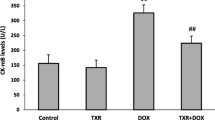
Abstract
The role of nitric oxide (NO) in doxorubicin (DOX; cancer chemotherapeutic)-induced cardiotoxicity is well established. In skeletal muscle (SM), NO regulation plays a critical role in health, biogenesis, and function. Despite the increasing evidence that indicates the negative impact of DOX on SM function, the effect of DOX on NO production in SM has yet to be examined. The purpose of the current study was to simultaneously examine intracellular and interstitial NO concentrations in the SM following the administration of DOX. A single dose of 1.5 or 4.5 mg/kg was administered intraperitoneally to male Sprague Dawley rats, and interstitial (IS) and intracellular (IC) NO was quantified every 24 up to 192 h post-injection. There was no significant difference in IC NO following the injection of 1.5 mg/kg DOX when compared to the control; however, the administration of 4.5 mg/kg DOX resulted in lower (P < 0.05) concentrations of NO in the IC. Interestingly, a consistently higher (P < 0.05) concentration of NO in the IS was established following the administration of 1.5 mg/kg compared to the control while no significant changes in IS NO resulted from the administration of the 4.5 mg/kg dose. The fluctuation of IS and IC NO was not a result of substrate availability as arginine concentrations remained stable throughout the experiment. By utilizing the microdialysis technique, we have simultaneously quantified for the first time the IS and IC concentrations of NO in SM following DOX administration. These data provide important insight in the possible mechanisms leading to DOX-related SM dysfunction.





Similar content being viewed by others
Abbreviations
- AKR:
-
Aldo-keto reductase
- Arg:
-
L-arginine
- CBR:
-
Carbonyl reductase
- DOX:
-
Doxorubicin
- eNOS:
-
Endothelial nitric oxide synthase
- iNOS:
-
Inducible nitric oxide synthase
- Jun-D:
-
Transcription factor Jun-D
- MCK:
-
Muscle creatine kinase
- MyoD:
-
Myoblast determination protein
- NF-kB:
-
Nuclear factor activated B cells
- NO:
-
Nitric oxide
- nNOS:
-
Neuronal nitric oxide synthase
- NOS:
-
Nitric oxide synthase
- O2 − :
-
Superoxide
- ONOO− :
-
Peroxynitrite
- PL:
-
Plantaris muscle
- ROS:
-
Radical oxygen species
- SOL:
-
Soleus muscle
- TOP2A:
-
Topoisomerase II
- WG:
-
Medial gastrocnemius
References
Aldieri E, Bergandi L, Riganti C, Costamagna C, Bosia A, Ghigo D. Doxorubicin induces an increase of nitric oxide synthesis in rat cardiac cells that is inhibited by iron supplementation. Toxicol Appl Pharmacol. 2002;185:85–90.
Antoun S, Baracos VE, Birdsell L, Escudier B, Sawyer MB. Low body mass index and sarcopenia associated with dose-limiting toxicity of sorafenib in patients with renal cell carcinoma. Ann Oncol. 2010;21:1594–8.
Archer SL, Huang JM, Hampl V, Nelson DP, Shultz PJ, Weir EK. Nitric oxide and cGMP cause vasorelaxation by activation of a charybdotoxin-sensitive K channel by cGMP-dependent protein kinase. Proc Natl Acad Sci U S A. 1994;91:7583–7.
Arola OJ, Saraste A, Pulkki K, Kallajoki M, Parvinen M, Voipio-Pulkki LM. Acute doxorubicin cardiotoxicity involves cardiomyocyte apoptosis. Cancer Res. 2000;60:1789–92.
Beckman JS, Beckman TW, Chen J, Marshall PA, Freeman BA. Apparent hydroxyl radical production by peroxynitrite: implications for endothelial injury from nitric oxide and superoxide. Proc Natl Acad Sci U S A. 1990;87:1620–4.
