Abstract
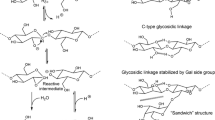
A novel synthetic method for co-oligomers of tri-O-methylated and unmodified cello-oligosaccharides was designed. These oligomers are of importance as model compounds for investigations on the dissolution behavior of commercial methylcelluloses. In this connection, insights into the chemical structure of ‘cross linking loci’ in the thermo reversible gelation of aqueous solution of methylcellulose are of particular significance. The synthetic procedure consists of glycosylation using glycosyl fluoride and oligomerization of sugar orthoester. Thus, phenyl 2,3,6-tri-O-methyl-β-d-glucopyranosyl-(1→4)-2,3,6-tri-O-methyl-1-thio-β-d-glucopyranoside (1) as a glycosyl acceptor was glycosylated with 4-O-acetyl-2,3,6-tri-O-methyl-β-d-glucopyranosyl-(1→4)-2,3,6-tri-O-methyl-d-glucopyranosyl fluoride (2) as a glycosyl donor converted to give a cellotetraose derivative (3). Both reactants have been prepared from commercially available cellobiose. After deacetylation of 3, 3-O-benzyl-6-O-pivaloyl-α-d-glucopyranose 1,2,4-orthopivalate (5) was reacted with 4-hydroxyl group at non-reducing-end of cellotetraose derivative (4) to give the block co-oligomer (6). After the deprotection of compound 6, tri-O-methylated-block-unmodified cello-oligosaccharides (18 and 18′) (DP = 4 − 8, DS = 2.79 − 1.38) were obtained, monitored by MALDI-TOF MS spectra. Chloroform-soluble methylated cellotetraose derivatives (18 and 18′ (DP=4, n=0), DS=2.57, and 2.79, respectively) were also soluble in the water solution of tri-O-methylated-block-unmodified cello-oligosaccharides (DP=4−8, DS=2.79−1.50). This fact indicated that hydrophobic methylated cello-tetraose derivatives were encapsulated within a micelle of amphiphlic tri-O-methylated-block-unmodified cello-oligosaccharides. It was found that solubilities of 18 and 18′ (DP=4−8, DS=2.79−1.38) in water and chloroform were obviously different in the mixtures, depending on their DP and DS values. The substituent distribution of the tri-O-methylated-block-unmodified cello-oligosaccharides along one molecule and between molecules plays an important role in its solubility in water and chloroform.
Similar content being viewed by others
References
Adden R., Bosch A. and Mischnick P. (2004). Novel possibilities by cationic ring-opening polymerisation of cyclodextrin derivatives: Preparation of a copolymer bearing block-like sequences of tri-o-methylglucosyl units. Macromolecular Chemistry and Physics 205(15):2072–2079
Arisz P.W., Kauw H.J.J. and Boon J.J. (1995). Substituent distribution along the cellulose backbone in o-methylcelluloses using gc and fab-ms for monomer and oligomer analysis. Carbohydrate Research 271:1–14
Bahrke S., Einarsson J.M., Gislason J., Haebel S., Letzel M.C., Peter-Katalinic J. and Peter M.G. (2002). Sequence analysis of chitooligosaccharides by matrix-assisted laser desorption ionization postsource decay mass spectrometry. Biomacromolecules 3(4):696–704
Erler U., Mischnick P., Stein A. and Klemm D. (1992). Determination of the substitution patterns of cellulose methyl ethers by hplc and glc - comparison of methods. Polymer Bulletin 29(3–4):349–356
Freudenberg K. and Blomqvist G. (1935). Hydrolyse der cellulose und ihrer oligosaccharide. Berichite der Deutschen Chemischen Gesellschaft zu Berlin 68:2066–2082
Freudenberg K., Friedlich K. and Bumann I. (1932). Über cellulose und stärke. Justus Liebigs Annalen der Chemie 494:41–62
Fukuyama T., Laird A.A. and Hotchkiss L.M. (1985). Para-anisyl group - a versatile protecting group for primary alcohols. Tetrahedron Letters 26(51):6291–6292
Hashimoto S., Hayashi M. and Noyori R. (1984). Glycosylation using glucopyranosyl fluorides and silicon-based catalysts - solvent dependency of the stereoselection. Tetrahedron Lett. 25(13):1379–1382
