Abstract
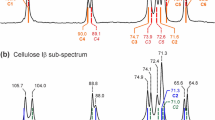
Reducing chain ends in various hydrolyzed cellulose samples were quantitatively analyzed by solid-state 13C NMR. Cellulose di- and tetramers served as model systems to demonstrate that distinct signals at 97 ppm and 92.7 ppm can be detected quantitatively and assigned unambiguously to reducing chain ends in β and α anomers, respectively. In standard Avicel microcrystalline cellulose, the same signals were detected at the expected low intensity; their strength increased significantly upon further hydrolysis. The assignment of the 97 and 92.7 ppm signals to reducing chain ends was confirmed by their absence after ethanolysis, which instead produced an α-ethyl-ether end group signal at 99 ppm. Due to moderate-amplitude chain-end mobility, chain-end signals were relatively enhanced in direct-polarization 13C NMR with heteronuclear Overhauser enhancement. In 13C-enriched hydrolyzed cellulose, the assignment of the C–OH chain-end signals was further corroborated by hydroxyl-proton selection and two-dimensional 13C-13C NMR. From the fractional chain-end signal intensity, the number-average degree of polymerization (DPn) was determined. For Avicel, this yielded DPn = 43 (+ 50, -6), consistent with gel-permeation chromatography but significantly lower than deduced from a more indirect optical method likely hampered by limited chain-end accessibility. After 60 min of hydrolysis of ball-milled Avicel or cellulose from maize, highly reliable values of DPn = 18 ± 3 and 15 ± 3, respectively, were obtained. Solid-state NMR completely avoids the potential loss of low-molar-mass chains in solution-based approaches. The accurate, solvent-free solid-state NMR method introduced here can serve as a primary standard to calibrate other methods for molar-mass determination in hydrolyzed cellulose.
Graphical abstract









Similar content being viewed by others
References
Bennett AE, Rienstra CM, Auger M et al (1995) Heteronuclear decoupling in rotating solids. J Chem Phys 103:6951–6958. https://doi.org/10.1063/1.470372
Bielecki A, Kolbert AC, De Groot HJM et al (1990) Frequency-Switched Lee—Goldburg Sequences in Solids. Adv Magn Opt Reson. https://doi.org/10.1016/b978-0-12-025514-6.50011-3
Caravatti P, Braunschweiler L, Ernst RR (1983) Heteronuclear correlation spectroscopy in rotating solids. Chem Phys Lett 100:305–310. https://doi.org/10.1016/0009-2614(83)80276-0
Chaerunisaa AY, Sriwidodo S, Abdassah M (2020) Microcrystalline Cellulose as Pharmaceutical Excipient. In: Pharmaceutical Formulation Design - Recent Practices. IntechOpen. doi:https://doi.org/10.5772/intechopen.88092
Crawford R (1981) Lignin biodegradation and transformation. Wiley, New York
Daňhelka J, Kössler I, Boháčková V (1976) Determination of molecular weight distribution of cellulose by conversion into tricarbanilate and fractionation. J Polym Sci, Part a: Polym Chem 14:287–298. https://doi.org/10.1002/pol.1976.170140202
Dixon WT, Schaefer J, Sefcik MD et al (1982) Total suppression of sidebands in CPMAS C-13 NMR. J Magn Reson 49:341–345. https://doi.org/10.1016/0022-2364(82)90199-8
Duan P, Schmidt-Rohr K (2017) Composite-pulse and partially dipolar dephased multiCP for improved quantitative solid-state 13C NMR. J Magn Reson 285:68–78. https://doi.org/10.1016/j.jmr.2017.10.010
Duan P, Schmidt-Rohr K (2019) Quick, selective NMR spectra of C-OH moieties in 13C-enriched solids. J Magn Reson 301:80–84. https://doi.org/10.1016/j.jmr.2019.02.007
Engel P, Hein L, Spiess AC (2012) Derivatization-free gel permeation chromatography elucidates enzymatic cellulose hydrolysis. Biotechnol Biofuels 5:77. https://doi.org/10.1186/1754-6834-5-77
