, Huitinga I, Tilders FJ, Van Dam AM, Boddeke HW (2001) Expression of interleukin-1 beta in rat dorsal root ganglia. J Neuroimmunol 118:203–211. doi:10.1016/S0165-5728(01)00324-1
Cornell-Bell AH, Finkbeiner SM, Cooper MS, Smith SJ (1990) Glutamate induces calcium waves in cultured astrocytes: long-range glial signaling. Science 247:470–473. doi:10.1126/science.1967852
Cotrina ML, Lin JH, Alves-Rodrigues A, Liu S, Li J, Azmi-Ghadimi H, Kang J, Naus CC, Nedergaard M (1998) Connexins regulate calcium signaling by controlling ATP release. Proc Natl Acad Sci USA 95:15735–15740. doi:10.1073/pnas.95.26.15735
Coull JA, Beggs S, Boudreau D, Boivin D, Tsuda M, Inoue K, Gravel C, Salter MW, De Koninck Y (2005) BDNF from microglia causes the shift in neuronal anion gradient underlying neuropathic pain. Nature 438:1017–1021. doi:10.1038/nature04223
Dani JW, Chernjavsky A, Smith SJ (1992) Neuronal activity triggers calcium waves in hippocampal astrocyte networks. Neuron 8:429–440. doi:10.1016/0896-6273(92)90271-E
Del Bigio MR, Fedoroff S, Qualtiere LF (1992) Morphology of astroglia in colony cultures following transient exposure to potassium ion, hypoosmolarity and vasopressin. J Neurocytol 21:7–18. doi:10.1007/BF01206894
Del Bigio MR, Omara F, Fedoroff S (1994) Astrocyte proliferation in culture following exposure to potassium ion. NeuroReport 5:639–641. doi:10.1097/00001756-199401000-00027
DeLeo JA, Colburn RW, Rickman AJ (1997) Cytokine and growth factor immunohistochemical spinal profiles in two animal models of mononeuropathy. Brain Res 759:50–57. doi:10.1016/S0006-8993(97)00209-6
Djouhri L, Lawson SN (2004) Abeta-fiber nociceptive primary afferent neurons: a review of incidence and properties in relation to other afferent A-fiber neurons in mammals. Brain Res Brain Res Rev 46:131–145. doi:10.1016/j.brainresrev.2004.07.015
Falsig J, Pörzgen P, Lotharius J, Leist M (2004) Specific modulation of astrocyte inflammation by inhibition of mixed lineage kinases with CEP-1347. J Immunol 173:2762–2770
Fellin T, Pascual O, Gobbo S, Pozzan T, Haydon PG, Carmignoto G (2004) Neuronal synchrony mediated by astrocytic glutamate through activation of extrasynaptic NMDA receptors. Neuron 43:729–743. doi:10.1016/j.neuron.2004.08.011
Ferreira SH, Lorenzetti BB, Bristow AF, Poole S (1988) Interleukin-1 beta as a potent hyperalgesic agent antagonized by a tripeptide analogue. Nature 334:698–700. doi:10.1038/334698a0
Fiacco TA, McCarthy KD (2004) Intracellular astrocyte calcium waves in situ increase the frequency of spontaneous AMPA receptor currents in CA1 pyramidal neurons. J Neurosci 24:722–732. doi:10.1523/JNEUROSCI.2859-03.2004
Finkbeiner S (1992) Calcium waves in astrocytes-filling in the gaps. Neuron 8:1101–1108. doi:10.1016/0896-6273(92)90131-V
Fyffe RE, Perl ER (1984) Is ATP a central synaptic mediator for certain primary afferent fibers from mammalian skin? Proc Natl Acad Sci USA 81:6890–6893. doi:10.1073/pnas.81.21.6890
Gallagher CJ, Salter MW (2003) Differential properties of astrocyte calcium waves mediated by P2Y1 and P2Y2 receptors. J Neurosci 23:6728–6739
Gao YJ, Ji RR (2008) Activation of JNK pathway in persistent pain. Neurosci Lett 437:180–183. doi:10.1016/j.neulet.2008.03.017
Garrison CJ, Dougherty PM, Kajander KC, Carlton SM (1991) Staining of glial fibrillary acidic protein (GFAP) in lumbar spinal cord increases following a sciatic nerve constriction injury. Brain Res 565:1–7. doi:10.1016/0006-8993(91)91729-K
Garrison CJ, Dougherty PM, Carlton SM (1994) GFAP expression in lumbar spinal cord of naive and neuropathic rats treated with MK-801. Exp Neurol 129:237–243. doi:10.1006/exnr.1994.1165