Heinrich J. and Mischnick P. (1996). Rapid method for the determination of the substitution pattern of o-methylated 1,4-glucans by high-ph anion-exchange chromatography with pulsed amperometric detection. Journal of Chromatography A 749(1–2):41–45
Hernandez-Torres J.M., Liew S.T., Achkar J. and Wei A. 2002. Optimized synthesis of an orthogonally protected glucosamine. Synthesis-Stuttgart (4): 487–490
Heymann E. (1935). Studies on sol-gel transitions. 1. The inverse sol-gel transformation of methylcellulose in water. Trans. Faraday Soc. 31:846
Hiranuma S., Kanie O. and Wong C.H. (1999). Formation of the 1,2,6-orthoester of mannose and its utilization in the glycosylation reaction. Tetrahedron Lett. 40(35):6423–6426
Kamitakahara H. and Nakatsubo F. (1996). Substituent effect on ring-opening polymerization of regioselectively acylated 1,4-anhydro-alpha-d-glucopyranose derivatives. Macromolecules 29(4):1119–1122
Kamitakahara H., Nakatsubo F. and Murakami K. (1994). Syntheses of 1,4-anhydro-alpha-d-glucopyranose derivatives having acyl-groups. Mokuzai Gakkaishi 40(3):302–307
Kamitakahara H., Hori M. and Nakatsubo F. (1996). Substituent effect on ring-opening polymerization of regioselectively acylated alpha-d-glucopyranose 1,2,4- orthopivalate derivatives. Macromolecules 29(19):6126–6131
Kanie O., Ito Y. and Ogawa T. (1994). Orthogonal glycosylation strategy in oligosaccharide synthesis. J. Am. Chem. Soc. 116(26):12073–12074
Karakawa M., Kamitakahara H., Takano T. and Nakatsubo F. (2002). The utility of a 3-o-allyl group as a protective group for ring-opening polymerization of alpha-d-glucopyranose 1,2,4- orthopivalate derivatives. Biomacromolecules 3(3):538–546
Karakawa M., Mikawa Y., Kamitakahara H. and Nakatsubo F. (2002). Preparations of regioselectively methylated cellulose acetates and their h-1 and c-13 nmr spectroscopic analyses. J. Polym. Sci. Pol. Chem. 40(23):4167–4179
Kato T., Yokoyama M. and Takahashi A. (1978). Melting temperatures of thermally reversible gels. Colloid and Polymer Science 256:15–21
Kato T., Kozaki N. and Takahashi A. (1986). Lubrication enhancement by surface gelation of thin liquid-films of methylcellulose and hydroxypropyl methylcellulose solutions. Polymer Journal 18(2):189–191
Kawada T., Nakatsubo F. and Murakami K. (1989). Synthetic studies of cellulose.4. Preparation of perbenzylated allyl 4′-o-p-methoxybenzyl cellobioside by the imidate method. Mokuzai Gakkaishi 35(1):21–25
Kern H., Choi S., Wenz G., Heinrich J., Ehrhardt L., Mischnick P., Garidel P. and Blume A. (2000). Synthesis, control of substitution pattern and phase transitions of 2,3-di-o-methylcellulose. Carbohydrate Research 326(1):67–79
Konradsson P., Udodong U.E. and Fraserreid B. (1990). Iodonium promoted reactions of disarmed thioglycosides. Tetrahedron Lett. 31(30):4313–4316
Kuhn W., Moser, P., Majer, H. (1961). Aggregation von methylcellulose in lösung. Helv. Chim. Acta 44:770
Malet C., Viladot J.L., Ochoa a., Gallego B., Brosa C. and Planas a. (1995). Synthesis of 4-methylumbelliferyl-beta-d-glucan oligosaccharides as specific chromophoric substrates of (1-]3),(1-]4)-beta-d-glucan 4-glucanohydrolases. Carbohydr. Res. 274:285–301
Matsumoto T., Maeta H., Suzuki K. and Tsuchihashi G. (1988). New glycosidation reaction.1. Combinational use of cp2zrcl2-agclo4 for activation of glycosyl fluorides and application to highly beta-selective glycosidation of d-mycinose. Tetrahedron Lett. 29(29):3567–3570
Mischnick P. (2001). Challenges in structure analysis of polysaccharide derivatives. Cellulose 8(4):245–257
Momcilovic D., Wittgren B., Wahlund K.G., Karlsson J. and Brinkmalm G. (2003). Sample preparation effects in matrix-assisted laser desorption/ionisation time-of-flight mass spectrometry of partially depolymerised methyl cellulose. Rapid Communications in Mass Spectrometry 17(11):1116–1124
