Abstract
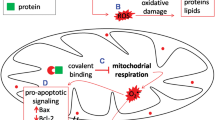
Drug-induced liver injury (DILI) is a leading cause of discontinuation of new drug approval or withdrawal of marketed medicine based on safety due to organ vulnerability. The aim of this research is to investigate the potential abilities of four different in vitro cell models (L-02, HepG2, HepaRG, and hiHeps cell lines) in assessing marketed drugs labeled with apparently different types of liver injury. A total of 17 drugs with versatile pharmacological profiles were chosen, of which, 14 drugs are recognized as DILI agents and 3 drugs are DILI irrelevant. Preliminary cellular screening assays indicated that the HepaRG cell line had an advantage over other cell lines in predicting drugs associated with DILI in vitro as it had the highest Youden’s index (71.4 %). A multi-parametric screening assay showed that oxidative stress, mitochondrial damage, and disorders of neutral lipid metabolism were changed notably in the HepaRG cell line after DILI-related drugs exposure, accounting for its high sensitivity in comparison with other three cell lines. In addition, aspartate aminotransferase (AST), lactate dehydrogenase (LDH), and malate dehydrogenase (MDH) all correlated with the cytotoxic effects of diclofenac sodium (p < 0.05), buspirone hydrochloride (p < 0.01), and danazol (p < 0.01) in the HepaRG cell line. We conclude that the HepaRG cell line is a superior in vitro cell model to other three cell lines for evaluating drugs with DILI potential.













Similar content being viewed by others
Abbreviations
- ALP:
-
Alkaline phosphatase
- ALT:
-
Alanine aminotransferase
- APAP:
-
Acetaminophen
- AST:
-
Aspartate aminotransferase
- CCK-8:
-
Cell counting kit-8
- CHE:
-
Cholinesterase
- DILI:
-
Drug-induced liver injury
- GLDH:
-
Glutamate dehydrogenase
- γ-GT:
-
γ-Glutamyl transferase
- HCS:
-
High-content screening
- LDH:
-
Lactate dehydrogenase
- MAO:
-
Monoamine oxidase
- MDH:
-
Malate dehydrogenase
- NAT:
-
N-acetyltransferase
- 5’-NT:
-
5’-nucleotidase
- PSTC:
-
Predictive Safety Testing Consortium
- TPL:
-
Triptolide
References
Bjӧrnsson ES. Drug-induced liver injury: an overview over the most critical compounds. Arch Toxicol. 2015;89(3):327–34.
Cerec V, Glaise D, et al. Transdifferentiation of hepatocyte-like cells from the human hepatoma HepaRG cell line through bipotent progenitor. Hepatology. 2007;45(4):957–67.
Chen M, Vijay V, et al. FDA-approved drug labeling for the study of drug-induced liver injury. Drug Discov Today. 2011;16(15–16):697–703.
Cheong N, Madesh M, et al. Functional and trafficking defects in ATP binding cassette A3 mutants associated with respiratory distress syndrome. J Biol Chem. 2006;281(14):9791–800.
Danan, G, Teschke, R. RUCAM in drug and herb induced liver injury: the update. Int J Mol Sci. 2015;17(1). pii: E14.
Du Y, Wang J, et al. Human hepatocytes with drug metabolic function induced from fibroblasts by lineage reprogramming. Cell Stem Cell. 2014;14(3):394–403.
Esch MB, Mahler GJ, et al. Body-on-a-chip simulation with gastrointestinal tract and liver tissues suggests that ingested nanoparticles have the potential to cause liver injury. Lab Chip. 2014;14(16):3081–92.
Gomez-Lechon MJ, Tolosa L, et al. Mechanism-based selection of compounds for the development of innovative in vitro approaches to hepatotoxicity studies in the LIINTOP project. Toxicol In Vitro. 2010;24(7):1879–89.
Gripon P, Rumin S, et al. Infection of a human hepatoma cell line by hepatitis B virus. Proc Natl Acad Sci U S A. 2002;99(24):15655–60.
Hanumegowda UM, Copple BL, et al. Basement membrane and matrix metalloproteinases in monocrotaline-induced liver injury. Toxicol Sci. 2003;76(1):237–46.
Hu X, Yang T, et al. Human fetal hepatocyte line, L-02, exhibits good liver function in vitro and in an acute liver failure model. Transplant Proc. 2013;45(2):695–700.
Huang X, Qin J, et al. Magnesium isoglycyrrhizinate protects hepatic L02 cells from ischemia/reperfusion induced injury. Int J Clin Exp Pathol. 2014;7(8):4755–64.
Kia R, Kelly L, et al. MicroRNA-122: a novel hepatocyte-enriched in vitro marker of drug-induced cellular toxicity. Toxicol Sci. 2015;144(1):173–85.