Buck M, Chojkier M. Muscle wasting and dedifferentiation induced by oxidative stress in a murine model of cachexia is prevented by inhibitors of nitric oxide synthesis and antioxidants. EMBO J. 1996;15:1753–65.
Calabrese V, Mancuso C, Calvani M, Rizzarelli E, Butterfield DA, Stella AM. Nitric oxide in the central nervous system: neuroprotection versus neurotoxicity. Nat Rev Neurosci. 2007;8:766–75.
Carter SK. Adriamycin-a review. J Natl Cancer Inst. 1975;55:1265–74.
Conklin KA. Chemotherapy-associated oxidative stress: impact on chemotherapeutic effectiveness. Integr Cancer Ther. 2004;3:294–300.
De Palma C, Morisi F, Pambianco S, Assi E, Touvier T, Russo S, et al. Deficient nitric oxide signalling impairs skeletal muscle growth and performance: involvement of mitochondrial dysregulation. Skelet Muscle. 2014;4:22.
Delgado JM, Defeudis FV, Roth RH, Ryugo DK, Mitruka BM. Dialytrode for long term intracerebral perfusion in awake monkeys. Arch Int Pharmacodyn Ther. 1972;198:9–21.
Di Marco S, Mazroui R, Dallaire P, Chittur S, Tenenbaum SA, Radzioch D, et al. NF-kappa B-mediated MyoD decay during muscle wasting requires nitric oxide synthase mRNA stabilization, HuR protein, and nitric oxide release. Mol Cell Biol. 2005;25:6533–45.
Evig CB, Kelley EE, Weydert CJ, Chu Y, Buettner GR, Burns CP. Endogenous production and exogenous exposure to nitric oxide augment doxorubicin cytotoxicity for breast cancer cells but not cardiac myoblasts. Nitric Oxide. 2004;10:119–29.
Fabris S, MacLean DA. Skeletal muscle an active compartment in the sequestering and metabolism of doxorubicin chemotherapy. PLoS One. 2015;10:e0139070.
Garner AP, Paine MJ, Rodriguez-Crespo I, Chinje EC, Ortiz de Montellano P, Stratford IJ, et al. Nitric oxide synthases catalyze the activation of redox cycling and bioreductive anticancer agents. Cancer Res. 1999;59:1929–34.
Gewirtz DA. A critical evaluation of the mechanisms of action proposed for the antitumor effects of the anthracycline antibiotics adriamycin and daunorubicin. Biochem Pharmacol. 1999;57:727–41.
Gille L, Nohl H. Analyses of the molecular mechanism of adriamycin-induced cardiotoxicity. Free Radic Biol Med. 1997;23:775–82.
Gilliam LA, St Clair DK. Chemotherapy-induced weakness and fatigue in skeletal muscle: the role of oxidative stress. Antioxid Redox Signal. 2011;15:2543–63.
Guerra J, De Jesus A, Santiago-Borrero P, Roman-Franco A, Rodriguez E, Crespo MJ. Plasma nitric oxide levels used as an indicator of doxorubicin-induced cardiotoxicity in rats. Hematol J. 2005;5:584–8.
Hayward R, Hydock D, Gibson N, Greufe S, Bredahl E, Parry T. Tissue retention of doxorubicin and its effects on cardiac, smooth, and skeletal muscle function. J Physiol Biochem. 2013;69:177–87.
Hellsten Y, MacLean D, Radegran G, Saltin B, Bangsbo J. Adenosine concentrations in the interstitium of resting and contracting human skeletal muscle. Circulation. 1998;98:6–8.
Iqbal M, Dubey K, Anwer T, Ashish A, Pillai KK. Protective effects of telmisartan against acute doxorubicin-induced cardiotoxicity in rats. Pharmacol Rep. 2008;60:382–90.
Jackson MJ. Use of microdialysis to study interstitial nitric oxide and other reactive oxygen and nitrogen species in skeletal muscle. Methods Enzymol. 2005;396:514–25.