Gu JG (2003) P2X receptor-mediated modulation of sensory transmission to the spinal cord dorsal horn. Neuroscientist 9:370–378. doi:10.1177/1073858403252788
Gu JG, MacDermott AB (1997) Activation of ATP P2X receptors elicits glutamate release from sensory neuron synapses. Nature 389:749–753. doi:10.1038/39639
Guhring H, Gorig M, Ates M, Coste O, Zeilhofer HU, Pahl A, Rehse K, Brune K (2000) Suppressed injury-induced rise in spinal prostaglandin E2 production and reduced early thermal hyperalgesia in iNOS-deficient mice. J Neurosci 20:6714–6720
Guo W, Wang H, Watanabe M, Shimizu K, Zou S, LaGraize SC, Wei F, Dubner R, Ren K (2007) Glial-cytokine-neuronal interactions underlying the mechanisms of persistent pain. J Neurosci 27:6006–6018. doi:10.1523/JNEUROSCI.0176-07.2007
Guthrie PB, Knappenberger J, Segal M, Bennett MV, Charles AC, Kater SB (1999) ATP released from astrocytes mediates glial calcium waves. J Neurosci 19:520–528
Haas CA, Reddington M, Kreutzberg GW (1991) Calcitonin gene-related peptide stimulates the induction of c-fos gene expression in rat astrocyte cultures. Eur J NeuroSci 3:708–712. doi:10.1111/j.1460-9568.1991.tb00856.x
Hald A, Lotharius J (2005) Oxidative stress and inflammation in Parkinson’s disease: is there a causal link? Exp Neurol 193:279–290. doi:10.1016/j.expneurol.2005.01.013
Hald A, Nedergaard S, Hansen RR, Ding M, Heegaard AM (2009) Differential activation of spinal cord glial cells in murine models of neuropathic and cancer pain. Eur J Pain 13:138–145. doi:10.1016/j.ejpain.2008.03.014
Hashizume H, DeLeo JA, Colburn RW, Weinstein JN (2000) Spinal glial activation and cytokine expression after lumbar root injury in the rat. Spine 25:1206–1217. doi:10.1097/00007632-200005150-00003
Hassinger TD, Guthrie PB, Atkinson PB, Bennett MV, Kater SB (1996) An extracellular signaling component in propagation of astrocytic calcium waves. Proc Natl Acad Sci USA 93:13268–13273. doi:10.1073/pnas.93.23.13268
Himes BT, Tessler A (1989) Death of some dorsal root ganglion neurons and plasticity of others following sciatic nerve section in adult and neonatal rats. J Comp Neurol 284:215–230. doi:10.1002/cne.902840206
Honore P, Rogers SD, Schwei MJ, Salak-Johnson JL, Luger NM, Sabino MC, Clohisy DR, Mantyh PW (2000) Murine models of inflammatory, neuropathic and cancer pain each generates a unique set of neurochemical changes in the spinal cord and sensory neurons. Neuroscience 98:585–598. doi:10.1016/S0306-4522(00)00110-X
Ikeda H, Tsuda M, Inoue K, Murase K (2007) Long-term potentiation of neuronal excitation by neuron–glia interactions in the rat spinal dorsal horn. Eur J NeuroSci 25:1297–1306. doi:10.1111/j.1460-9568.2007.05386.x
Inagaki N, Fukui H, Ito S, Wada H (1991) Type-2 astrocytes show intracellular Ca2+ elevation in response to various neuroactive substances. Neurosci Lett 128:257–260. doi:10.1016/0304-3940(91)90274-W
Innocenti B, Parpura V, Haydon PG (2000) Imaging extracellular waves of glutamate during calcium signaling in cultured astrocytes. J Neurosci 20:1800–1808
Inoue A, Ikoma K, Morioka N, Kumagai K, Hashimoto T, Hide I, Nakata Y (1999) Interleukin-1beta induces substance P release from primary afferent neurons through the cyclooxygenase-2 system. J Neurochem 73:2206–2213
Jahr CE, Jessell TM (1983) ATP excites a subpopulation of rat dorsal horn neurones. Nature 304:730–733. doi:10.1038/304730a0
Ji RR, Kohno T, Moore KA, Woolf CJ (2003) Central sensitization and LTP: do pain and memory share similar mechanisms? Trends Neurosci 26:696–705. doi:10.1016/j.tins.2003.09.017
