Abstract
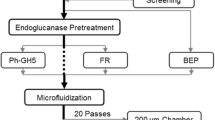
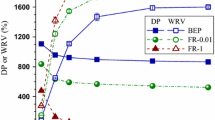
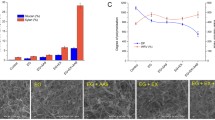
A GH5 hyperthermostable endoglucanase from the archaeon Pyrococcus horikoshii (Ph-GH5) and a commercial endoglucanase FR were used to treat bleached eucalyptus pulp (BEP) fibers to produce cellulose nanofibrils (CNFs) through subsequent microfluidization. Enzymatic treatments facilitated CNF production due to the reduced degree of polymerization (DP) of the fibers. SEM imaging indicated that FR reduced fiber DP drastically and resulted in much shorter fibers than with Ph-GH5, even at very low dosages (1 mg protein/g fiber) of FR treatment compared with a high dosage (10 mg protein/g fiber) of Ph-GH5. The fibers treated with FR were much more uniform in length perhaps due to the presence of exoglucanase and beta-glucosidase saccharifying short microfibers into glucose. TEM imaging indicated that Ph-GH5 produced longer and entangled CNFs than FR with the same number of microfluidization passes. However, the CNF diameters were approximately the same for all CNFs from enzyme-treated fibers using both endoglucanases at two dosages (1 or 10 mg protein/g fiber). CNFs produced from BEP fibers without enzymatic treatment showed larger diameters than those with enzymatic treatment.







Similar content being viewed by others
References
Abdul Khalil HPS, Bhat AH, Ireana Yusra AF (2012) Green composites from sustainable cellulose nanofibrils: a review. Carbohydr Polym 87(2):963–979
Abdul Khalil HPS, Davoudpour Y, Islam MN, Mustapha A, Sudesh K, Dungani R, Jawaid M (2014) Production and modification of nanofibrillated cellulose using various mechanical processes: a review. Carbohydr Polym 99:649–665
Ando S, Ishida H, Kosugi Y, Ishikawa K (2002) Hyperthermostable endoglucanase from Pyrococcus horikoshii. Appl Environ Microbiol 68(1):430–433
Belbekhouche S, Bras J, Siqueira G, Chappey C, Lebrun L, Khelifi B, Marais S, Dufresne A (2011) Water sorption behavior and gas barrier properties of cellulose whiskers and microfibrils films. Carbohydr Polym 83(4):1740–1748
Bradford M (1976) A rapid and sensitive for the quantitation of microgram quantitites of protein utilizing the principle of protein-dye binding. Anal Biochem 72(1–2):248–254
de Souza Lima MM, Borsali R (2004) Rodlike cellulose microcrystals: structure, properties, and applications. Macromol Rapid Commun 25:771–787
Eichhorn SJ, Dufresne A, Aranguren M, Marcovich NE, Capadona JR, Rowan SJ, Weder C, Thielemans W, Roman M, Renneckar S et al (2010) Review: current international research into cellulose nanofibres and nanocomposites. J Mater Sci 45(1):1–33
Fukuzumi H, Saito T, Wata T, Kumamoto Y, Isogai A (2009) Transparent and high gas barrier films of cellulose nanofibers prepared by TEMPO-mediated oxidation. Biomacromolecules 10:162–165
Ghose TK (1987) Measurement of cellulase activities. Pure Appl Chem 59:257–268
Henriksson M, Henriksson G, Berglund LA, Lindström T (2007) An environmentally friendly method for enzyme-assisted preparation of microfibrillated cellulose (MFC) nanofibers. Eur Polym J 43:3434–3441
Henriksson M, Berglund LA, Isaksson P, Lindström T, Nishino TD (2008) Cellulose nanopaper structures of high toughness. Biomacromolecules 9:1579–1585
Kang HJ, Ishikawa K (2007) Analysis of active center in hyperthermophilic cellulase from Pyrococcus horikoshii. J Microbiol Biotechnol 17(8):1249–1253
Kang HJ, Uegaki K, Fukada H, Ishikawa K (2007) Improvement of the enzymatic activity of the hyperthermophilic cellulase from Pyrococcus horikoshii. Extremophiles 11(2):251–256
Kashima Y, Mori K, Fukada H, Ishikawa K (2005) Analysis of the function of a hyperthermophilic endoglucanase from Pyrococcus horikoshii that hydrolyzes crystalline cellulose. Extremophiles 9(1):37–43
Kim H-W, Mino K, Ishikawa K (2008) Crystallization and preliminary X-ray analysis of endoglucanase from Pyrococcus horikoshii. Acta Crystallogr Sect F 64(12):1169–1171
