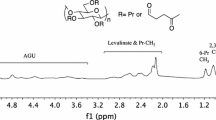
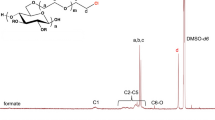
Abstract
The regioselective esterification of cellulose by reaction with bulky acyl halides including pivaloyl chloride, adamantoyl chloride and 2,4,6-trimethylbenzoyl chloride was studied. Functionalization conditions to achieve a given degree of substitution (DS) and the resulting ester substitution pattern were described in detail. One- and two-dimensional NMR spectroscopy techniques were used to confirm the structure of the esters obtained. We compared the effects on substitution of using different esterifying reagents and solvent systems including DMAc/LiCl, DMSO/TBAF, and ionic liquids (ILs).









Similar content being viewed by others
References
Cai S, Hakomori S, Toyokuni T (1992) Application of protease-catalyzed regioselective esterification in synthesis of 6′-deoxy-6′-fluoro- and 6-deoxy-6-fluorolactosides. J Org Chem 57(12):3431–3437
Camacho Gomez JA, Erler UW, Klemm DO (1996) 4-Methoxy substituted trityl groups in 6-O protection of cellulose: homogeneous synthesis, characterization, detritylation. Macromol Chem Phys 197(3):953–964
Chan BKM, Chang N-H, Grimmett MR (1977) The synthesis and thermolysis of imidazole quaternary salts. Aust J Chem 30(9):2005–2013
Corey EJ, Achiwa K, Katzenellenbogen JA (1969) Total synthesis of dl-sirenin. J Am Chem Soc 91(15):4318–4320
Dawsey TR, McCormick CL (1990) The lithium chloride/dimethylacetamide solvent for cellulose: a literature review. Polym Rev 30:405–440
Edgar KJ, Arnold KM, Blount WW, Lawniczak JE, Lowman DW (1995) Synthesis and properties of cellulose acetoacetates. Macromolecules 28(12):4122–4128
Einfeldt L, Guenther W, Klemm D, Heublein B (2005) Peracetylated cellulose: end group modification and structural analysis by means of 1H-NMR spectroscopy. Cellulose 12(1):15–24
El Seoud OA, Koschella A, Fidale LC, Dorn S, Heinze T (2007) Applications of ionic liquids in carbohydrate chemistry: a window of opportunities. Biomacromolecules 8(9):2629–2647. doi:10.1021/bm070062i
Graebner D, Liebert T, Heinze T (2002) Synthesis of novel adamantoyl cellulose using differently activated carboxylic acid derivatives. Cellulose 9(2):193–201
Granstroem M, Kavakka J, King A, Majoinen J, Maekelae V, Helaja J, Hietala S, Virtanen T, Maunu S-L, Argyropoulos DS, Kilpelaeinen I (2008) Tosylation and acylation of cellulose in 1-allyl-3-methylimidazolium chloride. Cellulose 15(3):481–488
Hattori A, Kawahara T, Uemoto T, Suzuki F, Tada H, Ito S (2000) Ultrathin SiOx film coating effect on the wettability change of TiO2 surfaces in the presence and absence of UV light illumination. J Colloid Interface Sci 232(2):410–413
Heinze T (2004) Chemical functionalization of cellulose. Dumitriu polysaccharides 551–590
Heinze T, Liebert T (2001a) Unconventional methods in cellulose esterification. Prog Polym Sci 26:1689–1762
Heinze T, Liebert T (2001b) Unconventional methods in cellulose functionalization. Prog Polym Sci 26(9):1689–1762
Heinze T, Dicke R, Koschella A, Kull AH, Klohr E-A, Koch W (2000) Effective preparation of cellulose derivatives in a new simple cellulose solvent. Macromol Chem Phys 201(6):627–631
Hussain MA, Liebert T, Heinze T (2004) Acylation of cellulose with N, N′-carbonyldiimidazole-activated acids in the novel solvent dimethyl sulfoxide/Tetrabutylammonium fluoride. Macromol Rapid Commun 25(9):916–920
Iwata IO T, Okamura K, Azuma J, Tanaka F (1996) Molecular and crystal structure of cellulose acetate dipropanoate (CADP, 6-O-Acetyl-2, 3-di-O-propanoyl cellulose). Cellulose 3:107–124
Iwata K, Azuma J, Tanaka F (1996) Molecular and crystal structure of cellulose propanoate diacetate (CPDA, 2, 3-Di-O-acetyl-O-propanoyl cellulose). Cellulose 3:91–106
Iwata T, Azuma J, Okamura K, Muramoto M, Chun B (1992) Preparation and NMR assignments of cellulose mixed esters regioselectively substituted by acetyl and propanoyl groups. Carbohydr Res 224:277–283
Iwata T, Fukushima A, Okamura K, Azuma J (1997) DSC study on regioselectively substituted cellulose heteroesters. J Appl Polym Sci 65:1511–1515
Kamide K, Saito M (1994) Recent advances in molecular and supermolecular characterization of cellulose and cellulose derivatives. In: Macromolecular symposia 83 (international conference on advanced polymer materials, 1993), pp 233–271
