Abstract
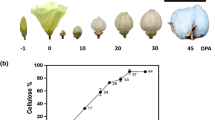
Cotton fiber maturity is a major yield component and an important fiber quality trait that is directly linked to the quantity of cellulose deposited during the secondary cell wall (SCW) biogenesis. Cotton fiber development consists of five major overlapping stages: differentiation, initiation, polar elongation, secondary cell wall development, and maturation. The transition period between 16 and 21 dpa (days post anthesis) is regarded to represent a major developmental stage between the primary cell wall and the SCW. Fourier Transform Infrared spectroscopy was used to investigate the structural changes that occur during the different developmental stages. The IR spectra of fibers harvested at different stages of development (10, 14, 17, 18, 19, 20, 21, 24, 27, 30, 36, 46, and 56 dpa) show the presence of vibrations located at 1,733 cm−1 (C=O stretching originating from esters or amides) and 1,534 cm−1 (NH2 deformation corresponding to proteins or amino acids). The results converge towards the conclusion that the transition phase between the primary cell wall and the secondary cell wall occurs between 17 and 18 dpa in fibers from TX19 cultivar, while this transition occurs between 21 and 24 dpa in fibers from TX55 cultivar.





Similar content being viewed by others
References
Abidi N, Hequet E, Cabrales L, Gannaway J, Wilkins T, Wells LW (2008) Evaluating cell wall structure and composition of developing cotton fibers using Fourier transform infrared spectroscopy and thermogravimetric analysis. J Appl Polym Sci 107:476–486
Carpita NC, Defernez M, Findlay K, Wells B, Shoue DA, Catchpole G, Wilson RH, McCann MC (2001) Cell wall architecture of the elongating Maize coleoptile. Plant Physiol 127:551–565
Chen L, Carpita NC, Reiter W-D, Wilson RH, Jeffries C, McCann MC (1998) A rapid method to screen for cell-wall mutants using discriminant analysis of Fourier transform spectra. Plant J 16(3):385–392
Gokani SJ, Kumar R, Thaker VS (1998) Potential role of abscisic acid in cotton fiber and ovule development. J Plant Growth Regul 17:1–5
Haigler CH, Zhang D, Wilkerson CG (2005) Biotechnological improvement of cotton fibre maturity. Physiol Plant 124:285–294
Hsieh Y-L, Hu X-P, Nguyen A (1997) Strength and crystalline structure of developing Acala cotton. Textile Res J 67(7):529–536
Huwyler HR, Franz G, Meier H (1979) Changes in the composition of cotton fibre cell walls during development. Planta 146:635–642
Ilharco LM, Garcia AR, Lopez da Silva J, Vieira Ferreira LF (1997) Infrared approach to the study of adsorption on cellulose: influence of cellulose crystallinity on the adsorption of benzophenone. Langmuir 13:4126–4132
Liang C-Y, Marchessault RH (1959) Infrared spectra of crystalline polysaccharides. II. Native celluloses in the region from 640 to 1700 cm−1. J Polym Sci XXXIX:269–278
Maltby D, Carpita NC, Montezinos D, Kulow C, Delmer DP (1979) β-1, 3-glucan in developing cotton fibers, structure, localization, and relationship of synthesis to that of secondary wall cellulose. Plant Physiol 63:1158–1164
McCann MC, Hammouri N, Wilson R, Belton P, Roberts K (1992) Fourier transform infrared microspectroscopy is a new way to look at plant cell walls. Plant Physiol 100:1940–1947
McCann MC, Stacey NJ, Wilson R, Roberts K (1993) Orientation of macromolecules in the walls of elongating carrot cells. J Cell Sci 106:1347–1356
McCann MC, Chen L, Roberts L, Kemsley EK, Sene C, Carpita NC, Stacey NJ, Wilson RH (1997) Infrared microspectroscopy: sampling heterogeneity in plant cell wall composition and architecture. Physiol Plant 100:729–738
McCann MC, Defernez M, Urbanowicz BR, Tewari JC, Langewisch T, Olek A, Wells B, Wilson RH, Carpita NC (2007) Neural network analyses of infrared spectra for classifying cell wall architectures. Plant Physiol 143:1314–1326
Meinert MC, Delmer DP (1977) Changes in biochemical composition of the cell wall of the cotton fiber during development. Plant Physiol 59:1088–1097
Muller LL, Jacks TJ (1975) Rapid chemical dehydration of samples for electron microscopic examinations. J Histochem Cytochem 23(2):107–110
Rajasekaran K, Muir AJ, Ingber BF, French AD (2006) A dehydration method for immature or wet cotton fibers for light and electron microscopy. Textile Res J 76(6):514–518
Salmén L, Åkerholm M, Hinterstoisser B (2005) Two-dimensional Fourier transform infrared spectroscopy applied to cellulose and paper. In: Dumitriu S (ed) Polysaccharides, structural diversity and functional versatility, 2nd edn. Marcel Dekker, New York, pp 159–187
Schwanninger M, Rodrigues JC, Pereira H, Hinterstoisser B (2004) Effects of short-time vibratory ball milling on the shape of FT-IR spectra of wood and cellulose. Vib Spectrosc 36:23–40
Séné CFB, McCann MC, Wilson RH, Grinter R (1994) Fourier-transform Raman and Fourier-transform infrared spectroscopy an investigation of five higher plant cell walls and their components. Plant Physiol 106:1623–1631
Timpa JD, Triplett BA (1993) Analysis of cell-wall polymers during cotton fiber development. Planta 189:101–108
Tokumoto H, Wakabayashi K, Kamisaka S, Hoson T (2002) Changes in the sugar composition and molecular mass distribution of matrix polysaccharides during cotton fiber development. Plant Cell Physiol 43(4):411–418
Wilkins TA, Jernstedt JA (1999) Molecular genetics of developing cotton fibers. In: Basra AS (ed) Cotton fibers, developmental biology, quality improvement, and textile processing. Food Products Press, New York, pp 231–269
Yong W, Link B, O’Malley R, Tewari J, Hunter CT, C-An Lu, Li X, Bleecker AB, Koch KE, McCann MC, McCarty DR, Patterson SE, Reiter W-D, Staiger C, Thomas SR, Vermerris W, Carpita NC (2005) Genomics of plant cell wall biogenesis. Planta 221:747–751
Acknowledgments
The authors would like to thank the Texas Department of Agriculture/Food and Fibers Research Grant Program.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Abidi, N., Cabrales, L. & Hequet, E. Fourier transform infrared spectroscopic approach to the study of the secondary cell wall development in cotton fiber. Cellulose 17, 309–320 (2010). https://doi.org/10.1007/s10570-009-9366-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-009-9366-1