Abstract
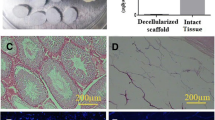
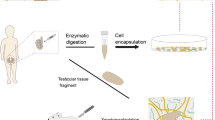
We determined histological aspects of implanted human decellularized testicular matrix (DTM) in C57BL6 as a primitive step for further testis tissue engineering. A total of 4 immature human testicles were obtained after bilateral orchiectomy from patients with testicular feminization syndrome. The optimal decellularization protocol was determined and the efficacy of decellularization was evaluated in two of the testicles. The remaining scaffolds were cut in 3 × 3 mm3 pieces and implanted between the tight muscles in 32 C57BL6. Biopsies were taken at 2, 4, 8, and 24 weeks postoperatively and stained with PLZF, protamine, and tekt1 markers. Histological examination of DTMs confirmed complete absence of nuclear remnants and preservation of the extracellular matrix. Successful cell seeding was observed in all follow-ups confirmed by H&E and IHC staining that increased continuously during the whole study. Interestingly, spermatogonial stem-like cells were observed on decellularized implants that were well differentiated during the follow-ups. Natural bioreactors may provide a good cell source for testes tissue regeneration. This technique may provide testis bioscaffold as a three-dimensional platform and further successful cell seeding to produce a functional testis. This novel technique may be beneficial for patients who require testicular supplementation.






Similar content being viewed by others
Abbreviations
- DTM:
-
Decellularized testicular matrix
- ECM:
-
Extracellular matrix
References
Abrishami M, Anzar M, Yang Y, Honaramooz A (2010) Cryopreservation of immature porcine testis tissue to maintain its developmental potential after xenografting into recipient mice. Theriogenology 73:86–96. https://doi.org/10.1016/j.theriogenol.2009.08.004
Abu Elhija M, Lunenfeld E, Schlatt S, Huleihel M (2012) Differentiation of murine male germ cells to spermatozoa in a soft agar culture system. Asian J Androl 14:285–293. doi:https://doi.org/10.1038/aja.2011.112
Amann RP (2008) The cycle of the seminiferous epithelium in humans: a need to revisit? J Androl 29:469–487. doi:https://doi.org/10.2164/jandrol.107.004655
Aslam I, Fishel S (1998) Short-term in-vitro culture and cryopreservation of spermatogenic cells used for human in-vitro conception . Hum Reprod (Oxf, Engl) 13:634–638
Baert Y, Stukenborg JB, Landreh M, De Kock J, Jornvall H, Soder O, Goossens E (2015) Derivation and characterization of a cytocompatible scaffold from human testis. Hum Reprod (Oxf, Engl) 30:256–267. https://doi.org/10.1093/humrep/deu330
Chan BP, Leong KW (2008) Scaffolding in tissue engineering: general approaches and tissue-specific considerations. Europ Spine J Off Publ Eur Spine Soc Eur Spinal Deform Soc Eur Sect Cerv Spine Res Soc 17(Suppl 4):467–479. https://doi.org/10.1007/s00586-008-0745-3
Del Vento F, Vermeulen M, de Michele F, Giudice MG, Poels J, des Rieux A, Wyns C, (2018) Tissue engineering to improve immature testicular tissue and cell transplantation outcomes: one step closer to fertility restoration for prepubertal boys exposed to gonadotoxic treatments. Int J Mol Sci. https://doi.org/10.3390/ijms19010286
Geens M, De Block G, Goossens E, Frederickx V, Van Steirteghem A, Tournaye H (2006) Spermatogonial survival after grafting human testicular tissue to immunodeficient mice . Hum Reprod (Oxf, Engl) 21:390–396. https://doi.org/10.1093/humrep/dei412
Goossens E, Van Saen D, Tournaye H (2013) Spermatogonial stem cell preservation and transplantation: from research to clinic. Hum Reprod (Oxf, Engl) 28:897–907. https://doi.org/10.1093/humrep/det039
Gow A et al (1999) CNS myelin and sertoli cell tight junction strands are absent in Osp/claudin-11 null mice. Cell 99:649–659
Jahnukainen K, Ehmcke J, Nurmio M, Schlatt S (2012a) Autologous ectopic grafting of cryopreserved testicular tissue preserves the fertility of prepubescent monkeys that receive sterilizing cytotoxic. Ther Cancer Res 72:5174–5178. doi:https://doi.org/10.1158/0008-5472.can-12-1317
