Abstract
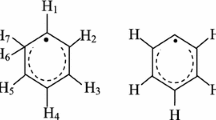
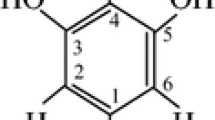
By use of 9.7 GHz and 94 GHz ESR spectra and electron spin echo (ESE)-detected spectra the six radical centres produced by γ-irradiation of cellobiose were identified. The radicals are localized on different carbon atoms. Use of high-frequency ESR spectra with computer resolution enhancement methods enabled unique radical identification and determination of g-factors and proton hyperfine splitting, A, with high accuracy. For radiation doses below 20 kGy three radicals dominate: on C1 with isotropic doublet A = 1.8 mT; on C2, C3 and C4 with triplet A = 2.9 mT; and localized on CH2 with anisotropic triplet. For doses above 100 kGy the radical on C1 dominates, because of cleavage of the glycosidic bonds. Electron spin–lattice relaxation shows that radiation damage of the cellulose structure around the radical centres is significant and radical molecules do not participate in phonon dynamics of the host lattice. The relaxation is because of tunnelling motions of the ring or OH-groups, with tunnelling splitting 2.4 cm−1. Electron spin echo dephasing results identify cellobiose ring torsions with activation energy 117 cm−1.
Similar content being viewed by others
References
Atalla R.H. (1999) In: Pinto B.N. (ed) Comprehensive natural product chemistry, vol 3. Carbohydrates and their derivatives including tannin, cellulose, and related lignins. Elsevier, Amsterdam, pp 529–598
Bowman M.K., Kevan L. (1977). An electron spin–lattice relaxation mechanism involving tunneling modes for trapped electrons in gamma-irradiated ethanol glasses. J. Phys. Chem. 81:456–461
Charlesby A. (1982). Crosslinking and degradation of polymers. Radiat. Phys. Chem. 18:59–66
Chu S.S.C., Jeffrey G.A. (1968). The refinement of the crystal structure of β-D-glucose and cellobiose. Acta Crystallogr. B 24:830–838
Delincee H., Soika Ch. (2002). Improvement of the ESR detection of irradiated food containing cellulose employing a simple extraction method. Radiat. Phys. Chem. 63:437–441
Engalytcheff A., Kolberg M., Barra A.L., Andersson K.K., Tilquin B. (2004). The use of multi-frequency EPR technique to identify the radicals produced in irradiated β-blockers. Free Radical Res 38:59–66
Ershov B.G., Isakova O.V. (1984). Formation and thermal transformations of free radicals in gamma-radiation cellulose. Bull. Acad. Sci. USSR. 33:1171–1175
Ershov B.G., Klimentov A.S., Bykov L.E. (1977). EPR spectra of radicals in γ-irradiated wood and cellulose. Khimiya Drevesiny (Russ.) 2:74–78
Ershov B.G. (1998). Radiation-chemical degradation of cellulose and other polysaccharides. Russian Chem. Rev. 67:315–334
Franco R.W.A., Martin-Neto L., Kato M.S.A., Furlan G.R., Walder J.M.M., Colagno L.A. (2004). Identification of irradiation treatment in black pepper by electron paramagnetic resonance. Int. J. Food Sci. Tech. 39:395–401
Gamble W.L., Miyagawa I., Hartman R.L. (1968). Some Effects of Quantization of Internal Rotation on Spin–lattice Relaxation and Hyperfine Structure. Phys. Rev. Lett. 20:415–418
Goslar J., Hoffmann S.K., Hilczer W. (2002). Local vibration mode mechanism of electron spin–lattice relaxation of PO 2−3 radicals in γ-irradiated (glycine) H3PO3 crystal and in its deuterated analogue. Sol. State Comm. 121:423–427
Hoffmann S.K., Goslar J., Hilczer W., Augustyniak-Jablokow M.A., Kiczka S. (2001). Dephasing relaxation of the electron spin echo of the vibronic Cu(H2O)6 complexes in Tutton salt crystals at low temperatures. J. Magn. Res. 153:56–68
Hoffmann S.K., Hilczer W., Radczyk T. (2003). Electron spin–lattice relaxation in polymers and crystals related to disorder and structure defects. Acta Phys. Pol. 103:373–385
Hoffmann S.K., Hilczer W., Goslar J., Kiczka S., Polus I. (2002). Resonance-type effects in free radical electron spin–lattice relaxation and electron spin echo dephasing due to a dynamics of a homogeneous-chain oligomeric system. Phys. Chem. Chem. Phys. 4:4944–4951
