Abstract
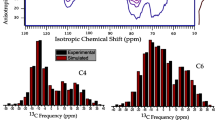
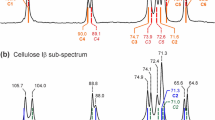
In our previous studies of the crystal structure of native cellulose (cellulose I) by solid-state two-dimensional (2D) 13C–13C INADEQUATE, it was revealed that cellulose Iα contains two kinds of β-d-glucose residues (A and A′) in the unit cell and that cellulose Iβ contains another two kinds of residues (B and B′). In the present study, the sequence of residues A and A′ along the same chains in cellulose Iα and that of residues B and B′ in Iβ were investigated by 2D 13C–13C rotor-synchronized radiofrequency-driven recoupling (RFDR) experiments using, respectively, uniformly 13C6-labeled (U−13C6) bacterial cellulose and the same [U−13C6] cellulose sample after thermal treatment at 260 °C. The RFDR spectra recorded with a short mixing time (1.0 ms) showed dipolar-coupled 13C spin pairs of only the neighboring carbon of the both phases, while those recorded with a longer mixing time (3.0–15 ms) provided correlations between weakly coupled 13C spin pairs as well as strongly coupled 13C spin pairs such as neighboring carbon nuclei. In the RFDR spectrum of the [U−13C6] cellulose recorded with a mixing time of 15 ms, the inter-residue 13C–13C correlation between C4 of residue A and C2 of residue A′ and that between C3 of residue A and C4 of residue A′ were clearly observed. In the case of cellulose Iβ, however, inter-residue 13C–13C correlations between residues B and B′ could not be detected in the series of RFDR spectra recorded with different mixing times of annealed [U−13C6] cellulose. From these findings, that cellulose Iα was revealed to have the –A–A′– repeating units along the cellulose chain. For cellulose Iβ, it was revealed that the respective residues B and B′ are composed of independent chains (–B–B– and –B′–B′– repeating units) and that there are no –B–B′– repeating units in the chain.
Similar content being viewed by others
References
Arashida T., Ishino T., Kai A., Hatanaka K., Araike T., Matsuzaki K., Kaneko Y. and Miura T. (1993). Biosynthesis of cellulose from culture media containing 13C-labeled glucose as a carbon source. J. Carbohydr. Chem. 12:641–649
Atalla R.H. and VanderHart D.L. (1984). Native cellulose: a composite of two distinct crystalline forms. Science 223:283–285
Aue W.P., Bartholdi E. and Ernst R.R. (1976). Two-dimensional spectroscopy. Application to nuclear magnetic resonance. J. Chem. Phys. 64:2229–2246
Bax A., Freeman R. and Frenkiel T.A. (1981). An NMR technique for tracing out the carbon skeleton of an organic molecule. J. Am. Chem. Soc. 103:2102–2104
Bennett A.E., Ok J.H., Griffin R.G. and Vega S. (1992). Chemical shift correlation spectroscopy in rotating solids: radio frequency-driven dipolar recoupling and longitudinal exchange. J. Chem. Phys. 96:8624–8627
Bennett A.E., Rienstra C.M., Auger M., Lakshmi K.V. and Griffin R.G. (1995). Heteronuclear decoupling in rotating solids. J. Chem. Phys. 103:6951–6958
Debzi E.M., Chanzy H., Sugiyama J., Tekely P. and Excoffier G. (1991). The Iα→Iβ transformation of highly crystalline cellulose by annealing in various mediums. Macromolecules 24:6816–6822
Finkenstadt V.L. and Millane R.P. (1998). Crystal structure of Valonia cellulose Iβ. Macromolecules 31:3776–3778
Gardner K.H. and Blackwell J. (1974). The structure of native cellulose. Biopolymers 13:1975–2001
Horii F., Yamamoto H. and Kitamaru R. (1987). Transformation of native cellulose crystals induced by saturated stream at high temperatures. Macromolecules 20:2946–2949
Kai A., Arashida T., Hatanaka K., Araike T., Matsuzaki K., Miura T. and Kaneko Y. (1994). Analysis of the biosynthetic process of cellulose and curdlan using 13C-labeled glucose. Carbohydr. Polym. 23:235–239
Kono H., Yunoki S., Shikano T., Fujiwara M., Erata T. and Takai M. (2002). CP/MAS 13C NMR study of cellulose and cellulose derivatives. 1. Complete assignment of the CP/MAS 13C NMR spectrum of the native cellulose. J. Am. Chem. Soc. 124:7506–7511
Kono H., Erata T. and Takai M. (2003a). Complete assignment of the CP/MAS 13C NMR spectrum of the cellulose IIII. Macromolecules 36:3589–3592