Ji RR, Kawasaki Y, Zhuang ZY, Wen YR, Decosterd I (2006) Possible role of spinal astrocytes in maintaining chronic pain sensitization: review of current evidence with focus on bFGF/JNK pathway. Neuron Glia Biol 2:259–269. doi:10.1017/S1740925X07000403
Ju G, Hokfelt T, Brodin E, Fahrenkrug J, Fischer JA, Frey P, Elde RP, Brown JC (1987) Primary sensory neurons of the rat showing calcitonin gene-related peptide immunoreactivity and their relation to substance P-, somatostatin-, galanin-, vasoactive intestinal polypeptide- and cholecystokinin-immunoreactive ganglion cells. Cell Tissue Res 247:417–431. doi:10.1007/BF00218323
Julius D, Basbaum AI (2001) Molecular mechanisms of nociception. Nature 413:203–210. doi:10.1038/35093019
Kennedy C (2005) P2X receptors: targets for novel analgesics? Neuroscientist 11:345–356. doi:10.1177/1073858404274063
Kofuji P, Newman EA (2004) Potassium buffering in the central nervous system. Neuroscience 129:1045–1056. doi:10.1016/j.neuroscience.2004.06.008
Kozlova EN (2003) Differentiation and migration of astrocytes in the spinal cord following dorsal root injury in the adult rat. Eur J NeuroSci 17:782–790. doi:10.1046/j.1460-9568.2003.02518.x
Krasowska-Zoladek A, Banaszewska M, Kraszpulski M, Konat GW (2007) Kinetics of inflammatory response of astrocytes induced by TLR 3 and TLR4 ligation. J Neurosci Res 85:205–212. doi:10.1002/jnr.21088
Krishna M, Narang H (2008) The complexity of mitogen-activated protein kinases (MAPKs) made simple. Cell Mol Life Sci 65:636–3544. doi:10.1007/s00018-008-8170-7
Lazar P, Reddington M, Streit W, Raivich G, Kreutzberg GW (1991) The action of calcitonin gene-related peptide on astrocyte morphology and cyclic AMP accumulation in astrocyte cultures from neonatal rat brain. Neurosci Lett 130:99–102. doi:10.1016/0304-3940(91)90237-N
Lee JC, Simon PL, Young PR (1989) Constitutive and PMA-induced interleukin-1 production by the human astrocytoma cell line T24. Cell Immunol 118:298–311. doi:10.1016/0008-8749(89)90379-1
Lee SC, Dickson DW, Liu W, Brosnan CF (1993) Induction of nitric oxide synthase activity in human astrocytes by interleukin-1 beta and interferon-gamma. J Neuroimmunol 46:19–24. doi:10.1016/0165-5728(93)90229-R
Li Y, Ji A, Weihe E, Schafer MK (2004) Cell-specific expression and lipopolysaccharide-induced regulation of tumor necrosis factor alpha (TNFalpha) and TNF receptors in rat dorsal root ganglion. J Neurosci 24:9623–9631. doi:10.1523/JNEUROSCI.2392-04.2004
Li CY, Zhang XL, Matthews EA, Li KW, Kurwa A, Boroujerdi A, Gross J, Gold MS, Dickenson AH, Feng G, Luo ZD (2006) Calcium channel alpha2delta1 subunit mediates spinal hyperexcitability in pain modulation. Pain 125:20–34. doi:10.1016/j.pain.2006.04.022
Liu GJ, Kalous A, Werry EL, Bennett MR (2006) Purine release from spinal cord microglia after elevation of calcium by glutamate. Mol Pharmacol 70:851–859. doi:10.1124/mol.105.021436
Liu YL, Zhou LJ, Hu NW, Xu JT, Wu CY, Zhang T, Li YY, Liu XG (2007) Tumor necrosis factor-alpha induces long-term potentiation of C-fiber evoked field potentials in spinal dorsal horn in rats with nerve injury: the role of NF-kappa B, JNK and p38 MAPK. Neuropharmacology 52:708–715. doi:10.1016/j.neuropharm.2006.09.011
Luo ZD, Chaplan SR, Higuera ES, Sorkin LS, Stauderman KA, Williams ME, Yaksh TL (2001) Upregulation of dorsal root ganglion (alpha)2(delta) calcium channel subunit and its correlation with allodynia in spinal nerve-injured rats. J Neurosci 21:1868–1875