Hon D.N., Srinivasan K.S.V. (1983). Mechanochemical processes in cotton cellulose fiber. J. Appl. Polymer Sci. 28:1–10
Hon N.S. (1975). Formation of free radicals in photoirradiated cellulose. VI. Effect of lignin. J. Polymer Sci. Polym. Chem. 13:2653–2669
Jacobson R.A., Wunderlich J.A., Lipscomb W.N. (1961). The crystal and molecular structure of cellobiose. Acta Crys. B 14:598–607
Jesus E.F.O., Rossi A.M., Lopes R.T. (1999). An ESR study on identification of gamma-irradiated kiwi, papaya and tomato using fruit pulp. Int. J. Food Sci. Technol. 34:173–178
Kawano Y., Logarezzi A. (1995). X-ray induced degradation of regenerated cellulose membrane films. J. M. Polymer Degr. Stabil. 50:125–130
Kuzuya M., Yamauchi Y. (1998). Plasma-induced free radicals of polycrystalline dicarbohydrates studied by electron spin resonance. Thin Solid Films. 316:158–164
Kubota H., Ogiwara Y., Matsuzaki K. (1975). Photo-induced radicals in glucose and cellobiose. J. Appl. Polymer Sci. 19:1291–1296
Kuzina S.I., Brezgunov A.Yu., Dubinskii A.A., Mikhailov A.I. (2004). Free radicals in the photolysis of polymers: IV. Radicals in γ- and UV-irradiated wood and lignin. High Energy Chem. 38:298–305
Kuzuya M., Yamauchi Y., Kondo (1999). Mechanolysis of glucose-based polysaccharides as studied by electron spin resonance. S. J. Phys. Chem. B 103:8051–8059
Mazeau K., Heux L. (2003). Molecular dynamics simulations of bulk native crystalline and amorphous structures of cellulose. J. Phys. Chem. B 107:2394–2403
Meissner D., Einfeldt L., Einfeldt (2001). Dielectric relaxation analysis of cellulose oligomers and polymers in dependency on their chain length. J. J. Polymer Sci. B. Polymer Phys. 39:2491–2500
Misra S.K. (1998). Spin–lattice relaxation time in amorphous materials as affected by exchange interaction and tunneling. Spectrochim. Acta A 54:2257–2267
Mohanty A.K., Singh B.C. (1988). Radiation-induced and photoinduced grafting onto cellulose and cellulosic materials. Polym. Plast. Techn. Eng. 27:435–466
Moncrief J.W. and Sims S.O. 1969. Absolute configuration determination using the anomalous scattering of Cu-Kα X-rays by oxygen atoms: Cellobiose. J. Chem. Soc. D 914–915
Murphy J. (1966). Spin–lattice Relaxation Due to Local Vibrations with Temperature-Independent Amplitudes. Phys. Rev. 145:241–247
Nakamura Y., Ogiwara Y., Phillips G.O. (1985). Free-radical formation and degradation of cellulose by ionizing-radiation. Polym. Photochem. 6:135–159
Ranby B., Rabek J.F. (1977). In ESR Spectroscopy in Polymer Research. Springer-Verlag, Berlin
Tang H.R., Belton P. (2002). Solid-state NMR studies of cellobiose motions. S. Sol. State Nucl. Magn. Res. 21:117–133
Wach R.A., Mitomo H., Yoshii F.J. (2004). ESR investigation of gamma-irradiated methylcellulose and hydroxyethylcellulose in dry state and in aqueous solution. Radioanal. Nucl. Chem. 261:113–118
Wach R.A., Mitomo H., Nagasawa N., Yoshii F. (2003). Radiation crosslinking of carboxymethylcellulose of various degree of substitution at high concentration in aqueous solution of natural pH. Rad. Phys. Chem. 68:771–779
Yamauchi Y., Sugito M., Kuzuya M. (1999). Plasma-induced free radicals of polycrystalline monocarbohydrates studied by Electron Spin Resonance. Chem. Pharm. Bull. 47:273–278
Acknowledgements
The authors thanks Professor Gunnar Jeschke, in whose laboratory at Max-Planck Institute for Polymer Research in Mainz the W-band ESR measurements were carried out, for valuable suggestions and comments, and Dr Yevhen Polyhach for technical assistance during the measurements.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wencka, M., Wichlacz, K., Kasprzyk, H. et al. Free radicals and their electron spin relaxation in cellobiose. X-band and W-band ESR and electron spin echo studies. Cellulose 14, 183–194 (2007). https://doi.org/10.1007/s10570-006-9097-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-006-9097-5