Motawia M.S., Olsen C.E., Denyer K., Smith A.M. and Moller B.L. (2001). Synthesis of 4 ′-o-acetyl-maltose and alpha-d-galactopyranosyl-(1->4)-d-glucopyranose for biochemical studies of amylose biosynthesis. Carbohydr. Res. 330(3):309–318
Mukaiyama T., Hashimoto S. and Shoda S. 1981. An efficient method for glucosylation of hydroxy compounds using glucopyranosyl fluoride. Chem. Lett.: 431–432
Nakatsubo F., Kamitakahara H. and Hori M. (1996). Cationic ring-opening polymerization of 3,6-di-o-benzyl-alpha- d-glucose 1,2,4-orthopivalate and the first chemical synthesis of cellulose. J. Am. Chem. Soc. 118(7):1677–1681
Nicolaou K.C., Seitz S.P. and Pavia M.R. (1982). Carbohydrates in organic-synthesis - synthesis of 16-membered-ring macrolide antibiotics.6. Total synthesis of o-mycinosyltylonolide - coupling of key intermediates and macrocyclization. J. Am. Chem. Soc. 104(7):2030–2031
Nicolaou K.C., Randall J.L. and Furst G.T. (1985). Stereospecific synthesis of rhynchosporosides - a family of fungal metabolites causing scald disease in barley and other grasses. J. Am. Chem. Soc. 107(19):5556–5558
Nishimura T. and Nakatsubo F. (1996). First synthesis of cellooctaose by a convergent synthetic method. Carbohydr. Res. 294:53–64
Pringsheim H. and Leibowitz J. (1925). Über die beziehunh zwischen optischem drehungsvermögen und structur in der polysaccharid-chemie. Berichite der Deutschen Chemischen Gesellschaft zu Berlin 58:2808–2814
Rees D.A. (1972). Polysaccharide gels, a molecular view. Chem. & Ind. (London) 19:630–636
Richardson S. and Gorton L. (2003). Characterisation of the substituent distribution in starch and cellulose derivatives. Analytica Chimica Acta 497(1–2):27–65
Savage A.B. (1957). Temperature-viscosity relationships for water-soluble cellulose ethers. Ind. Eng. Chem. 49:99
Suzuki K., Maeta H. and Matsumoto T. (1989). An improved procedure for metallocene-promoted glycosidation - enhanced reactivity by employing 1–2-ratio of cp2hfcl2-agclo4. Tetrahedron Lett. 30(36):4853–4856
Veeneman G.H., Vanleeuwen S.H. and Vanboom J.H. (1990). Iodonium ion promoted reactions at the anomeric center.2. An efficient thioglycoside mediated approach toward the formation of 1,2-trans linked glycosides and glycosidic esters. Tetrahedron Lett. 31(9):1331–1334
Wagner G. and Metzner R. (1969). Synthesis of aromatic disaccharide thioglycosides and disaccharide sulfonylglycosides. Pharmazie 24(5):245–50
Yamaura M., Suzuki T., Hashimoto H., Yoshimura J., Okamoto T. and Shin C. (1985). Oxidative removal of n-(4-methoxybenzyl) group on 2,5-piperazinediones with cerium(iv) diammonium nitrate. Bulletin of the Chemical Society of Japan 58(5):1413–1420
Acknowledgements
H. Kamitakahara acknowledges the Alexander von Humboldt Foundation for a research fellowship at University of Jena. The authors thank Dr. W. Poppitz and his co-workers, Institute for Inorganic and Analytical Chemistry, University of Jena, for measuring ESI mass spectra. The authors thank Dr. W. Günther and his co-workers, Institute for Organic Chemistry and Macromolecular Chemistry, University of Jena, for measuring a part of NMR spectra. This investigation was supported in part by a Grant-in-Aid for Scientific Research from the Ministry of Education, Science, and Culture of Japan (Nos. 13760132 and 15780124).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kamitakahara, H., Nakatsubo, F. & Klemm, D. Block co-oligomers of tri-O-methylated and unmodified cello-oligosaccharides as model compounds for methylcellulose and its dissolution/gelation behavior. Cellulose 13, 375–392 (2006). https://doi.org/10.1007/s10570-005-9003-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-005-9003-6