Abstract
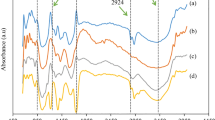
The molecular and physical structures of cellulosic rayon fiber during the stepwise heat treatment for the fabrication of activated carbon fiber (ACF) were researched by Fourier transform infrared spectroscopy, wide angle X-ray diffraction and elemental analysis. Dibasic ammonium phosphate (DAP) lowers the dehydration temperature of rayon evidently and acts also as a reactant besides as a catalyst. Group C=O is generated at 200 °C while group C=C appears at 250 °C. About 220–250 °C is a key temperature range where the chain, ring and crystal structures of rayon macromolecules are damaged completely to lose the basic characteristics of cellulose by dehydration. The drastic changes of weight loss, discoloration and shrinkage occur simultaneously. The bands of chemical groups decrease rapidly and go to disappear during carbonization. The activation at a lower temperature (750 °C) decreases the contents of oxygen-containing groups, but increases them at a higher temperature (950 °C). The microcrystalline structures are gradually formed near 800 °C and activation seems to be favor to the crystallization. A higher usage of the phosphate does not only affect the weight-losses of rayon fiber significantly, it also enhances the specific surface areas of ACF obviously.







Similar content being viewed by others
References
Arthur JC (1983) Cellulosic chemistry and technology (Chinese). Light Industry Press, Beijing, pp 135–149
Basch A, Lewin M (1975) The influence of structure on the flame retardance of cellulose. Text Res J 45(3):246–255
Blanchard EJ, Graves EE (2003) Phosphorylation cellulose with some phosphonic acid derivatives. Text Res J 73(1):22–26
Donnet JB, Bansal RC (1990) Carbon fibers, 2nd edn. Marcel Dekker Inc., New York, pp 99–138
Gottlieb IM (1956) A theory of flame-retardant finishes. Text Res J 26(1):156–167
Hendrix JE, Drake GL (1972) Pyrolysis and combustion of cellulose. III. Mechanistic basis for the synergism involving organic phosphates and nitrogenous bases. J Appl Polym Sci 16(2):257–274
Kilzer FJ, Broido A (1965) Speculations on the nature of cellulose pyrolysis. Pyrodynamics 2:151–163
Lewin M, Sello SB (1983) Handbook of fiber science and technology: Vol. II, Chemical processing of fiber and fabrics, functional finishes: Part B. Marcel Dekker, Inc., New York, pp 2–93
Morterra C, Low MJD (1983) IR studied carbon—II. The vaccum pyrolysis of cellulose. Carbon 21(3):283–288
Pastor AC, Rodriguez-Reinoso F, Marsh H, Martinez MA (1999) Preparation of activated carbon cloth from viscose rayon. Part I. Carbonization procedures. Carbon 37(8):1275–1283
Perkins RM, Drake GL, Reeves WA (1966) DTA and TGA studies of flame-resistant fabrics. J Appl Polymer Sci 10(7):1041–1066
Schwenker RF, Beck LR (1963) Study of pyrolytic decomposition of cellulose by gas chromatography. J Polym Sci Part C (2):331–340
Sekiguchi Y, Shafizadeh F (1984) The effect of inorganic additives on the formation, composition, and combustion of cellulosic char. J Appl Polymer Sci 29:1267–1286
Tang M.M., Bacon R (1964) Carbonization of cellulose fibers—I, low temperature pyrolysis. Carbon 2(3):211–220
Wodley FA (1971) Pyrolysis products of untreated and flame retardant-treated α-cellulose and levoglucosan. J Appl Polym Sci 15:835–851
Wu Y (2000) Plant chemistry (Chinese), 2nd version. Light Industry Press of China, Peking, pp 174–177
Zeng FL, Pan D, Pan N (2005) Choosing the impregnants for rayon-based activated carbon fiber by thermogravimetric analysis. J Inorg and Organomet Polym and Mat 15(2):261–267
Acknowledgment
This paper received the financial aid from Natural Scientific Foundation of Shanghai, China (05ZR14003).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zeng, F., Pan, D. The structural transitions of rayon under the promotion of a phosphate in the preparation of ACF. Cellulose 15, 91–99 (2008). https://doi.org/10.1007/s10570-007-9148-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-007-9148-6