López-Rubio A, Lagaron JM, Ankerfors M, Lindström T, Nordqvist D, Mattozzi A, Hedenqvist MS (2007) Enhanced film forming and film properties of amylopectin using micro-fibrillated cellulose. Carbohydr Polym 68(4):718–727
Mazumder BB, Ohtani Y, Cheng Z, Sameshima K (2000) Combination treatment of kenaf bast fiber for high viscosity pulp. J Wood Sci 46:364–370
Moon RJ, Martini A, Nairn J, Simonsen J, Youngblood J (2011) Cellulose nanomaterials review: structure, properties and nanocomposites. Chem Soc Rev 40:3941–3994
Nogi M, Iwamoto S, Nakagaito AN, Yano H (2009) Optically Transparent Nanofiber Paper. Adv Mater 21(16):1595–1598
Pääkko M, Ankerfors M, Kosonen H, Nykänen A, Ahola S, Österberg M, Ruokolainen J, Laine J, Larsson PT, Ikkala O et al (2007) Enzymatic hydrolysis combined with mechanical shearing and high-pressure homogenization for nanoscale cellulose fibrils and strong gels. Biomacromolecules 8:1934–1941
Qing Y, Sabo R, Zhu JY, Agarwal U, Cai Z, Wu Y (2013) A comparative study of cellulose nanofibrils disintegrated via multiple processing approaches. Carbohydr Polym 97(1):226–234
Siddiqui N, Mills RH, Gardner DJ, Bousfield D (2010) Production and characterization of cellulose nanofibers from wood pulp. J Adhes Sci Technol 25(6–7):709–721
Sluiter A, Hames B, Ruiz R, Scarlata C, Sluiter J, Templeton D, Crocker D (2011) Determination of structural carbohydrates and Lignin in Biomass Laboratory Analytical Procedure (LAP). NREL Technical Report NREL/TP-510-42618 (Version 07-08-2011)
Studier FW (2005) Protein production by auto-induction in high-density shaking cultures. Protein Expr Purif 41(1):207–234
Svagan AJ, Azizi Samir MAS, Berglund LA (2007) Biomimetic polysaccharide nanocomposites of high cellulose content and high toughness. Biomacromolecules 8(8):2556–2563
Tanpichai S, Quero F, Nogi M, Yano H, Young RJ, Lindström T, Sampson WW, Eichhorn SJ (2012) Effective young’s modulus of bacterial and microfibrillated cellulose fibrils in fibrous networks. Biomacromolecules 13(5):1340–1349
TAPPI Standard Test Method (1999) T230 om-99, Viscosity of pulp (capillary viscometer method)
Teugjas H, Väljamäe P (2013) Selecting β-glucosidases to support cellulases in cellulose saccharification. Biotechnol Biofuels 6:105
Wang QQ, Zhu JY, Reiner RS, Verrill SP, Baxa U, McNeil SE (2012) Approaching zero cellulose loss in cellulose nanocrystal (CNC) production: recovery and characterization of cellulosic solid residues (CSR) and CNC. Cellulose 19(6):2033–2047
Zhu JY, Sabo R, Luo X (2011) Integrated production of nano-fibrillated cellulose and cellulosic biofuel (ethanol) by enzymatic fractionation of wood fibers. Green Chem 13(5):1339–1344
Acknowledgments
We acknowledge the financial supports by a USDA Agriculture and Food Research Initiative (AFRI) Competitive Grant (No. 2011-67009-20056), Chinese Scholarship Council (CSC), National Natural Science Foundation of China (Grant Nos. 31070512 and 31370571). The funding from these programs made the visiting appointment of Wang at the USDA Forest Products Laboratory (FPL) possible. We also acknowledge Thomas Kuster of the Analytical Chemistry and Microscopy Lab of FPL for SEM imaging and Debra Sherman of DS imaging LLC, West Lafayette, IN, for TEM imaging.
Author information
Authors and Affiliations
Corresponding authors
Additional information
This work was conducted on official government time of Zhu, Kersten, Mozuch, and Sabo while Wang was a visiting student at the US Forest Service, Forest Products Lab.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, W., Mozuch, M.D., Sabo, R.C. et al. Production of cellulose nanofibrils from bleached eucalyptus fibers by hyperthermostable endoglucanase treatment and subsequent microfluidization. Cellulose 22, 351–361 (2015). https://doi.org/10.1007/s10570-014-0465-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-014-0465-2