Abstract
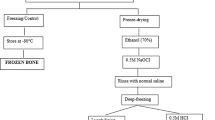
Reconstruction of large skeletal defects is a significant and challenging issue. Bone allografts are often used for such reconstructions. However, sterilizing bone allografts by using γ-irradiation, damages collagen and causes the bone to become weak, brittle and less fatigue resistant. In a previous study, we successfully protected the mechanical properties of human cortical bone by conducting a pre-treatment with ribose, a natural and biocompatible agent. This study focuses on examining possible mechanisms by which ribose might protect the bone. We examined the mechanical properties, crosslinking, connectivity and free radical scavenging potentials of the ribose treatment. Human cortical bone beams were treated with varying concentration of ribose (0.06–1.2 M) and γ-irradiation before testing them in 3-point bending. The connectivity and amounts of crosslinking were determined with Hydrothermal-Isometric-Tension testing and High-Performance-Liquid-Chromatography, respectively. The free radical content was measured using Electron Paramagnetic Resonance. Ribose pre-treatment improved the mechanical properties of irradiation sterilized human bone in a pre-treatment concentration-dependent manner. The 1.2 M pre-treatment provided >100% of ultimate strength of normal controls and protected 76% of the work-to-fracture (toughness) lost in the irradiated controls. Similarly, the ribose pre-treatment improved the thermo-mechanical properties of irradiation-sterilized human bone collagen in a concentration-dependent manner. Greater free radical content and pentosidine content were modified in the ribose treated bone. This study shows that the mechanical properties of irradiation-sterilized cortical bone allografts can be protected by incubating the bone in a ribose solution prior to irradiation.






Similar content being viewed by others
References
Akkus O, Rimnac CM (2001) Fracture resistance of gamma radiation sterilized cortical bone allografts. J Orthop Res 19:927–934. doi:10.1016/S0736-0266(01)00004-3
Akkus O, Belaney RM (2005) Sterilization by gamma radiation impairs the tensile fatigue life of cortical bone by two orders of magnitude. J Orthop Res 23(1054–1058):2005. doi:10.1016/J.Orthres.2005.03.003
Akkus O, Belaney RM, Das P (2005) Free radical scavenging alleviates the biomechanical impairment of gamma radiation sterilized bone tissue. J Orthop Res 23(838–845):2005. doi:10.1016/J.Orthres.2005.01.007
Benedict SR (1909) A reagent for the detection of reducing sugars. J Biol Chem 5:485–487
Breen SL, Battista JJ (1995) Radiation dosimetry in human bone using electron paramagnetic resonance. Phys Med Biol 40:2065–2077. doi:10.1088/0031-9155/40/12/005
Burton B (2013) Improving the mechanical properties of irradiation sterilized bone allografts master of applied science Thesis, Institute of Biomaterials and Biomedical Engineering University of Toronto
Burton B, Gaspar A, Josey D, Tupy J, Grynpas MD, Willett TL (2014) Bone embrittlement and collagen modifications due to high-dose gamma-irradiation sterilization. Bone 61C:71–81. doi:10.1016/j.bone.2014.01.006
Catanese J, III, Bank RA, Tekoppele JM, Keaveny TM (1999) Increased cross-linking by non-enzymatic glycation reduces the ductility of bone and bone collagen bioengineering conference. ASME 42:267–268
Currey JD, Foreman J, Laketic I, Mitchell J, Pegg DE, Reilly GC (1997) Effects of ionizing radiation on the mechanical properties of human bone. J Orthop Res 15:111–117. doi:10.1002/Jor.1100150116
Dong XN, Qin A, Xu JK, Wang XD (2011) In situ accumulation of advanced glycation endproducts (AGEs) in bone matrix and its correlation with osteoclastic bone resorption. Bone 49:174–183. doi:10.1016/j.bone.2011.04.009
Enneking WE, Campanacci DA (2001) Retrieved human allografts—a clinicopathological study. J Bone Joint Surg Am 83A:971–986
Goldberg VM (2008) Biology of bone allograft and clinical application. In: Pietrzak W (ed) Musculoskeletal tissue regeneration: biological materials and methods, vol 5. Humana Press, New York, pp 81–92
Greenwald AS, Boden SD, Goldberg VM, Khan Y, Laurencin CT, Rosier RN, Implants CB (2001) Bone-graft substitutes: facts, fictions, and applications. J Bone Joint Surg Am 83A:98–103
Houben JL (1971) Free radicals produced by ionizing radiation in bone and its constituents. Int J Radiat Biol Relat Stud Phys Chem Med 20:373–389
Kawaguchi S, Hart RA (2015) The need for structural allograft biomechanical guidelines. J Am Acad Orthop Surg 23:119–125. doi:10.5435/JAAOS-D-14-00263
Komender A (1976) Influence of preservation on some mechanical properties of human haversian bone. Mater Med Pol 1:13–17
Lietman SA, Tomford WW, Gebhardt MC, Springfield DS, Mankin HJ (2000) Complications of irradiated allografts in orthopaedic tumor surgery. Clin Orthop Relat Res 375:214–217
