Abstract
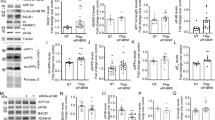
Amyotrophic lateral sclerosis (ALS) is a progressive neurodegenerative disease defined by motor neuron loss. Transgenic mouse model (Tg SOD1G93A) shows pathological features that closely mimic those seen in ALS patients. An hypothetic link between AD and ALS was suggested by finding an higher amount of amyloid precursor protein (APP) in the spinal cord anterior horn neurons, and of Aβ peptides in ALS patients skin. In this work, we have investigated the expression of some genes involved in Alzheimer’s disease, as APP, β- and γ-secretase, in an animal model of ALS, to understand some possible common molecular mechanisms between these two pathologies. For gene expression analysis, we carried out a quantitative RT-PCR in ALS mice and in transgenic mice over-expressing human wild-type SOD1 (Tg hSOD1). We found that APP and BACE1 mRNA levels were increased 1.5-fold in cortical cells of Tg SOD1G93A mice respect to Tg hSOD1, whereas the expression of γ-secretase genes, as PSEN1, PSEN2, Nicastrin, and APH1a, showed no statistical differences between wild-type and ALS mice. Biochemical analysis carried out by immunostaining and western blotting, did not show any significant modulation of the protein expression compared to the genes, suggesting the existence of post-translational mechanisms that modify protein levels.




Similar content being viewed by others
Abbreviations
- APP:
-
Amyloid precursor protein
- BACE1:
-
β-Site APP cleaving enzyme
- ALS:
-
Amyotrophic lateral sclerosis
- CNS:
-
Central nervous system
- PSEN1:
-
Presenilin 1
- PSEN2:
-
Presenilin 2
- NCSTN:
-
Nicastrin
- APH1:
-
Anterior pharynx defective 1
References
Abrahams S, Leigh PN, Goldstein LH (2005) Cognitive change in ALS: a prospective study. Neurology 64:1222–1226
Beauchamp C, Fridovich I (1971) Superoxide dismutase: improved assays and an assay applicable to acrylamide gels. Anal Biochem 44(1):276–287. doi:10.1016/0003-2697(71)90370-8
Bossy-Wetzel E, Schwarzenbacher R, Lipton SA (2004) Molecular pathways to neurodegeneration. Nat Med 10:S2–S9. doi:10.1038/nm1067
Butterfield DA, Bush AI (2004) Alzheimer’s amyloid β-peptide (1–42): involvement of methionine residue 35 in the oxidative stress and neurotoxicity properties of this peptide. Neurobiol Aging 25(5):563–568. doi:10.1016/j.neurobiolaging.2003.12.027
Carunchio I, Pieri M, Ciotti MT, Albo F, Zona C (2007) Modulation of AMPA receptors in cultured cortical neurons induced by the antiepileptic drug levetiracetam. Epilepsia 48(4):654–662. doi:10.1111/j.1528-1167.2006.00973.x
Forman MS, Trojanowski JQ, Lee VM (2004) Neurodegenerative diseases: a decade of discoveries paves the way for therapeutic breakthroughs. Nat Med 10(10):1055–1063. doi:10.1038/nm1113
Hamilton RL, Bowser R (2004) Alzheimer disease pathology in amyotrophic lateral sclerosis. Acta Neuropathol 107:515–522. doi:10.1007/s00401-004-0843-1
Hensley K, Carney JM, Mattson MP, Aksenova M, Harris M, Wu JF, Floyd RA, Butterfield DA (1994) A model for beta-amyloid aggregation and neurotoxicity based on free radical generation by the peptide: relevance to Alzheimer disease. Proc Natl Acad Sci USA 91(8):3270–3274. doi:10.1073/pnas.91.8.3270
Koistinen H, Prinjha R, Soden P, Harper A, Banner SJ, Pradat PF, Loeffler JP, Dingwall C (2006) Elevated levels of amyloid precursor protein in muscle of patients with amyotrophic lateral sclerosis and a mouse model of the disease. Muscle Nerve 34(4):444–450. doi:10.1002/mus.20612
Kolecki R, Lafauci G, Rubenstein R, Mazur-Kolecka B, Kaczmarski W, Frackowiak J (2008) The effect of amyloidosis-beta and ageing on proliferation of neuronal progenitor cells in APP-transgenic mouse hippocampus and in culture. Acta Neuropathol (in press)
Lomen-Hoerth C, Murphy J, Langmore S, Kramer JH, Olney RK, Miller B (2003) Are amyotrophic lateral sclerosis patients cognitively normal? Neurology 60:1094–1097
Misonou H, Morishima-Kawashima M, Ihara Y (2000) Oxidative stress induces intracellular accumulation of amyloid β-protein (Aβ) in human neuroblastoma cells. Biochemistry 39(23):6951–6959. doi:10.1021/bi000169p
Morimoto N, Nagai M, Ohta Y, Miyazaki K, Kurata T, Morimoto M, Murakami T, Takehisa Y, Ikeda Y, Kamiya T, Abe K (2007) Increased autophagy in transgenic mice with a G93A mutant SOD1 gene. Brain Res 1167:112–117. doi:10.1016/j.brainres.2007.06.045