Jahnukainen K, Ehmcke J, Nurmio M, Schlatt S (2012b) Fertility preservation after sterilizing therapy through autologous ectopic grafting of cryopreserved testicular tissue in prepubertal monkeys. Cancer Res (in press)
Kaneko H et al (2013) Generation of live piglets for the first time using sperm retrieved from immature testicular tissue cryopreserved and grafted into nude mice. PloS ONE 8:e70989. https://doi.org/10.1371/journal.pone.0070989
Lee JH, Kim HJ, Kim H, Lee SJ, Gye MC (2006) In vitro spermatogenesis by three-dimensional culture of rat testicular cells in collagen gel matrix. Biomaterials 27:2845–2853. https://doi.org/10.1016/j.biomater.2005.12.028
Mirone V et al (2017) European Association of urology position statement on the role of the urologist in the management of male hypogonadism and testosterone therapy. Eur Urol 72:164–167. https://doi.org/10.1016/j.eururo.2017.02.022
Reda A, Hou M, Landreh L, Kjartansdottir KR, Svechnikov K, Soder O, Stukenborg JB (2014) In vitro Spermatogenesis—optimal culture conditions for testicular cell survival, germ cell differentiation, and steroidogenesis in rats. Front Endocrinol 5:21. https://doi.org/10.3389/fendo.2014.00021
Reuter K et al (2014) Reassembly of somatic cells and testicular organogenesis in vitro. Tissue Cell 46:86–96. doi:https://doi.org/10.1016/j.tice.2013.12.001
Sato T et al (2011) In vitro production of functional sperm in cultured neonatal mouse testes. Nature 471:504–507. https://doi.org/10.1038/nat09850
Storm D, Redden T, Aguiar M, Wilkerson M, Jordan G, Sumfest J (2007) Histologic evaluation of the testicular remnant associated with the vanishing testes syndrome: is surgical management necessary? Urology 70:1204–1206. https://doi.org/10.1016/j.urol.2007.08.020
Stukenborg JB et al (2009) New horizons for in vitro spermatogenesis? An update on novel three-dimensional culture systems as tools for meiotic and post-meiotic differentiation of testicular germ cells. Mol Hum Reprod 15:521–529. doi:https://doi.org/10.1093/molehr/gap052
Tanaka A, Nagayoshi M, Awata S, Mawatari Y, Tanaka I, Kusunoki H (2003) Completion of meiosis in human primary spermatocytes through in vitro coculture with Vero cells. Fertil Steril 79(Suppl 1):795–801
Tesarik J, Greco E, Rienzi L, Ubaldi F, Guido M, Cohen-Bacrie P, Mendoza C (1998) Differentiation of spermatogenic cells during in-vitro culture of testicular biopsy samples from patients with obstructive azoospermia: effect of recombinant follicle stimulating hormone . Hum Reprod (Oxf, Engl) 13:2772–2781
Valli H, Phillips BT, Shetty G, Byrne JA, Clark AT, Meistrich ML, Orwig KE (2014) Germline stem cells: toward the regeneration of spermatogenesis. Fertil Steril 101:3–13. doi:https://doi.org/10.1016/j.fertnstert.2013.10.052
Van Saen D, Goossens E, De Block G, Tournaye H (2009) Regeneration of spermatogenesis by grafting testicular tissue or injecting testicular cells into the testes of sterile mice: a comparative study. Fertil Steril 91:2264–2272. doi:https://doi.org/10.1016/j.fertnstert.2008.02.100
Yokonishi T, Sato T, Katagiri K, Komeya M, Kubota Y, Ogawa T (2013) In vitro reconstruction of mouse seminiferous tubules supporting germ cell differentiation. Biol Reprod 89:15. https://doi.org/10.1095/biolreprod.113.108613
Acknowledgements
The authors gratefully acknowledge Tehran University of Medical Sciences and Health Services for funding this manuscript (Grant Number: 33177).
Funding
This work was supported by the Tehran University of Medical Sciences (Grant Number: 33177).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None of the authors have any conflict of interest, and will sign the Disclosing Form.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sabetkish, S., Kajbafzadeh, AM. & Sabetkish, N. Recellularization of testicular feminization testis in C57bl6 as a natural bioreactor for creation of cellularized seminiferous tubules: an experimental study. Cell Tissue Bank 22, 287–295 (2021). https://doi.org/10.1007/s10561-020-09877-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10561-020-09877-6