Mitchell EJ, Stawarz AM, Kayacan R, Rimnac CM (2004) The effect of gamma radiation sterilization on the fatigue crack propagation resistance of human cortical bone. J Bone Joint Surg Am 86A:2648–2657
Nakamura K, Nakazawa Y, Ienaga K (1997) Acid-stable fluorescent advanced glycation end products: vesperlysines A, B, and C are formed as crosslinked products in the maillard reaction between lysine or proteins with glucose. Biochem Biophys Res Commun 232:227–230. doi:10.1006/Bbrc.1997.6262
Nguyen H, Morgan DA, Forwood MR (2007) Sterilization of allograft bone: effects of gamma irradiation on allograft biology and biomechanics. Cell Tissue Bank 8:93–105. doi:10.1007/s10561-006-9020-1
Norman TL, Wang Z (1997) Microdamage of human cortical bone: incidence and morphology in long bones. Bone 20:375–379. doi:10.1016/S8756-3282(97)00004-5
Ritchie RO, Kinney JH, Kruzic JJ, Nalla RK (2005) A fracture mechanics and mechanistic approach to the failure of cortical bone. Fatigue Fract Eng Mater Struct 28:345–371. doi:10.1111/J.1460-2695.2005.00878.X
Salehpour A, Butler DL, Proch E, Schwartz HE, Feder SM, Doxey CM, Ratcliffe A (1995) Dose-dependent response of gamma irradiation on mechanical properties and related biochemical composition of coat bone-patellar tendon-bone allografts. J Orthop Res 13:898–906. doi:10.1002/Jor.1100130614
Seto A, Gatt CJ, Dunn MG (2008) Radioprotection of tendon tissue via crosslinking and free radical scavenging. Clin Orthop Relat Res 466:1788–1795. doi:10.1007/S11999-008-0301-9
Seto A, Gatt CJ, Dunn MG (2009) Improved Tendon Radioprotection by Combined Cross-linking and Free Radical Scavenging. Clin Orthop Relat Res 467:2994–3001. doi:10.1007/S11999-009-0934-3
Sroga GE, Siddula A, Vashishth D (2015) Glycation of human cortical and cancellous bone captures differences in the formation of maillard reaction products between glucose and ribose. PLoS ONE 10:e0117240. doi:10.1371/journal.pone.0117240
Tang SY, Vashishth D (2011) The relative contributions of non-enzymatic glycation and cortical porosity on the fracture toughness of aging bone. J Biomech 44:330–336. doi:10.1016/j.jbiomech.2010.10.016
Thompson RC Jr, Garg A, Clohisy DR, Cheng EY (2000) Fractures in large-segment allografts. Clin Orthop Relat Res 370:227–235
Turner CH (2006) Bone strength: current concepts. Ann NY Acad Sci 1068:429–446. doi:10.1196/Annals.1346.039
Vashishth AK (2009) Advanced glycation end-products and bone fractures. IBMS BoneKEy 6:268–278
Vashishth D, Gibson GJ, Khoury JI, Schaffler MB, Kimura J, Fyhrie DP (2001) Influence of nonenzymatic glycation on biomechanical properties of cortical bone. Bone 28:195–201. doi:10.1016/S8756-3282(00)00434-8
Wang X, Shen X, Li X, Agrawal CM (2002) Age-related changes in the collagen network and toughness of bone. Bone 31:1–7. doi:10.1016/S8756-3282(01)00697-4
Willett TL, Sutty S, Gaspar A, Avery N, Grynpas M (2013) In vitro non-enzymatic ribation reduces post-yield strain accommodation in cortical bone. Bone 52:611–622. doi:10.1016/j.bone.2012.11.014
Willett TL, Burton B, Woodside M, Wang Z, Gaspar A, Attia T (2015) Gamma-irradiation sterilized bone strengthened and toughened by ribose pre-treatment. J Mech Behav Biomed Mater 44C:147–155. doi:10.1016/j.jmbbm.2015.01.003
Woodside M, Willett TL (2016) Elastic–plastic fracture toughness and rising J(R)-curve behavior of cortical bone is partially protected from irradiation–sterilization-induced degradation by ribose protectant. J Mech Behav Biomed Mater 64:53–64. doi:10.1016/j.jmbbm.2016.07.001
Zimmermann EA, Gludovatz B, Schaible E, Busse B, Ritchie RO (2014) Fracture resistance of human cortical bone across multiple length-scales at physiological strain rates. Biomaterials 35(5472–5481):2014. doi:10.1016/J.Biomaterials.03.066
Zioupos P, Currey JD, Hamer AJ (1999) The role of collagen in the declining mechanical properties of aging human cortical bone. J Biomed Mater Res 45:108–116. doi:10.1002/(Sici)1097-4636(199905)45:2<108:Aid-Jbm5>3.0.Co;2-A
Zioupos P, Gresle M, Winwood K (2008) Fatigue strength of human cortical bone: age, physical, and material heterogeneity effects. J Biomed Mater Res, Part A 86A:627–636. doi:10.1002/Jbm.A.31576
Acknowledgements
This work was funded by the Canadian Institute of Health Research, the Natural Science and Engineering Research Council of Canada, and scholarships from the University of Toronto Institute for Biomaterials and Biomedical Engineering and Toronto Musculoskeletal Centre. The authors also acknowledge the contributions made by Mr. Jindra Tupy and Mr. Doug Holmyard. We acknowledge the contributions of our tissue-banking partner, Mount Sinai Allograft Technologies.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Attia, T., Woodside, M., Minhas, G. et al. Development of a novel method for the strengthening and toughening of irradiation-sterilized bone allografts. Cell Tissue Bank 18, 323–334 (2017). https://doi.org/10.1007/s10561-017-9634-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10561-017-9634-5