Octave JN (2005) Alzheimer disease: cellular and molecular aspects. Bull Mem Acad R Med Belg 160(10–12):445–449
Ohyagi Y, Yamada T, Nishioka K, Clarke NJ, Tomlinson AJ, Naylor S, Nakabeppu Y, Kira J, Younkin SG (2000) Selective increase in cellular A-beta 42 is related to apoptosis but not necrosis. Neuroreport 11(1):167–171. doi:10.1097/00001756-200001170-00033
Pieri M, Gaetti C, Spalloni A, Cavalcanti S, Mercuri N, Bernardi G, Longone P, Zona C (2003) Alpha-Amino-3-hydroxy-5-methyl-isoxazole-4-propionate receptors in spinal cord motor neurons are altered in transgenic mice overexpressing human Cu, Zn superoxide dismutase (Gly93 → Ala) mutation. Neuroscience 122:47–58. doi:10.1016/j.neuroscience.2003.07.003
Piscopo P, Bernardo A, Calamandrei G, Venerosi A, Valanzano A, Bianchi D, Confaloni A, Minghetti L (2008) Exp Neurol 209(1):192–198. doi:10.1016/j.expneurol.2007.09.014
Gurney ME, Pu H, Chiu AY, Dal Canto MC, Polchow, CY, Alexander DD, Caliendo J, Hentati A, Kwon YW, Deng HX (1994) Motor neuron degeneration in mice that express a human Cu, Zn superoxide dismutase mutation. Science 264:1772–1775
Puttaparthi K, Van Kaer L, Elliott JL (2007) Assessing the role of immuno-proteasomes in a mouse model of familial ALS. Exp Neurol 206(1):53–58. doi:10.1016/j.expneurol.2007.03.024
Ringholz GM, Appel SH, Bradshaw M, Cooke NA, Mosnik DM, Schulz PE (2005) Prevalence and patterns of cognitive impairment in sporadic ALS. Neurology 65:586–590. doi:10.1212/01.wnl.0000172911.39167.b6
Roßner S, Sastre M, Bourne K, Lichtenthaler SF (2006) Transcriptional and translational regulation of BACE1 expression—implications for Alzheimer’s disease. Prog Neurobiol 79(2):95–111. doi:10.1016/j.pneurobio.2006.06.001
Sasaki S, Iwata M (1999) Immunoreactivity of beta-amyloid precursor protein in amyotrophic lateral sclerosis. Acta Neuropathol 97(5):463–468. doi:10.1007/s004010051015
Selkoe DJ (2001) Alzheimer’s disease: genes, proteins, and therapy. Physiol Rev 81(2):741–766
Selkoe DJ (2004) Cell biology of protein misfolding: the examples of Alzheimer’s and Parkinson’s diseases. Nat Cell Biol 6:1054–1061. doi:10.1038/ncb1104-1054
Strong MJ, Grace GM, Orange JB, Leeper HA, Menon RS, Aere C (1999) A prospective study of cognitive impairment in ALS. Neurology 53:1665–1670
Tamaoka A, Matsuno S, Ono S, Shimizu N, Shoji S (2000) Increased amyloid β-protein in the skin of patients with amyotrophic lateral sclerosis. J Neurol 247(8):633–635. doi:10.1007/s004150070133
Velliquette RA, O’Connor T, Vassar R (2005) Energy inhibition elevates β-secretase levels and activity and is potentially amyloidogenic in APP transgenic mice: possible early events in Alzheimer’s disease pathogenesis. J Neurosci 25(47):10874–10883. doi:10.1523/JNEUROSCI.2350-05.2005
Wheaton MW, Salamone AR, Mosnik DM, McDonald RO, Appel SH, Schmolck HI, Ringholz GM, Schulz PE (2007) Cognitive impairment in familial ALS. Neurology 69(14):1411–1417. doi:10.1212/01.wnl.0000277422.11236.2c
Xiong K, Cai H, Luo XG, Struble RG, Clough RW, Yan XX (2007) Mitochondrial respiratory inhibition and oxidative stress elevate beta-secretase (BACE1) proteins and activity in vivo in the rat retina. Exp Brain Res 181(3):435–446. doi:10.1007/s00221-007-0943-y
Yu WH, Cuervo AM, Kumar A, Peterhoff CM, Schmidt SD, Lee JH, Mohan PS, Mercken M, Farmery MR, Tjernberg LO, Jiang Y, Duff K, Uchiyama Y, Näslund J, Mathews PM, Cataldo AM, Nixon RA (2005) Macroautophagy a novel Beta-amyloid peptide-generating pathway activated in Alzheimer’s disease. J Cell Biol 171(1):87–98. doi:10.1083/jcb.200505082
Zhang X, Zhou K, Wang R, Cui J, Lipton SA, Liao FF, Xu H, Zhang YW (2007) Hypoxia-inducible factor 1alpha (HIF-1alpha)-mediated hypoxia increases BACE1 expression and beta-amyloid generation. J Biol Chem 282(15):10873–10880. doi:10.1074/jbc.M608856200
Acknowledgement
This work was supported by MIUR Italy (FIRB: contract RBAU01A7T4 to C.Z.)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Spadoni, O., Crestini, A., Piscopo, P. et al. Gene Expression Profiles of APP and BACE1 in Tg SOD1G93A Cortical Cells. Cell Mol Neurobiol 29, 635–641 (2009). https://doi.org/10.1007/s10571-009-9356-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-009-9356-8