Evans R, Wearne RH, Wallis AFA (1989) Molecular weight distribution of cellulose as its tricarbanilate by high performance size exclusion chromatography. J Appl Polym Sci 37:3291–3303. https://doi.org/10.1002/app.1989.070371202
Fung BM, Khitrin AK, Ermolaev K (2000) An improved broadband decoupling sequence for liquid crystals and solids. J Magn Reson 142:97–101. https://doi.org/10.1006/jmre.1999.1896
Gupta R, Lee YY (2009) Mechanism of cellulase reaction on pure cellulosic substrates. Biotechnol Bioeng 102:1570–1581. https://doi.org/10.1002/bit.22195
Hahn EL (1950) Spin Echoes Phys Rev 80:580–594. https://doi.org/10.1103/PhysRev.80.580
Haslinger S, Hietala S, Hummel M et al (2019) Solid-state NMR method for the quantification of cellulose and polyester in textile blends. Carbohydr Polym 207:11–16. https://doi.org/10.1016/j.carbpol.2018.11.052
Holding AJ, Makela V, Tolonen L et al (2016) Solution-state one- and two-dimensional nmr spectroscopy of high-molecular-weight cellulose. Chemsuschem 9:880–892. https://doi.org/10.1002/cssc.201501511
Jeener J, Meier BH, Bachmann P et al (1979) Investigation of exchange processes by two-dimensional NMR spectroscopy. J Chem Phys 71:4546–4553. https://doi.org/10.1063/1.438208
Johnson RL, Anderson JM, Shanks BH et al (2013) Spectrally edited 2D 13C–13C NMR spectra without diagonal ridge for characterizing 13C-enriched low-temperature carbon materials. J Magn Reson 234:112–124. https://doi.org/10.1016/j.jmr.2013.06.006
Kang X, Kirui A, Dickwella Widanage MC et al (2019) Lignin-polysaccharide interactions in plant secondary cell walls revealed by solid-state NMR. Nat Commun 10:347. https://doi.org/10.1038/s41467-018-08252-0
King AWT, Makela V, Kedzior SA et al (2018) Liquid-state NMR analysis of nanocelluloses. Biomacromol 19:2708–2720. https://doi.org/10.1021/acs.biomac.8b00295
Koso T, Rico del Cerro D, Heikkinen S et al (2020) 2D Assignment and quantitative analysis of cellulose and oxidized celluloses using solution-state NMR spectroscopy. Cellulose 27:7929–7953. https://doi.org/10.1007/s10570-020-03317-0
Lu Q, Hu B, Zhang Z-x et al (2018) Mechanism of cellulose fast pyrolysis: the role of characteristic chain ends and dehydrated units. Combust Flame 198:267–277. https://doi.org/10.1016/j.combustflame.2018.09.025
Matsuoka S, Kawamoto H, Saka S (2014) What is active cellulose in pyrolysis? An approach based on reactivity of cellulose reducing end. J Anal Appl Pyrol 106:138–146. https://doi.org/10.1016/j.jaap.2014.01.011
Oberlerchner JT, Rosenau T, Potthast A (2015) Overview of methods for the direct molar mass determination of cellulose. Molecules 20:10313–10341. https://doi.org/10.3390/molecules200610313
Odian G (2004) Principles of polymerization. Wiley-Interscience, Hoboken, New Jersey. https://doi.org/10.1002/047147875X
Pala H, Mota M, Gama F (2007) Enzymatic depolymerisation of cellulose. Carbohydrate Polym 68:101–108. https://doi.org/10.1016/j.carbpol.2006.07.015
Pintiaux T, Heuls M, Vandenbossche V et al (2019) Cellulose consolidation under high-pressure and high-temperature uniaxial compression. Cellulose 26:2941–2954. https://doi.org/10.1007/s10570-019-02273-8
Puri VP (1984) Effect of crystallinity and degree of polymerization of cellulose on enzymatic saccharification. Biotechnol Bioeng 26:1219–1222. https://doi.org/10.1002/bit.260261010
Reich HJ Hans Reich's Collection. NMR Spectroscopy. https://organicchemistrydata.org/hansreich/resources/nmr/?index=nmr_index%2F13C_shift#cdata01. Accessed 05/17 2021
Röder T, Morgenstern B, Schelosky N et al (2001) Solutions of cellulose in N N -Dimethylacetamide/lithium Chloride Studied by Light Scattering Methods. Polymer 42:6765–6773. https://doi.org/10.1016/s0032-3861(01)00170-7
Roslund MU, Tahtinen P, Niemitz M et al (2008) Complete assignments of the 1H and 13C chemical shifts and JH, H coupling constants in NMR spectra of D-glucopyranose and all D-glucopyranosyl-D-glucopyranosides. Carbohydr Res 343:101–112. https://doi.org/10.1016/j.carres.2007.10.008
