Abstract
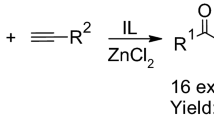
The synthesis of a series of fourteen 4-alkoxy-1,1,1-trihalo-3-alken-2-ones (2,3) [CX3COC(R2)=C(R1)OMe, where X = Cl, F; R1/R2 = Me/H, Bu/H, i-Bu/H, Ph/H, Thien-2-yl/H, –(CH2)4–, –CH(CH2)4CH(CH2)2–] from the acylation reactions of acetals (1) with trichloroacetyl chloride or trifluoroacetic anhydride in the presence of equimolar amounts of pyridine and imidazolium based ionic liquid ([BMIM][BF4] or [BMIM][PF6]) is reported. The reaction time, yields and IL recyclation are also investigated and this method showed advantages over the methods described in the literature.
Similar content being viewed by others
References
Martins MAP, Cunico W, Pereira CMP, Sinhorin AP, Flores AFC, Bonacorso HG, Zanatta N (2004) Curr Org Chem 1:391
Druzhinin SV, Balenkova ES, Nenajdenko VG (2007) Tetrahedron 63:7753
Seyferth D, Weinstein RM, Hui RC, Wang WL, Archer CM (1991) J Org Chem 56:5768
Ryu I, Hayama Y, Hirai A, Sonoda N, Orita A, Ohe K, Murai S (1990) J Am Chem Soc 112:7061
Chatani N, Fukuyama T, Kakiuchi F, Murai S (1996) J Am Chem Soc 118:493
Larock RC (1999) Comprehensive organic transformation. Wiley-VCH, New York
Fischer P (1980) Enol ethers—structure, synthesis and reactions, in the chemistry of functional groups. Wiley-Interscience, New York. Chap. 17, suppl E, part 2
Colla A, Martins MAP, Clar G, Krimmer S, Fischer P (1991) Synthesis 483
Martins MAP, Guarda EA, Frizzo CP, Scapin E, Beck P, Costa AC, Zanatta N, Bonacorso HG (2006) J Mol Catal A Chem 266:100
Bonacorso HG, Martins MAP, Bittencourt SRT, Lourega RV, Zanatta N, Flores AFC (1999) J Fluor Chem 99:177
Martins MAP, Bastos GP, Bonacorso HG, Zanatta N, Flores AFC, Siqueira GM (1999) Tetrahedron Lett 40:430
Flores AFC, Siqueira GM, Freitag R, Zanatta N, Martins MAP (1994) Quim Nova 17:24 Chem Abstr, 121:230377
Flores AFC, Brondani S, Zanatta N, Rosa A, Martins MAP (2002) Tetrahedron Lett 43:870
Chiappe C, Pieraccini D (2005) J Phys Org Chem 18:275
Olivier-Bourbigou H, Magna L (2002) J Mol Catal A Chem 182–183:419
Wasserscheid P, Keim W (2000) Angew Chem Int Ed 39:3772
Welton T (1999) Chem Rev 99:2071
Pârvulescu VI, Hardacre C (2007) Chem Rev 107:2615
Welton T (2004) Coord Chem Rev 248:2459
Souza R, Suarez PAZ, Consorti CS, Dupont J (2002) Org Synth 79:236
Wohl RA (1974) Synthesis 38
Shubin VG, Borodkin GI (2006) Rus J Org Chem 42:1761
Xu LW, Gao Y, Yin JJ, Li L, Chun-Gu X (2005) Tetrahedron Lett 46:5317
Martins MAP, Frizzo CP, Moreira DN, Zanata N, Bonacorso HG (2008) Chem Rev 108:2015
Martins MAP, Peres RL, Fiss GF, Dimer FA, Mayer R, Frizzo CP, Marzari MRB, Zanatta N, Bonacorso HG (2007) J Braz Chem Soc 18:1486
Acknowledgments
The authors thank the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq, Contracts No. 46.0804/00-6 and 480067/01-5), Fundação de Amparo à Pesquisa do Estado do Rio Grande do Sul (FAPERGS) and CAPES for financial support and fellowships.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Martins, M.A.P., Guarda, E.A., Frizzo, C.P. et al. Ionic Liquids Promoted the C-Acylation of Acetals in Solvent-free Conditions. Catal Lett 130, 93–99 (2009). https://doi.org/10.1007/s10562-009-9873-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-009-9873-6