Jensen RA, Acton EM, Peters JH. Doxorubicin cardiotoxicity in the rat: comparison of electrocardiogram, transmembrane potential, and structural effects. J Cardiovasc Pharmacol. 1984;6:186–200.
Jung ID, Lee JS, Yun SY, Park CG, Han JW, Lee HW, et al. Doxorubicin inhibits the production of nitric oxide by colorectal cancer cells. Arch Pharm Res. 2002;25:691–6.
Kalivendi SV, Kotamraju S, Zhao H, Joseph J, Kalyanaraman B. Doxorubicin-induced apoptosis is associated with increased transcription of endothelial nitric-oxide synthase. Effect of antiapoptotic antioxidants and calcium. J Biol Chem. 2001;276:47266–76.
Kassner N, Huse K, Martin HJ, Godtel-Armbrust U, Metzger A, Meineke I, et al. Carbonyl reductase 1 is a predominant doxorubicin reductase in the human liver. Drug Metab Dispos. 2008;36:2113–20.
Kotamraju S, Konorev EA, Joseph J, Kalyanaraman B. Doxorubicin-induced apoptosis in endothelial cells and cardiomyocytes is ameliorated by nitrone spin traps and ebselen. Role of reactive oxygen and nitrogen species. J Biol Chem. 2000;275:33585–92.
Kremer LC, van Dalen EC, Offringa M, Ottenkamp J, Voute PA. Anthracycline-induced clinical heart failure in a cohort of 607 children: long-term follow-up study. J Clin Oncol. 2001;19:191–6.
Lim S, Shin JY, Jo A, Jyothi KR, Nguyen MN, Choi TG, et al. Carbonyl reductase 1 is an essential regulator of skeletal muscle differentiation and regeneration. Int J Biochem Cell Biol. 2013;45:1784–93.
MacLean DA, Bangsbo J, Saltin B. Muscle interstitial glucose and lactate levels during dynamic exercise in humans determined by microdialysis. J Appl Physiol (1985). 1999;87:1483–90.
MacLean DA, Vickery LM, Sinoway LI. Elevated interstitial adenosine concentrations do not activate the muscle reflex. Am J Physiol Heart Circ Physiol. 2001;280:H546–53.
Megeney LA, Kablar B, Garrett K, Anderson JE, Rudnicki MA. MyoD is required for myogenic stem cell function in adult skeletal muscle. Genes Dev. 1996;10:1173–83.
Minotti G, Menna P, Salvatorelli E, Cairo G, Gianni L. Anthracyclines: molecular advances and pharmacologic developments in antitumor activity and cardiotoxicity. Pharmacol Rev. 2004;56:185–229.
Momparler RL, Karon M, Siegel SE, Avila F. Effect of adriamycin on DNA, RNA, and protein synthesis in cell-free systems and intact cells. Cancer Res. 1976;36:2891–5.
Nagi MN, Mansour MA. Protective effect of thymoquinone against doxorubicin-induced cardiotoxicity in rats: a possible mechanism of protection. Pharmacol Res. 2000;41:283–9.
Octavia Y, Tocchetti CG, Gabrielson KL, Janssens S, Crijns HJ, Moens AL. Doxorubicin-induced cardiomyopathy: from molecular mechanisms to therapeutic strategies. J Mol Cell Cardiol. 2012;52:1213–25.
Okutsu M, Call JA, Lira VA, Zhang M, Donet JA, French BA, et al. Extracellular superoxide dismutase ameliorates skeletal muscle abnormalities, cachexia, and exercise intolerance in mice with congestive heart failure. Circ Heart Fail. 2014;7:519–30.
Olson RD, Mushlin PS. Doxorubicin cardiotoxicity: analysis of prevailing hypotheses. FASEB J. 1990;4:3076–86.
Paulides M, Kremers A, Stohr W, Bielack S, Jurgens H, Treuner J, et al. Prospective longitudinal evaluation of doxorubicin-induced cardiomyopathy in sarcoma patients: a report of the late effects surveillance system (LESS). Pediatr Blood Cancer. 2006;46:489–95.
