Abstract
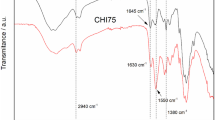
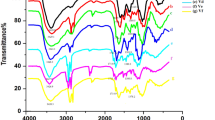
Four novel hydrogels composed of carboxymethyl chitosan (CM-chitosan) cross-linked with various contents of terephthaloyl thiourea moieties (TTUCM-chitosan-1, TTUCM-chitosan-2, TTUCM-chitosan-3, TTUCM-chitosan-4) have been successfully synthesized. The hydrogels were characterized by elemental analyses, FTIR, 13C NMR, SEM, XRD, solubility and swell ability in various solvents. The antimicrobial activities of these hydrogels against three crop-threatening pathogenic fungi (Aspergillus fumigatus, Geotrichum candidum and Candida albicans) and against three bacterial species (Bacillis subtilis, Staphylococcus aureus and Escherichia coli) were investigated. The hydrogels showed higher antibacterial activity than the parent CM-chitosan as judged by their higher inhibition zone diameter and lower minimum inhibition concentration. They are more active against Gram-positive bacteria than against Gram-negative bacteria. The results also indicated that the hydrogels have effective antifungal activity as compared with CM-chitosan. The antimicrobial activity of the hydrogels increased with increasing their cross-linking density.






Similar content being viewed by others
References
Aranaz I, Harris R, Heras A (2010) Chitosan amphiphilic derivatives. Chemistry and applications. Curr Org Chem 14:308–330
Arslan H, Duran N, Borekci G, Ozer CK, Akbay C (2009) Antimicrobial activity of some thiourea derivatives and their nickel and copper complexes. Molecules 14:519–527
Avadi MR, Sadeghi AMM, Tahzibi A, Bayati KH, Ponladzadeh M, Zohuriaan-Mehr MJ, Rafiee-Tehran M (2004) Antimicrobial action of novel chitin derivative. Eur Polym J 40:1355–1361
Bidgoli H, Zamani A, Taherzadeh MJ (2010) Effect of carboxymethylation conditions on the water-binding capacity of chitosan-based superabsorbents. Carbohydr Res 345:2683–2689
Chen XG, Park HJ (2003) Chemical characterization of O-carboxymethyl chitosan related to the preparation conditions. Carbohydr Polym 53:355–359
Criado JJ, Rodriguez-Femandez E, Garcia E, Hermosa MR, Monte E (1998) Thiourea derivatives of α- aminoacids. Synthesis and characterization of Ni(II), Cu(II) and Pt(II) complexes with l-valinate derivatives. Antifungal activity. J Inorg Biochem 69:113–119
Cuero RG, Osuji G, Washington A (1991) N-carboxymethylchitosan inhibition of aflatoxin production: role of zinc. Biotechnol Lett 13:441–444
Damyanova S, Gomez LM, Banares MA, Fierro JLG (2000) Thermal stability of titania-supported 12-molybdophosphoric heteropoly compounds. Chem Mater 12:501–510
El-Ghaouth A, Arul J, Grenier J, Asselin A (1992) Antifungal activity of chitosan on two postharvest pathogens of strawberry fruits. Phytopathology 82:398–402
Emara AAA, Tawab MA, El-ghamry MA, Elsabee MZ (2011) Metal uptake by chitosan derivatives and structure studies of the polymer metal complexes. Carbohydr Polym 83:192–202
Eweis M, Elkholy SS, Elsabee MZ (2006) Antifungal efficacy of chitosan and its thiourea derivatives upon the growth of some sugar-beet pathogens. Int J Biol Macromol 38:1–8
Eyler RW, Kluge ED, Diephuis F (1947) Determination of degree of substitution of sodium carboxymethyl cellulose. Anal Chem 79:24–27
Feng QL, Wu J, Chen GQ, Cui FZ, Kim TN, Kim JO (2000) A mechanistic study of the antibacterial effect of silver ions on Escherichia coli and Staphylococcus aureus. J Biomed Mat Res 52:662–668
Guilherme MR, Reis AV, Takahashi SH, Rubira AF, Feitosa JPA, Muniz EC (2005) Synthesis of a novel superabsorbent hydrogel by copolymerization of acrylamide and cashew gum modified with glycidyl methacrylate. Carbohydr Polym 61:464–471
Hadwiger LA, Kendra DF, Fristensky BW, Wagoner W (1986) Chitosan both activates genes in plants and inhibits RNA synthesis in fungi. In: Muzzarelli RAA, Jeuniaux C, Gooday CW (eds) Chitin in nature and technology, Plenum Press, New York, pp 209–214
Hongmei K, Xie J (2003) Effect of concentration and pH of solutions on the absorbency of polyacrylate superabsorbents. J Appl Polym Sci 88:494–499
Huehr OF, Marsh JL, Mizzoni RH, Mull RP, Schroeder DC, Troxell HA, Scholz CR (1953) A new class of antitubercular compounds. J Am Chem Soc 75:2274–2275
Mohamed NA, Abd El-Ghany NA (2012) Preparation and antimicrobial activity of some carboxymethyl chitosan acyl thiourea derivatives. Int J Biol Macromol 50:1280–1285
Mourya VK, Inamdar NN, Tiwari A (2010) Carboxymethyl chitosan and its applications. Adv Mat Lett 1:11–33
Nester EW, Anderson DG, Roberts E, Pearshall NN, Nester MT (2003) Synthesis, characterization and antifungal properties of (N,O-(acyl)-N-trimethyl) chitosan chloride. Microbiology. McGraw-Hill, Boston, pp 518–524
Phuong T, Khac-Minh T, Van Hal NT, Phuong HT (2004) Synthesis and antifungal activities of phenylenedithioureas. Bioorg Med Chem Lett 14:653–656
Rahman A, Choudhary MI, Thompson WJ (2001) Bioassay techniques for drug development, Harwood Academic Publishers, The Netherlands 16, pp 2024–2027
Ramadus K, Suresh G, Janarthanan N, Masilamani S (1998) Antifungal activity of 1,3-disubstituted symmetrical and unsymmetrical thioureas. Pestic Sci 52:145–151
Rathore HS, Mittal S, Kumar S (2000) Synthesis, characterization and antifungal activities of 3d-transition metal complexes of 1-acetylpiperazinyldithiocarbamate, M(acpdtc)2. Pestic Res J 12:103–107
Ravi Kumar MNV (2000) A review of chitin and chitosan applications. React Funct Polym 46:1–27
Xie WM, Xu PX, Wang W, Liu Q (2002) Preparation and antibacterial activity of a water-soluble chitosan derivative. Carbohydr Polym 50:35–40
Yoshimura T, Uchikoshi I, Yoshiura Y, Fujioka R (2005) Effects of modified vermiculite on water absorbency and swelling behavior of chitosan-g-poly (acrylic acid)/vermiculite superabsorbent composite. Carbohydr Polym 61:322–326
Zamani A, Henriksson D, Taherzadeh MJ (2010) A new foaming technique for production of superabsorbents from carboxymethyl chitosan. Carbohydr Polym 80:1091–1101
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mohamed, N.A., Abd El-Ghany, N.A. Synthesis and antimicrobial activity of some novel terephthaloyl thiourea cross-linked carboxymethyl chitosan hydrogels. Cellulose 19, 1879–1891 (2012). https://doi.org/10.1007/s10570-012-9789-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-012-9789-y