Abstract
Purpose
Increased myocardial infarct (MI) size is associated with higher risk of developing left ventricular dysfunction, heart failure and mortality. Experimental studies have suggested that metformin treatment reduces MI size after induced ischaemia but human data is lacking. We aimed to investigate the effect of metformin on MI size in patients presenting with an acute MI.
Methods
All consecutive patients (n = 3,288) presenting to our hospital with ST-segment elevation myocardial infarction (STEMI) undergoing primary PCI between January 2004 and December 2010 were included in this retrospective analysis. Patients with diabetes were divided according to metformin versus non-metformin based pharmacotherapy. MI size was estimated using peak values of serum creatine kinase (CK), myocardial band of CK (CK-MB), and troponin-T.
Results
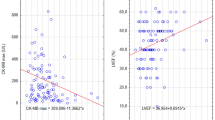
We identified 677 (20.6 %) patients with diabetes, of whom 189 (27.9 %) were treated with metformin. Chronic metformin treatment was associated with lower peak levels of CK (1,101 vs. 1,422 U/L, P = 0.005), CK-MB (152 vs. 182 U/L, P = 0.018) and troponin-T (2.5 vs. 4.0 ng/L, P = 0.021) compared to non-metformin using diabetics. After adjustment for age, sex, TIMI flow post PCI, and previous MI, the use of metformin treatment remained an independent predictor of smaller MI size. Patient with diabetes treated with metformin had even smaller MI size than patients without diabetes.
Conclusions
Chronic metformin treatment is associated with reduced MI size compared to non-metformin based strategies in diabetic patients presenting with STEMI. Metformin might have additional beneficial effects beyond glucose lowering efficacy.

Similar content being viewed by others
References
Fokkema ML, James SK, Albertsson P, et al. Population trends in percutaneous coronary intervention: 20-year results from the SCAAR (Swedish Coronary Angiography and Angioplasty Registry). J Am Coll Cardiol. 2013;61:1222–30.
Task Force on the management of ST-segment elevation acute myocardial infarction of the European Society of Cardiology (ESC), Steg PG, James SK, Atar D, et al. ESC Guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation. Eur Heart J. 2012;33:2569–619.
Burns RJ, Gibbons RJ, Yi Q, CORE Study Investigators, et al. The relationship of left ventricular ejection fraction, end-systolic volume index and infarct size to six-month mortality after hospital discharge following myocardial infarction treated by thrombolysis. J Am Coll Cardiol. 2002;39:30–6.
van der Vleuten PA, Rasoul S, Huurnink W, et al. The importance of left ventricular function for long-term outcome after primary percutaneous coronary intervention. BMC Cardiovasc Disord. 2008;8:4.
Bailey CJ. Metformin: effects on micro and macrovascular complications in type 2 diabetes. Cardiovasc Drugs Ther. 2008;22:215–24.
UK Prospective Diabetes Study (UKPDS) group. Effect of intensive blood-glucose control with metformin on complications in overweight patients with type 2 diabetes (UKPDS 34). Lancet. 1998;352:854–65.
Mellbin LG, Malmberg K, Norhammar A, Wedel H, Rydén L, DIGAMI 2 Investigators. Prognostic implications of glucose-lowering treatment in patients with acute myocardial infarction and diabetes: experiences from an extended follow-up of the Diabetes Mellitus Insulin-Glucose Infusion in Acute Myocardial Infarction (DIGAMI) 2 study. Diabetologia. 2011;54:1308–17.
Zhao JL, Fan CM, Yang YJ, et al. Chronic pretreatment of metformin is associated with the reduction of the no-reflow phenomenon in patients with diabetes mellitus after primary angioplasty for acute myocardial infarction. Cardiovasc Ther. 2013;31:60–4.
Yin M, van der Horst IC, van Melle JP, et al. Metformin improves cardiac function in a nondiabetic rat model of post-MI heart failure. Am J Physiol Heart Circ Physiol. 2011;301:H459–68.
Gundewar S, Calvert JW, Jha S, et al. Activation of AMP-activated protein kinase by metformin improves left ventricular function and survival in heart failure. Circ Res. 2009;104:403–11.
Paiva M, Riksen NP, Davidson SM, et al. Metformin prevents myocardial reperfusion injury by activating the adenosine receptor. J Cardiovasc Pharmacol. 2009;53:373–8.
Solskov L, Løfgren B, Kristiansen SB, et al. Metformin induces cardioprotection against ischaemia/reperfusion injury in the rat heart 24 h after administration. Basic Clin Pharmacol Toxicol. 2008;103:82–7.
Calvert JW, Gundewar S, Jha S, et al. Acute metformin therapy confers cardioprotection against myocardial infarction via AMPK-eNOS-mediated signaling. Diabetes. 2008;57:696–705.
Whittington HJ, Hall AR, McLaughlin CP, Hausenloy DJ, Yellon DM, Mocanu MM. Chronic metformin associated cardioprotection against infarction: not just a glucose lowering phenomenon. Cardiovasc Drugs Ther. 2013;27:5–15.
Cittadini A, Napoli R, Monti MG, et al. Metformin prevents the development if chronic heart failure in the SHHF rat model. Diabetes. 2012;61:944–53.