Prado CM, Baracos VE, Mccargar LJ, Reiman T, Mourtzakis M, Tonkin K, et al. Sarcopenia as a determinant of chemotherapy toxicity and time to tumor progression in metastatic breast cancer patients receiving capecitabine treatment. Clin Cancer Res. 2009;15:2920–6.
Ramamoorthy S, Donohue M, Buck M. Decreased Jun-D and myogenin expression in muscle wasting of human cachexia. Am J Physiol Endocrinol Metab. 2009;297:E392–401.
Riddell DR, Owen JS. Nitric oxide and platelet aggregation. Vitam Horm. 1999;57:25–48.
Riede UN, Forstermann U, Drexler H. Inducible nitric oxide synthase in skeletal muscle of patients with chronic heart failure. J Am Coll Cardiol. 1998;32:964–9.
Sawyer DB, Fukazawa R, Arstall MA, Kelly RA. Daunorubicin-induced apoptosis in rat cardiac myocytes is inhibited by dexrazoxane. Circ Res. 1999;84:257–65.
Smuder AJ, Kavazis AN, Min K, Powers SK. Exercise protects against doxorubicin-induced markers of autophagy signaling in skeletal muscle. J Appl Physiol (1985). 2011;111:1190–8.
Stamler JS, Meissner G. Physiology of nitric oxide in skeletal muscle. Physiol Rev. 2001;81:209–37.
Teraoka K, Hirano M, Yamaguchi K, Yamashina A. Progressive cardiac dysfunction in adriamycin-induced cardiomyopathy rats. Eur J Heart Fail. 2000;2:373–8.
van Norren K, van Helvoort A, Argiles JM, van Tuijl S, Arts K, Gorselink M, et al. Direct effects of doxorubicin on skeletal muscle contribute to fatigue. Br J Cancer. 2009;100:311–4.
Vasquez-Vivar J, Martasek P, Hogg N, Masters BS, Pritchard KA, Kalyanaraman B. Endothelial nitric oxide synthase-dependent superoxide generation from adriamycin. Biochemistry. 1997;36:11293–7.
Wallimann T, Wyss M, Brdiczka D, Nicolay K, Eppenberger HM. Intracellular compartmentation, structure and function of creatine kinase isoenzymes in tissues with high and fluctuating energy demands: the ‘phosphocreatine circuit’ for cellular energy homeostasis. Biochem J. 1992;281(Pt 1):21–40.
Wang S, Konorev EA, Kotamraju S, Joseph J, Kalivendi S, Kalyanaraman B. Doxorubicin induces apoptosis in normal and tumor cells via distinctly different mechanisms. intermediacy of H(2)O(2)- and p53-dependent pathways. J Biol Chem. 2004;279:25535–43.
Xi L, Zhu SG, Das A, Chen Q, Durrant D, Hobbs DC, et al. Dietary inorganic nitrate alleviates doxorubicin cardiotoxicity: mechanisms and implications. Nitric Oxide. 2012;26:274–84.
Yu AP, Pei XM, Sin TK, Yip SP, Yung BY, Chan LW, et al. Acylated and unacylated ghrelin inhibit doxorubicin-induced apoptosis in skeletal muscle. Acta Physiol (Oxf). 2014;211:201–13.
Zhang S, Liu X, Bawa-Khalfe T, Lu LS, Lyu YL, Liu LF, et al. Identification of the molecular basis of doxorubicin-induced cardiotoxicity. Nat Med. 2012;18:1639–42.
Acknowledgments
The authors wish to thank Ms. Pamela Chenard, Ms. Talia Ryan, and Ms. Josée Lalanne for their excellent technical assistance. This study was supported by the Natural Sciences and Engineering Research Council and the Ontario Institute for Cancer Research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fabris, S., MacLean, D.A. Doxorubicin chemotherapy affects intracellular and interstitial nitric oxide concentrations in skeletal muscle. Cell Biol Toxicol 32, 121–131 (2016). https://doi.org/10.1007/s10565-016-9325-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10565-016-9325-1