Abstract
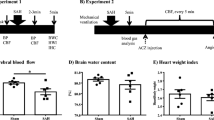
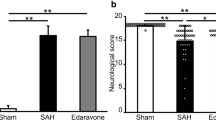
The study aimed to investigate the involvement of cerebral microcirculation turbulence after subarachnoid hemorrhage (SAH). Wistar rats were divided into non-SAH and SAH groups. Autologous arterial hemolysate was injected into rat’s cisterna magna to induce SAH. Changes of pial microcirculation within 2 h were observed. It was found that there were no obvious changes of the diameters, flow velocity, and fluid state of microvessels in non-SAH group. With the exception of rare linear-granular flow in A4 arteriole, linear flow was observed in most of the arterioles. There was no blood agglutination in any of the arterioles. After SAH, abnormal cerebral pial microcirculation was found. Spasm of microvessels, decreased blood flow, and agglutination of red blood cells occurred. Five minutes following the induction of SAH, the diameters of the arterioles and venules significantly decreased. The decreased diameters persisted for 2 h after cisternal injection. Decreased flow velocity of venules was found from 5 to 90 min after induction of SAH. Spasm of the basilar artery and increased brain malondialdehyde were also found after SAH. We concluded that cerebral microcirculation turbulence plays an important role in the development of secondary cerebral ischemia following SAH.




Similar content being viewed by others
References
Asano T (1999) Oxyhemoglobin as the principal cause of cerebral vasospasm: a holistic view of its actions. Crit Rev Neurosurg 9:303–318
Bariskaner H, Ustun ME, Ak A, Yosunkaya A, Ulusoy HB, Gurbilek M (2003) Effects of magnesium sulfate on tissue lactate and malondialdehyde levels after cerebral ischemia. Pharmacology 68:162–168
Dreier JP, Windmuller O, Petzold G, Lindauer U, Einhaupl KM, Dirnagl U (2002) Ischemia triggered by red blood cell products in the subarachnoid space is inhibited by nimodipine administration or moderate volume expansion/hemodilution in rats. Neurosurgery 51:1457–1465
Farrugia A, Shea N (1992) Frozen thawed blood stored at −20°C to −25°C. Med J Aust 157:716–717
Germano A, d’Avella D, Imperatore C, Caruso G, Tomasello F (2000) Time-course of blood-brain barrier permeability changes after experimental subarachnoid haemorrhage. Acta Neurochir (Wien) 142:575–581
Grasso G (2004) An overview of new pharmacological treatments for cerebrovascular dysfunction after experimental subarachnoid hemorrhage. Brain Res Rev 44:49–63
Grubb RL Jr, Raichle ME, Eichling JO, Gado MH (1977) Effects of subarachnoid hemorrhage on cerebral blood volume, blood flow, and oxygen utilization in humans. J Neurosurg 46:446–453
Hino A, Mizukawa N, Tenjin H, Imahori Y, Takemoto S, Yano I, Nakahashi H, Hirakawa K (1989) Postoperative hemodynamic and metabolic changes in patients with subarachnoid hemorrhage. Stroke 20:1504–1510
Ishikawa M, Sekizuka E, Shimizu K, Yamaguchi N, Kawase T (1998) Measurement of RBC velocities in the rat pial arteries with an image-intensified high-speed video camera system. Microvasc Res 56:166–172
Juvela S (2002) Plasma endothelin and big endothelin concentrations and serum endothelin-converting enzyme activity following aneurysmal subarachnoid hemorrhage. J Neurosurg 97:1287–1293
Manno EM (2004) Subarachnoid hemorrhage. Neurol Clin 22:347–366
Megyesi JF, Vollrath B, Cook DA, Findlay JM (2004) In vivo animal models of cerebral vasospasm: a review. Neurosurgery 46:448–461
Nicholls MD, Davies VJ, Au-Yeung D, Wedder C, Lovric A, Klarkowski DB (1989) Transfusion of frozen thawed blood stored at −20°C to −25°C. Lancet 1:71–73
Nihei H, Kassell NF, Dougherty DA, Sasaki T (1991) Does vasospasm occur in small pial arteries and arterioles of rabbits? Stroke 22:1419–1425
Park KW, Metais C, Dai HB, Comunale ME, Sellke FW (2001) Microvascular endothelial dysfunction and its mechanism in a rat model of subarachnoid hemorrhage. Anesth Analg 92:990–996
Pradilla G, Thai QA, Legnani FG, Hsu W, Kretzer RM, Wang PP, Tamargo RJ (2004) Delayed intracranial delivery of a nitric oxide donor from a controlled-release polymer prevents experimental cerebral vasospasm in rabbits. Neurosurgery 55:1393–1400
Satomura Y, Seki J, Ooi Y, Yanagida T, Seiyama A (2004) In vivo imaging of the rat cerebral microvessels with optical coherence tomography. Clin Hemorheol Microcirc 31:31–40
Sehba FA, Mostafa G, Friedrich V Jr, Bederson JB (2005) Acute microvascular platelet aggregation after subarachnoid hemorrhage. J Neurosurg 102:1094–1100
Sun BL, Xia ZL, Yang MF, Qiu PM (2000) Effects of Ginkgo biloba extract on somatosensory evoked potential, nitric oxide levels in serum and brain tissue in rats with cerebral vasospasm after subarachnoid hemorrhage. Clin Hemorheol Microcirc 23:139–144
Sun BL, Zhang SM, Xia ZL, Yang MF, Yuan H, Zhang J, Xiu RJ (2003) The effects of nimodipine on regional cerebral blood flow, brain water and electrolyte contents in rats with subarachnoid hemorrhage. Clin Hemorheol Microcirc 29:337–344
Ueda Y, Wei EP, Kontos HA, Suehiro E, Povlishock JT (2003) Effects of delayed, prolonged hypothermia on the pial vascular response after traumatic brain injury in rats. J Neurosurg 99:899–906
Uhl E, Lehmberg J, Steiger HJ, Messmer K (2003) Intraoperative detection of early microvasospasm in patients with subarachnoid hemorrhage by using orthogonal polarization spectral imaging. Neurosurgery 52:1307–1315
Veelken JA, Laing RJ, Jakubowski J (1995) The Sheffield model of subarachnoid hemorrhage in rats. Stroke 26:1279–1284
Wolf EW, Banerjee A, Soble-Smith J, Dohan FC Jr, White RP, Robertson T (1998) Reversal of cerebral vasospasm using an intrathecally administered nitric oxide donor. J Neurosurg 89:279–288
Yundt KD, Grubb RL Jr, Diringer MN, Powers WJ (1998) Autoregulatory vasodilation of parenchymal vessels is impaired during cerebral vasospasm. J Cereb Blood Flow Metab 18:419–424
Zimmermann M, Seifert V (1998) Endothelin and subarachnoid hemorrhage: an overview. Neurosurgery 43:863–876
Acknowledgments
This work was supported by the National Natural Science Foundation of China (No. 30570651, 30670724) and the Natural Medicine Research Foundation of Shandong, China (2005–231).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Sun, BL., Zheng, CB., Yang, MF. et al. Dynamic Alterations of Cerebral Pial Microcirculation During Experimental Subarachnoid Hemorrhage. Cell Mol Neurobiol 29, 235–241 (2009). https://doi.org/10.1007/s10571-008-9316-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-008-9316-8