Kono H., Erata T. and Takai M. (2003b). Determination of through-bond carbon-carbon and carbon-proton connectivities of the native celluloses in the solid state. Macromolecules 36:5131–5138
Kono H., Numata Y., Erata T. and Takai M. (2004). 13C and 1H Resonance assignment of mercerized cellulose II by two-dimensional MAS NMR spectroscopies. Macromolecules 37:5310–5316
Kono H. and Numata Y. (2004). Two-dimensional spin-exchange solid-state NMR studies of 13C-enriched cellulose II. Polymer 45:2843–2852
Lesage A., Sakellariou D., Steuernagel S. and Emsley L. (1998). Carbon-proton chemical shift correlation in solid-state NMR by through-bond multiple-quantum spectroscopy. J. Am. Chem. Soc. 120:13194–13201
Lesage A., Bardet M. and Emsley L. (1999). Through-bond carbon-carbon connectivities in disordered solids by NMR. J. Am. Chem. Soc. 121:10987–10993
Marion D. and Wüthrich K. (1983). Application of phase sensitive two-dimensional correlated spectroscopy (COSY) for measurements of 1H−1H spin–spin coupling constants in proteins. Biochem. Biophys. Res. Commun. 113:967–974
Metz G., Wu X. and Smith S.O. (1994). Ramped-amplitude cross polarization in magic-angle-spinning NMR. J. Magn. Reson. A 110:219–227
Mueller L. (1979). Sensitivity enhanced detection of weak nuclei using heteronuclear multiple quantum coherence. J. Am. Chem. Soc. 101:4481–4484
Nishiyama Y., Langan P. and Chanzy H. (2002). Crystal structure and hydrogen-bonding system in cellulose Iβ from synchrotron X-ray and neutron fiber diffraction. J. Am. Chem. Soc. 124:9074–9082
Nishiyama Y., Sugiyama J., Chanzy H. and Langan P. (2003). Crystal structure and hydrogen bonding system in cellulose Iα from synchrotron X-ray and neutron fiber diffraction. J. Am. Chem. Soc. 125:14300–14306
Numata Y., Kono H., Kawano S., Erata T. and Takai M. (2003). Cross-polarization/magic-angle spinning 13C nuclear magnetic resonance study of cellulose I–ethylenediamine complex. J. Biosci. Bioeng. 96:461–466
Sarko A. and Muggli R. (1974). Packing analysis of carbohydrates and polysaccharides III. Valonia cellulose and cellulose II. Macromolecules 7:486–494
Shramm M. and Hestrin S. (1954). Factors affecting production of cellulose at the air/liquid interface of a culture of Acetobacter xylinum. J. Gen. Microbiol. 11:123–129
Sugiyama J., Okano T., Yamamoto H. and Horii F. (1990). Transformation of Valonia cellulose crystals by an alkaline hydrothermal treatment. Macromolecules 23:3196–3198
Sugiyama J., Persson J. and Chanzy H. (1991a). Combined infrared and electron diffraction study of the polymorphism of native celluloses. Macromolecules 24:2461–2466
Sugiyama J., Vuong R. and Chanzy H. (1991b). Electron diffraction study on the two crystalline phases occurring in native cellulose from an algal cell wall. Macromolecules 24:4168–4175
VanderHart D.L. and Atalla R.H. (1984). Studies of microstructure in native celluloses using solid-state 13C NMR. Macromolecules 17:1465–1472
Yamamoto H. and Horii F. (1993). CP/MAS 13C NMR analysis of the crystal transformation induced Valonia cellulose by annealing at high temperatures. Macromolecules 26:1313–1317
Yunoki S., Osada Y., Kono H. and Takai M. (2004). Role of ethanol in improvement of bacterial cellulose production: analysis using 13C-labeled carbon sources. Food Sci. Technol. Res. 10:307–313
Zugenmaier P. (2001). Conformation and packing of various crystalline cellulose fibers. Prog. Polym. Sci. 26:1341–1417
Acknowledgements
We wish to thank Prof. Dr. T. Erata and Prof. Dr. M. Takai (Hokkaido University) for preparing cellulose samples. In addition, H. Kono thanks Prof. Dr. F. Horii (Kyoto University) for helpful discussions and carefully checking the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kono, H., Numata, Y. Structural investigation of cellulose Iα and Iβ by 2D RFDR NMR spectroscopy: determination of sequence of magnetically inequivalent d-glucose units along cellulose chain. Cellulose 13, 317–326 (2006). https://doi.org/10.1007/s10570-005-9025-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-005-9025-0