Klemm DO (1998) Regiocontrol in cellulose chemistry: principles and examples of etherification and esterification. In: ACS symposium series 688 (cellulose derivatives), pp 19–37
Klemm D, Heinze T, Philipp B, Wagenknecht W (1997) New approaches to advanced polymers by selective cellulose functionalization. Acta Polym 48:277–297
Kohler S, Heinze T (2007) New solvents for cellulose: dimethyl sulfoxide/ammonium fluorides. Macromol Biosci 7(3):307–314. doi:10.1002/mabi.200600197
Koschella A, Klemm D (1997) Silylation of cellulose regiocontrolled by bulky reagents and dispersity in the reaction media. In: Macromolecular symposia 120 (functional polysaccharides II), pp 115–125
Koschella A, Heinze T, Klemm D (2001) First synthesis of 3-O-functionalized cellulose ethers via 2, 6-di-O-protected silyl cellulose. Macromol Biosci 1(1):49–54
Liang T-s, Hu Y-l (2007) Preparation of 2, 4, 6-trimethylbenzoyl chloride. Huaxue Gongchengshi 21(11):11–12
Liebert T, Heinze T (2005) Tailored cellulose esters: synthesis and structure determination. Biomacromolecules 6:333–340
Liebert T, Wotschadlo J, Gericke M, Kohler S, Laudeley P, Heinze T (2009) Modification of cellulose in ionic liquids towards biomedical applications. In: Edgar KJ, Heinze T, Buchanan CM (eds) Polysaccharide materials: performance by design, vol 1017. American Chemical Society, Washington, DC
Liebert T, Heinze TL, Edgar KJ (2010) Cellulose solvents: for analysis, shaping, and chemical modification. In: ACS symposium series, vol 1033. American Chemical Society, Washington, DC
Matsushita Y-I, Sugamoto K, Kita Y, Matsui T (1997) Silica gel-catalyzed beta-O-glucosylation of alcohols with 1, 2-anhydro-3, 4, 6-tri-O-pivaloyl-alpha-d-glucopyranose. Tetrahedron Lett 38(50):8709–8712
Oestlund A, Lundberg D, Nordstierna L, Holmberg K, Nyden M (2009) Dissolution and gelation of cellulose in TBAF/DMSO solutions: the roles of fluoride ions and water. Biomacromolecules 10(9):2401–2407
Petzold K, Koschella A, Klemm D, Heublein B (2003) Silylation of cellulose and starch—selectivity, structure analysis, and subsequent reactions. Cellulose 10(3):251–269
Rao CB, Chinnababu B, Venkateswarlu Y (2009) An efficient protocol for alcohol protection under solvent- and catalyst-free conditions. J Org Chem 74(22):8856–8858
Sakairi N, Nomura K, Nishi N, Tokhura S (1997) A novel procedure for the preparation of fully 6-O-pivaloylated derivatives of cyclodextrin. Carbohydr Lett 2(4):253–258
Schlufter K, Schmauder H-P, Dorn S, Heinze T (2006) Efficient homogeneous chemical modification of bacterial cellulose in the ionic liquid 1-N-butyl-3-methylimidazolium chloride. Macromol Rapid Commun 27(19):1670–1676
Schnabelrauch M, Vogt S, Klemm D, Nehls I, Philipp B (1992) Readily hydrolyzable cellulose esters as intermediates for the regioselective derivatization of cellulose. Die Angewandte Makromolekulare Chemie 198:155–164
Sheldon R (2001) Catalytic reactions in ionic liquids. Chem Commun (23):2399–2407
Silverstein RM, Webster FX, Kiemle DJ (2005) Spectrometric identification of organic compounds 87–88
Sun H, DiMagno SG (2005) Anhydrous tetrabutylammonium fluoride. J Am Chem Soc 127(7):2050–2051. doi:10.1021/ja0440497
Swatloski RP, Spear SK, Holbrey JD, Rogers RD (2002) Dissolution of cellulose with ionic liquids. J Am Chem Soc 124(18):4974–4975
Takimoto S, Inanaga J, Katsuki T, Yamaguchi M (1976) Preparation of hindered esters from acid chlorides and alcohols in the presence of silver cyanide. Bull Chem Soc Japan 49(8):2335–2336
Tasaka K et al (2007) Cellulose ester film, its manufacturing method, optical retardation film, optical compensation sheet, elliptic polarizing plate, and image display. US Patent
Tsunashima Y, Hattori K (2000) Substituent distribution in cellulose acetates: its control and the effect on structure formation in solution. J Colloid Interface Sci 228(2):279–286
Acknowledgments
We thank ICTAS and MII at Virginia Tech for their financial, facilities, and educational support, Mr. Mark Flynn for performing SEC measurements and Dr. Hugo Azurmendi for performing HMBC measurements.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xu, D., Li, B., Tate, C. et al. Studies on regioselective acylation of cellulose with bulky acid chlorides. Cellulose 18, 405–419 (2011). https://doi.org/10.1007/s10570-010-9476-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-010-9476-9