Ma W, Quirion R (2002) Partial sciatic nerve ligation induces increase in the phosphorylation of extracellular signal-regulated kinase (ERK) and c-Jun N-terminal kinase (JNK) in astrocytes in the lumbar spinal dorsal horn and the gracile nucleus. Pain 99:175–184. doi:10.1016/S0304-3959(02)00097-0
Ma JY, Zhao ZQ (2002) The involvement of glia in long-term plasticity in the spinal dorsal horn of the rat. NeuroReport 13:1781–1784. doi:10.1097/00001756-200210070-00017
Magistretti PJ, Pellerin L, Rothman DL, Shulman RG (1999) Energy on demand. Science 283:496–497. doi:10.1126/science.283.5401.496
Maier SF, Wiertelak EP, Martin D, Watkins LR (1993) Interleukin-1 mediates the behavioral hyperalgesia produced by lithium chloride and endotoxin. Brain Res 623:321–324. doi:10.1016/0006-8993(93)91446-Y
Malmberg AB, Chen C, Tonegawa S, Basbaum AI (1997) Preserved acute pain and reduced neuropathic pain in mice lacking PKCgamma. Science 278:279–283. doi:10.1126/science.278.5336.279
Mancini I, Dumon JC, Body JJ (2004) Efficacy and safety of ibandronate in the treatment of opioid-resistant bone pain associated with metastatic bone disease: a pilot study. J Clin Oncol 22:3587–3592. doi:10.1200/JCO.2004.07.054
Martin WJ, Liu H, Wang H, Malmberg AB, Basbaum AI (1999) Inflammation-induced up-regulation of protein kinase C gamma immunoreactivity in rat spinal cord correlates with enhanced nociceptive processing. Neuroscience 88:1267–1274. doi:10.1016/S0306-4522(98)00314-5
McNaught KS, Jenner P (2000) Dysfunction of rat forebrain astrocytes in culture alters cytokine and neurotrophic factor release. Neurosci Lett 285:61–65. doi:10.1016/S0304-3940(00)00982-4
Meller ST, Dykstra C, Grzybycki D, Murphy S, Gebhart GF (1994) The possible role of glia in nociceptive processing and hyperalgesia in the spinal cord of the rat. Neuropharmacology 33:1471–1478. doi:10.1016/0028-3908(94)90051-5
Murray M, Wang SD, Goldberger ME, Levitt P (1990) Modification of astrocytes in the spinal cord following dorsal root or peripheral nerve lesions. Exp Neurol 110:248–257. doi:10.1016/0014-4886(90)90036-R
Nahin RL, Ren K, De Leon M, Ruda M (1994) Primary sensory neurons exhibit altered gene expression in a rat model of neuropathic pain. Pain 58:95–108. doi:10.1016/0304-3959(94)90189-9
Neary JT, Baker L, Jorgensen SL, Norenberg MD (1994) Extracellular ATP induces stellation and increases glial fibrillary acidic protein content and DNA synthesis in primary astrocyte cultures. Acta Neuropathol 87:8–13. doi:10.1007/BF00386249
Neprasova H, Anderova M, Petrik D, Vargova L, Kubinova S, Chvatal A, Sykova E (2007) High extracellular K(+) evokes changes in voltage-dependent K(+) and Na(+) currents and volume regulation in astrocytes. Pflugers Arch 453:839–849. doi:10.1007/s00424-006-0151-9
Neumann S, Doubell TP, Leslie T, Woolf CJ (1996) Inflammatory pain hypersensitivity mediated by phenotypic switch in myelinated primary sensory neurons. Nature 384:360–364. doi:10.1038/384360a0
Noble LJ, Hall JJ, Chen S, Chan PH (1992) Morphologic changes in cultured astrocytes after exposure to glutamate. J Neurotrauma 9:255–267
O’Callaghan JP, Sriram K (2005) Glial fibrillary acidic protein and related glial proteins as biomarkers of neurotoxicity. Expert Opin Drug Saf 4:433–442. doi:10.1517/14740338.4.3.433
Obata H, Eisenach JC, Hussain H, Bynum T, Vincler M (2006) Spinal glial activation contributes to postoperative mechanical hypersensitivity in the rat. J Pain 7:816–822. doi:10.1016/j.jpain.2006.04.004
Ohtori S, Takahashi K, Moriya H, Myers RR (2004) TNF-alpha and TNF-alpha receptor type 1 upregulation in glia and neurons after peripheral nerve injury: studies in murine DRG and spinal cord. Spine 29:1082–1088. doi:10.1097/00007632-200405150-00006