Rubin EM (2008) Genomics of cellulosic biofuels. Nature 454:841–845. https://doi.org/10.1038/nature07190
Saalwächter K, Burchard W, Klüfers P et al (2000) Cellulose solutions in water containing metal complexes†. Macromol 33:4094–4107. https://doi.org/10.1021/ma991893m
Schelosky N, Roeder T, Baldinger T (1999) Molecular mass distribution of cellulosic products by size exclusion chromatography in DMAc/LiCl Das Papier (Germany):728–738
Schmidt-Rohr K, Mao JD (2002) Efficient CH-group selection and identification in 13C solid-state NMR by dipolar DEPT and 1H chemical-shift filtering. J Am Chem Soc. 124:13938–13948. https://doi.org/10.1021/ja027362m
Schmidt-Rohr K, Spiess HW (1991) Chain diffusion between crystalline and amorphous regions in polyethylene detected by 2D exchange 13C NMR. Macromolecules. 24:5288–5293. https://doi.org/10.1021/ma00019a011
Shlieout G, Arnold K, Muller G (2002) Powder and mechanical properties of microcrystalline cellulose with different degrees of polymerization. AAPS PharmSciTech 3:E11. https://doi.org/10.1208/pt030211
Trache D, Hussin MH, Hui Chuin CT et al (2016) Microcrystalline cellulose: Isolation, characterization and bio-composites application-A review. Int J Biol Macromol 93:789–804. https://doi.org/10.1016/j.ijbiomac.2016.09.056
Tyufekchiev M, Kolodziejczak A, Duan P et al (2019) Reaction engineering implications of cellulose crystallinity and water-promoted recrystallization. Green Chem 21:5541–5555. https://doi.org/10.1039/c9gc02466b
Tyufekchiev M, Ralph K, Duan P et al (2020) Rapid Depolymerization of decrystallized cellulose to soluble products via ethanolysis under mild conditions. Chemsuschem 13:2634–2641. https://doi.org/10.1002/cssc.201903446
VanderHart DL, Atalla RH (1984) Studies of microstructure in native celluloses using solid-state 13C NMR. Macromolecules 17:1465–1472. https://doi.org/10.1021/ma00138a009
Waffenschmidt S, Jaenicke L (1987) Assay of reducing sugars in the nanomole range with 2,2’-bicinchoninate. Anal Biochem 165:337–340. https://doi.org/10.1016/0003-2697(87)90278-8
Wood BF, Conner AH, Hill CG (1989) The heterogeneous character of the dilute acid hydrolysis of crystalline cellulose. J Appl Polym Sci 37:1373–1394. https://doi.org/10.1002/app.1989.070370520
Yang J, Tu M, Xia C et al (2019) Effect of fenton pretreatment on C1 and C6 oxidation of cellulose and its enzymatic hydrolyzability ACS sustainable. Chem Eng 7:7071–7079. https://doi.org/10.1021/acssuschemeng.8b06850
Zhang YH, Lynd LR (2004) Toward an aggregated understanding of enzymatic hydrolysis of cellulose: noncomplexed cellulase systems. Biotechnol Bioeng 88:797–824. https://doi.org/10.1002/bit.20282
Zhang YH, Lynd LR (2005) Determination of the number-average degree of polymerization of cellodextrins and cellulose with application to enzymatic hydrolysis. Biomacromol 6:1510–1515. https://doi.org/10.1021/bm049235j
Zhang YHP, Cui J, Lynd LR et al (2006) A transition from cellulose swelling to cellulose dissolution byo-phosphoric acid: evidence from enzymatic hydrolysis and supramolecular structure. Biomacromolecules 7:644–648. https://doi.org/10.1021/bm050799c
Funding
The solid-state NMR spectrometer used in this work was funded by the NSF MRI program (Award No. 1726346). Partial student support for MVT was provided by the U.S. National Science Foundation (ENG/#1554283).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yuan, S., Tyufekchiev, M.V., Timko, M.T. et al. Direct quantification of the degree of polymerization of hydrolyzed cellulose by solid-state NMR spectroscopy. Cellulose 29, 2131–2144 (2022). https://doi.org/10.1007/s10570-022-04433-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-022-04433-9