Standards of medical care. Diabetes Care. 2011;34(S1):S11–61.
Austen WG, Edwards JE, Frye RL, et al. A reporting system on patients evaluated for coronary artery disease. Report of the Ad Hoc Committee for Grading of Coronary Artery Disease, Council on Cardiovascular Surgery, American Heart Association. Circulation. 1975;51:5–40.
Chesebro JH, Knatterud G, Roberts R, et al. Thrombolyis in myocardial infarction (TIMI) trial, phase I: a comparison between intravenous tissue plasminogen activator and intravenous streptokinase. Clinical findings through hospital discharge. Circulation. 1987;76:142–54.
van’t Hof AW, Liem A, Suryapranata H, Hoorntje JC, de Boer MJ, Zijlstra F. Angiographic assessment of myocardial reperfusion in patients treated with primary angioplasty for acute myocardial infarction: myocardial blush grade. Circulation. 1998;97:2302–6.
Ingkanisorn WP, Rhoads KL, Aletras AH, Kellman P, Arai AE. Gadolineum delayed enhancement cardiovascular magnetic resonance correlates with clinical measures of myocardial infarction. J Am Coll Cardiol. 2004;43:2253–9.
Whittington HJ, Babu GG, Mocanu MM, et al. The diabetic heart: too sweet for its own good? Cardiol Res Pract. 2012;2012:845698.
Timmer JR, van der Horst IC, Ottervanger JP, et al. Prognostic value of admission glucose in non-diabetic patients myocardial infarction. Am Heart J. 2004;148:399–404.
Timmer JR, Hoekstra M, Nijsten MW, et al. Prognostic value of admission glycosylated hemoglobin and glucose in nondiabetic patients with ST-segment-elevation myocardial infarction treated with percutaneous coronary intervention. Circulation. 2011;124:704–11.
Mather AN, Creab A, Abidin N, et al. Relationship of dysglycemia to acute myocardial infarct size and cardiovascular outcome as determined by cardiovascular magnetic resonance. J Cardiovasc Magn Reson. 2010;12:61.
El Messaoudi S, Rongen GA, de Boer RA, Riksen NP. The cardioprotective effects of metformin. Curr Opin Lipidol. 2011;22:445–53.
Jalving M, Gietema JA, Lefrandt JD, et al. Metformin: taking away the candy for cancer? Eur J Cancer. 2010;46:2369–80.
Bhamra GS, Hausenloy DJ, Davidson SM, et al. Metformin protects the ischemic heart by the Akt-mediated inhibition of mitochondrial permeability transition pore opening. Basic Res Cardiol. 2008;103:274–84.
Lexis CP, van der Horst IC, Lipsic E, for the GIPS-III Investigators, et al. Metformin in non-diabetic patients presenting with ST elevation myocardial infarction: rationale and design of the glycometabolic intervention as adjunct to primary percutaneous intervention in ST elevation myocardial infarction (GIPS)-III trial. Cardiovasc Drugs Ther. 2012;26:417–26.
Cuthbertson J, Patterson S, O’Harte FP, Bell PM. Investigation of the effect of oral metformin on dipeptidylpeptidase-4 (DPP-4) activity in Type 2 diabetes. Diabet Med. 2009;26:649–54.
Timmers L, Henriques JP, de Kleijn DP, et al. Exenatide reduces infarct size and improves cardiac function in a porcine model of ischemia and reperfusion injury. J Am Coll Cardiol. 2009;53:501–10.
Lønborg J, Kelbæk H, Vejlstrup N, et al. Exenatide reduces final infarct size in patients with ST-segment-elevation myocardial infarction and short-duration of ischemia. Circ Cardiovasc Interv. 2012;5:288–95.
Lexis CP, van der Horst IC, Lipsic E. Effects of metformin on insulin resistance in heart failure. Which came first: the chicken or the egg? Eur J Heart Fail. 2012;14:1197–8.
Tsikouris JP, Suarez JA, Meyerrose GE. Plasminogen activator inhibitor-1: physiologic role, regulation, and the influence of common pharmacologic agents. J Clin Pharmacol. 2002;42:1187–99.
Ersoy C, Kiyici S, Budak F, et al. The effect of metformin treatment on VEGF and PAI-1 levels in obese type 2 diabetic patients. Diabetes Res Clin Pract. 2008;81:56–60.
Protti A, Lecchi A, Fortunato F, et al. Metformin overdose causes platelet mitochondrial dysfunction in humans. Crit Care. 2012;16:R180.
Owen MR, Doran E, Halestrap AP. Evidence that metformin exerts its anti-diabetic effects through inhibition of complex 1 of the respiratory chain. Biochem J. 2000;348:607–14.
Funding Source
This study was supported by grant 95103007 from ZonMW, the Netherlands Organization for Health Research and Development, The Hague, the Netherlands.
Conflict of Interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lexis, C.P.H., Wieringa, W.G., Hiemstra, B. et al. Chronic Metformin Treatment is Associated with Reduced Myocardial Infarct Size in Diabetic Patients with ST-segment Elevation Myocardial Infarction. Cardiovasc Drugs Ther 28, 163–171 (2014). https://doi.org/10.1007/s10557-013-6504-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10557-013-6504-7