Parpura V, Basarsky TA, Liu F, Jeftinija K, Jeftinija S, Haydon PG (1994) Glutamate-mediated astrocyte-neuron signalling. Nature 369:744–747. doi:10.1038/369744a0
Raghavendra V, Tanga FY, DeLeo JA (2004) Complete Freunds adjuvant-induced peripheral inflammation evokes glial activation and proinflammatory cytokine expression in the CNS. Eur J NeuroSci 20:467–473. doi:10.1111/j.1460-9568.2004.03514.x
Sachs D, Cunha FQ, Poole S, Ferreira SH (2002) Tumour necrosis factor-alpha, interleukin-1beta and interleukin-8 induce persistent mechanical nociceptor hypersensitivity. Pain 96:89–97. doi:10.1016/S0304-3959(01)00433-X
Sanderson MJ, Charles AC, Dirksen ER (1990) Mechanical stimulation and intercellular communication increases intracellular Ca2+ in epithelial cells. Cell Regul 1:585–596
Scemes E, Giaume C (2006) Astrocyte calcium waves: what they are and what they do. Glia 54:716–725. doi:10.1002/glia.20374
Schafers M, Lee DH, Brors D, Yaksh TL, Sorkin LS (2003a) Increased sensitivity of injured and adjacent uninjured rat primary sensory neurons to exogenous tumor necrosis factor-alpha after spinal nerve ligation. J Neurosci 23:3028–3038
Schafers M, Sorkin LS, Geis C, Shubayev VI (2003b) Spinal nerve ligation induces transient upregulation of tumor necrosis factor receptors 1 and 2 in injured and adjacent uninjured dorsal root ganglia in the rat. Neurosci Lett 347:179–182. doi:10.1016/S0304-3940(03)00695-5
Shimizu I, Iida T, Guan Y, Zhao C, Raja SN, Jarvis MF, Cockayne DA, Caterina MJ (2005) Enhanced thermal avoidance in mice lacking the ATP receptor P2X3. Pain 116:96–108. doi:10.1016/j.pain.2005.03.030
Sneyd J, Charles AC, Sanderson MJ (1994) A model for the propagation of intercellular calcium waves. Am J Physiol 266:C293–C302
Sommer C, Myers RR (1995) Neurotransmitters in the spinal cord dorsal horn in a model of painful neuropathy and in nerve crush. Acta Neuropathol 90:478–485. doi:10.1007/BF00294809
Staton PC, Wilson AW, Bountra C, Chessell IP, Day NC (2006) Changes in dorsal root ganglion CGRP expression in a chronic inflammatory model of the rat knee joint: differential modulation by rofecoxib and paracetamol. Eur J Pain 11:283–289
Stevens B, Fields RD (2000) Response of Schwann cells to action potentials in development. Science 287:2267–2271. doi:10.1126/science.287.5461.2267
Steward O, Torre ER, Tomasulo R, Lothman E (1991) Neuronal activity up-regulates astroglial gene expression. Proc Natl Acad Sci USA 88:6819–6823. doi:10.1073/pnas.88.15.6819
Sweitzer SM, Colburn RW, Rutkowski M, DeLeo JA (1999) Acute peripheral inflammation induces moderate glial activation and spinal IL-1beta expression that correlates with pain behavior in the rat. Brain Res 829:209–221. doi:10.1016/S0006-8993(99)01326-8
Tracey DJ, Walker JS (1995) Pain due to nerve damage: are inflammatory mediators involved? Inflamm Res 44:407–411. doi:10.1007/BF01757696
Trebino CE, Stock JL, Gibbons CP, Naiman BM, Wachtmann TS, Umland JP, Pandher K, Lapointe JM, Saha S, Roach ML, Carter D, Thomas NA, Durtschi BA, McNeish JD, Hambor JE, Jakobsson PJ, Carty TJ, Perez JR, Audoly LP (2003) Impaired inflammatory and pain responses in mice lacking an inducible prostaglandin E synthase. Proc Natl Acad Sci USA 100:9044–9049. doi:10.1073/pnas.1332766100
Tsuda M, Shigemoto-Mogami Y, Koizumi S, Mizokoshi A, Kohsaka S, Salter MW, Inoue K (2003) P2X4 receptors induced in spinal microglia gate tactile allodynia after nerve injury. Nature 424:778–783. doi:10.1038/nature01786
Wang XF, Huang LD, Yu PP, Hu JG, Yin L, Wang L, Xu XM, Lu PH (2006) Upregulation of type I interleukin-1 receptor after traumatic spinal cord injury in adult rats. Acta Neuropathol 111:220–228. doi:10.1007/s00401-005-0016-x
