Abstract
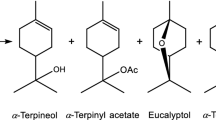
This study aims to produce terpineol from α-pinene by direct one-stage hydration reaction mechanism. The study was focused to investigate the influence of organic acids and mixture acid catalyst between organic acid and phosphoric acid. Based on the experimental results and depending on the acid strength and affinity of catalyst to the carbocation mechanism, p-toluensulfonic acid was shown to have a better result compared to acetic and formic acid. The presence of phosphoric acid could boost the performance of acetic acid in term of acidity strength that increased the conversion of the reaction. The highest yield of terpineol from α-pinene in this hydration reaction in this study was 53.5 wt% using mixed phosphoric and acetic acid.
Graphical Abstract

Similar content being viewed by others
References
Nuhfendi (2014) Feasibility analysis of perhutani gondarumen and trupentine-derived plant development. Master Thesis, Gadjah Mada University
Panda H (2005) Aromatic plants cultivation, processing and uses. Asia Pacific Business Press, Delhi
Sheffield DH (1936) Method for the production of terpineol from pinene, US Patent 2060597
Sheffield DH (1939) Method for the production of terpineol from pinene, US Patent 2178349
Utami H, Budiman A, Sutijan S, Roto R, Sediawan WB (2010) Synthesis of α-terpineol from turpentine by hydration in a batch reactor. In: 17th ASEAN regional symposium on Chemical Engineering, Bangkok, Thailand
Pakdel H, Sarron S, Roy C (2001) α-Terpineol from hydration of crude sulfate turpentine oil. J Agric Food Chem 49:4337
Mckim PA (1949) Terpinol hydrate and terpineol from sulfate turpentine, PhD Dissertation, Louisiana State University
Wijayati N, Pranowo HD, Jumina T (2011) Synthesis of terpineol from α-pinene catalyzed by TCA/Y-Zeolite. Indo J Chem 11:234
Amilia N (2013) Pengaruh Temperatur pada Reaksi Hidrasi α-Pinena menjadi α-Terpineol Sistem Heterogen dengan Katalis Zeolit Alam Teraktivasi (in Bahasa), Bachelor Thesis, Universitas Negeri Semarang
Indarto A, Yang DR, Choi JW, Lee H, Song HK (2007) CCl4 decomposition by gliding arc plasma: role of C2 compounds on products distribution. Chem Eng Commun 194:1111
Indarto A, Choi JW, Lee H, Song HK (2008) The kinetic studies of direct methane oxidation to methanol in the plasma process. Chin Sci Bull 18:2783
Hartanto Y, Yaswari Y, Zunita M, Soerawidjaja TH, Indarto A (2017) Decolorization of crude terpineol by adsorption. Sep Sci Technol 52:1967
Tama Y (2012) Synthesis Terpineol from Turpentine, Master Thesis, Institut Teknologi Bandung
Yang G, Liu Y, Zhou Z, Zhang Z (2011) Kinetic study of the direct hydration of turpentine. Chem Eng J 168:351
Herrlinger R (1959) Production of alpha terpineol, US Patent 1898380
Aguirre MR, De la Torre-Saenz L, Flores WA, Sanchez AR, Elguezabal AA (2005) Synthesis of terpineol from a-pinene by homogeneous acid catalysis. Catal Today 107–108:310
Baghernejad B (2011) Application of p-toluenesulfonic acid (PTSA) in organic synthesis. Curr Org Chem 15:3091
Falbe J, Bahrmann H, Lipps W, Mayer D (2000) Alcohols, aliphatic, ullman’s encyclopedia of industrial chemistry. Wiley, New York
Liu SW, Yu ST, Liu FS, Xie CX, Li L, Ji KH (2008) Reactions of pinene using acidic ionic liquids as catalysts. J Mol Catal A 279:177
Acknowledgements
The authors acknowledge a generous funding from the Program Penelitian, Pengabdian kepada Masyarakat, dan Inovasi (P3MI-2017) Kelompok Keahlian ITB 2017. Part of this research was also funded by 2018 KIST School Partnership Project of Korea Institute of Science and Technology (KIST). The author would like to thank Perhutani Pine Chemical Industry (PPCI), Pemalang, Indonesia for the research support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All contributing authors certify that they have NO affiliations with or involvement in any organization or entity with any financial or non-financial interest in the subject matter contained in this manuscript.
Rights and permissions
About this article
Cite this article
Prakoso, T., Hanley, J., Soebianta, M.N. et al. Synthesis of Terpineol from α-Pinene Using Low-Price Acid Catalyst. Catal Lett 148, 725–731 (2018). https://doi.org/10.1007/s10562-017-2267-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-017-2267-2