Lheureux PE, Hantson P. Carnitine in the treatment of valproic acid-induced toxicity. Clin Toxicol (Phila). 2009;47(2):101–11.
Li N, Xia Q, et al. Hepatotoxicity and tumorigenicity induced by metabolic activation of pyrrolizidine alkaloids in herbs. Curr Drug Metab. 2011;12(9):823–34.
Li Y, Tian Y, et al. Determination of diosbulbin B in rat plasma and urine by LC-MS/MS and its application in pharmacokinetic and urinary excretion studies. J Pharm Biomed Anal. 2013;77:133–8.
Li XJ, Jiang ZZ, et al. Triptolide: progress on research in pharmacodynamics and toxicology. J Ethnopharmacol. 2014;155(1):67–79.
Lin D, Li C, et al. Cytochrome p450-mediated metabolic activation of diosbulbin B. Drug Metab Dispos. 2014;42(10):1727–36.
McGill MR, Yan HM, et al. HepaRG cells: a human model to study mechanisms of acetaminophen hepatotoxicity. Hepatology. 2011;53(3):974–82.
Mueller D, Heinzle E, et al. 3D hepatic in vitro models as tools for toxicity studies. Curr Tissue Eng. 2013;2(1):78–89.
Navarro VJ, Senior JR. Drug-related hepatotoxicity. N Engl J Med. 2006;354(7):731–9.
Oda S, Yokoi T. Establishment of animal models of drug-induced liver injury and analysis of possible mechanisms. Yakugaku Zasshi. 2015;135(4):579–88.
Persson M, Løye AF, et al. A high content screening assay to predict human drug-induced liver injury during drug discovery. J Pharmacol Toxicol Methods. 2013;68(3):302–13.
Schomaker S, Warner R, et al. Assessment of emerging biomarkers of liver injury in human subjects. Toxicol Sci. 2013;132(2):276–83.
Tarantino G, Conca P, et al. A prospective study of acute drug-induced liver injury in patients suffering from non-alcoholic fatty liver disease. Hepatol Res. 2007;37(6):410–5.
Tarantino G, Di Minno MN, et al. Drug-induced liver injury: is it somehow foreseeable? World J Gastroenterol. 2009a;15(23):2817–33.
Tarantino G, Pezzullo MG, et al. Drug-induced liver injury due to “natural products” used for weight loss: a case report. World J Gastroenterol. 2009b;15(19):2414–7.
Teschke R, Andrade RJ. Drug-induced liver injury: expanding our knowledge by enlarging population analysis with prospective and scoring causality assessment. Gastroenterology. 2015;148(7):1271–3.
Tolosa L, Pinto S, et al. Development of a multiparametric cell-based protocol to screen and classify the hepatotoxicity potential of drugs. Toxicol Sci. 2012;127(1):187–98.
Tomida T, Okamura H, et al. Multiparametric assay using HepaRG cells for predicting drug-induced liver injury. Toxicol Lett. 2015;236(1):16–24.
Turpeinen M, Tolonen A, et al. Functional expression, inhibition and induction of CYP enzymes in HepaRG cells. Toxicol In Vitro. 2009;23(4):748–53.
Varecha M, Amrichová J, et al. Bioinformatic and image analyses of the cellular localization of the apoptotic proteins endonuclease G, AIF, and AMID during apoptosis in human cells. Apoptosis. 2007;12(7):1155–71.
Wang AG, Xia T, et al. Effects of phenobarbital on metabolism and toxicity of diclofenac sodium in rat hepatocytes in vitro. Food Chem Toxicol. 2004;42(10):1647–53.
Wang J, Peng X, et al. Construction and application of dynamic protein interaction network based on time course gene expression data. Proteomics. 2013;13(2):301–12.
Xu C, Bailly-Maitre B, et al. Endoplasmic reticulum stress: cell life and death decisions. J Clin Invest. 2005;115(10):2656–64.
Xu JJ, Henstock PV, et al. Cellular imaging predictions of clinical drug-induced liver injury. Toxicol Sci. 2008;105(1):97–105.
Youden WJ. Index for rating diagnostic tests. Cancer. 1950;3(1):32–5.
Acknowledgments
The authors are grateful for Prof. Deng Hong-kui and his research team for supplying ample hiHeps cell line as well as excellent technical assistance in cell culture and liver function maintenance. This work was supported by the National Key Technology R&D Program of China (2012ZX09302001).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest.
Electronic supplementary material
Supplementary data to this article can be found online.
ESM 1
(DOC 26947 kb)
Rights and permissions
About this article
Cite this article
Wu, Y., Geng, Xc., Wang, Jf. et al. The HepaRG cell line, a superior in vitro model to L-02, HepG2 and hiHeps cell lines for assessing drug-induced liver injury. Cell Biol Toxicol 32, 37–59 (2016). https://doi.org/10.1007/s10565-016-9316-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10565-016-9316-2