Watkins LR, Wiertelak EP, Goehler LE, Smith KP, Martin D, Maier SF (1994) Characterization of cytokine-induced hyperalgesia. Brain Res 654:15–26. doi:10.1016/0006-8993(94)91566-0
Weissner W, Winterson BJ, Stuart-Tilley A, Devor M, Bove GM (2006) Time course of substance P expression in dorsal root ganglia following complete spinal nerve transection. J Comp Neurol 497:78–87. doi:10.1002/cne.20981
Werry EL, Liu GJ, Bennett MR (2006) Glutamate-stimulated ATP release from spinal cord astrocytes is potentiated by substance P. J Neurochem 99:924–936. doi:10.1111/j.1471-4159.2006.04133.x
Winkelstein BA, Rutkowski MD, Sweitzer SM, Pahl JL, DeLeo JA (2001) Nerve injury proximal or distal to the DRG induces similar spinal glial activation and selective cytokine expression but differential behavioral responses to pharmacologic treatment. J Comp Neurol 439:127–139. doi:10.1002/cne.2000
Witting N, Kupers RC, Svensson P, Jensen TS (2006) A PET activation study of brush-evoked allodynia in patients with nerve injury pain. Pain 120:145–154. doi:10.1016/j.pain.2005.10.034
Wolf G, Gabay E, Tal M, Yirmiya R, Shavit Y (2006) Genetic impairment of interleukin-1 signaling attenuates neuropathic pain, autotomy, and spontaneous ectopic neuronal activity, following nerve injury in mice. Pain 120:315–324. doi:10.1016/j.pain.2005.11.011
Xu JT, Xin WJ, Zang Y, Wu CY, Liu XG (2006) The role of tumor necrosis factor-alpha in the neuropathic pain induced by Lumbar 5 ventral root transection in rat. Pain 123:306–321. doi:10.1016/j.pain.2006.03.011
Zelenka M, Schafers M, Sommer C (2005) Intraneural injection of interleukin-1beta and tumor necrosis factor-alpha into rat sciatic nerve at physiological doses induces signs of neuropathic pain. Pain 116:257–263. doi:10.1016/j.pain.2005.04.018
Zhang J, De Koninck Y (2006) Spatial and temporal relationship between monocyte chemoattractant protein-1 expression and spinal glial activation following peripheral nerve injury. J Neurochem 97:772–783. doi:10.1111/j.1471-4159.2006.03746.x
Zhang JM, Wang HK, Ye CQ, Ge W, Chen Y, Jiang ZL, Wu CP, Poo MM, Duan S (2003) ATP released by astrocytes mediates glutamatergic activity-dependent heterosynaptic suppression. Neuron 40:971–982. doi:10.1016/S0896-6273(03)00717-7
Zhang RX, Liu B, Wang L, Ren K, Qiao JT, Berman BM, Lao L (2005) Spinal glial activation in a new rat model of bone cancer pain produced by prostate cancer cell inoculation of the tibia. Pain 118:125–136. doi:10.1016/j.pain.2005.08.001
Zhuang ZY, Wen YR, Zhang DR, Borsello T, Bonny C, Strichartz GR, Decosterd I, Ji RR (2006) A peptide c-Jun N-terminal kinase (JNK) inhibitor blocks mechanical allodynia after spinal nerve ligation: respective roles of JNK activation in primary sensory neurons and spinal astrocytes for neuropathic pain development and maintenance. J Neurosci 26:3551–3560. doi:10.1523/JNEUROSCI.5290-05.2006
Zonta M, Sebelin A, Gobbo S, Fellin T, Pozzan T, Carmignoto G (2003) Glutamate-mediated cytosolic calcium oscillations regulate a pulsatile prostaglandin release from cultured rat astrocytes. J Physiol 553:407–414. doi:10.1113/jphysiol.2003.046706
Acknowledgments
I would like to thank Rikke Hansen, Kristoffer Egerod, and Anne-Marie Heegaard for critical proofreading of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hald, A. Spinal Astrogliosis in Pain Models: Cause and Effects. Cell Mol Neurobiol 29, 609–619 (2009). https://doi.org/10.1007/s10571-009-9390-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-009-9390-6