Abstract
Hypothesis
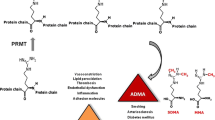
Asymmetric dimethylarginine (ADMA), an endogenous nitric oxide synthase inhibitor (NOS), may play an important role in endothelium dysfunction. Probucol, a potent antioxidant drug, may improve endothelium function via reduction of NOS inhibitor level. The present study examined whether the decreased level of ADMA by probucol is related to enhancement of protein arginine methyltransferase I (PRMT I) expression and reduction of dimethylarginine dimethylaminohydrolase (DDAH) activity.
Methods
Endothelial cells were cultured and used for all these studies. ADMA concentration and DDAH activity were determined by HPLC. Expression of PRMT I and eNOS were characterized by western blot.
Results
Pretreatment with oxidized-low density lipoprotein (ox-LDL) (10, 30 or 100 μg/ml) or lysophosphatidylcholine (LPC) (1.0, 2.5 or 5.0 μg/ml) for 12, 24 or 48 h markedly increased the activity of lactate dehydrogenase (LDH) in cultured endothelial cell. Incubation ofendothelial cells with ox-LDL (100 μg/ml) or LPC (5.0 μg/ml) for 48 h significantly increased the expression of PRMT I, and levels of MDA and ADMA, and decreased the concentration of nitrite/nitrate, the expression of eNOS and the activity of DDAH. Probucol significantly decreased the level of ADMA, concomitantly with reduction of PRMT I expression and elevation of DDAH activity and up-regulation of eNOS expression.
Conclusion
In summary, the present results suggest that the protective effect of probucol on endothelium is related to reduction of ADMA concentration by inhibition of PRMT I expression and enhancement of DDAH activity.
Similar content being viewed by others
References
Boger RH, Bode-Boger SM, Szuba A, et al. Asymmetric dimethylarginine (ADMA): a novel risk factor for endothelial dysfunction: its role in hypercholesterolemia. Circulation 1998;98:1842–7.
Yu XJ, Li YJ, Xiong Y. Increase of an endogenous inhibitor of nitric oxide synthesis in serum of high cholesterol fed rabbits. Life Sci 1994;54:753–8.
Bode-Böger SM, Böger RH, Kienke S, et al. Elevated l-arginine/dimethylarginine ratio contributes to enhanced systemic NO production by dietary l-arginine in hypercholesterolemic rabbits. Biochem Biophys Res Commun 1996;219:598–603.
Ito A, Tsao PS, Adimoolam S, et al. Novel mechanism for endothelial dysfunction: dysregulation of dimethylarginine dimethylaminohydrolase. Circulation 1999;99:3092–5.
Scott HS, Antonarakis SE, Lalioti MD, et al. Identification and characterization of two putative human arginine methyltransferases (HRMT1L1 and HRMT1L2). Genomics 1998;48:330–40.
Boisvert FM, Cote J, Boulanger MC, et al. A proteomic analysis of arginine-methylated protein complexes. Mol Cell Proteomics 2003;2:1319–30.
Leiper JM, Maria JS, Chubb A, et al. Identification of two human dimethylarginine dimethylaminohydrolases with distinct tissue distributions and homology with microbial arginine deaminases. Biochem J 1999;99:3092–95.
Boger RH, Sydow K, Borlak J, et al. LDL cholesterol upregulates synthesis of asymmetrical dimethylarginine in human endothelial cells: involvement of S-adenosylmethionine-dependent methyltransferases. Circ Res 2000;87:99–105.
Jiang JL, Li NS, Li YJ, et al. Probucol preserves endothelial function by reduction of the endogenous nitric oxide synthase inhibitor level. Br J Pharmacol 2002;135:1175–82.
Selley ML. Homocysteine increases the production of asymmetric dimethylarginine in cultured neurons. J Neurosci Res 2004;77:90–3.
Havel RJ, Eder HA, Bragdon JH. The distribution and chemical composition of ultra-centrifugally separated lipoproteins in human serum. J Clin Invest 1995;34:1345–53.
Ohkawa H, Ohishi N, Yagi K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem 1979;95:351–8.
Bessman JD, Gardner FH. Platelet size in thrombocytopenia due to sepsis. Surg Gynecol Obstet 1983;156:177–80.
Feng Q, Lu X, Jones DL, et al. Increased inducible nitric oxide synthase expression contributes to myocardial dysfunction and higher mortality after myocardial infarction in mice. Circulation 2001;104:700–4.
Chen BM, Xia LW, Zhao RQ. Determination of NG, NG-dimethylarginine in human plasma by high-performance liquid chromatography. J Chromatogr B Biomed Sci Appl 1997;692:467–71.
Lin KY, Ito A, Asagami T, et al. Impaired nitric oxide synthase pathway in diabetes mellitus: role of asymmetric dimethylarginine and dimethylarginine dimethylaminohydrolase. Circulation 2002;106:987–92.
Vallance P, Leone A, Calver A, et al. Accumulation of an endogenous inhibitor of NO synthesis in chronic renal failure. Lancet 1992;339:572–5.
Kurose I, Wolf R, Grisham MB, et al. Effects of an endogenous inhibitor of nitric oxide synthesis on postcapillary venules. Am J Physiol 1995;268:H2224–31.
Yu XJ, Li YJ, Xiong Y. Increase of an endogenous inhibitor of nitric oxide synthesis in serum of high cholesterol fed rabbits. Life Sci 1994;54:753–8.
Usui M, Matsuoka H, Miyazaki H, et al. Increased endogenous nitric oxide synthase inhibitor in patients with congestive heart failure. Life Sci 1998;62:2425–30.
Boger RH, Bode-Boger SM, Tsao PS, et al. An endogenous inhibitor of nitric oxide synthase regulates endothelial adhesiveness for monocytes. J Am Coll Cardiol 2000;3:2287–95.
Chan JR, Boger RH, Bode-Boger SM, et al. Asymmetric dimethylarginine increases mononuclear cell adhesiveness in hypercholesterolemic humans. Arterioscler Thromb Vasc Biol 2000;20:1040–6.
Osanai T, Saitoh M, Sasaki S, et al. Effect of shear stress on asymmetric dimethylarginine release from vascular endothelial cells. Hypertension 2003;42:985–90.
Johnson BA, Najbauer J, Aswad DW. Accumulation of substrates for protein l-isoaspartyl methyltransferase in adenosine dialdehyde-treated PC12 cells. J Biol Chem 1993;268:6174–81.
Hanzelka BL, Greenberg EP. Quorum sensing in Vibrio fischeri: evidence that S-adenosylmethionine is the amino acid substrate for autoinducer synthesis. J Bacteriol 1996;178:5291–4.
Najbauer J, Johnson BA, Aswad DW. Analysis of stable protein methylation in cultured cells. Arch Biochem Biophys 1992;293:85–92.
Stuhlinger MC, Tsao PS, Her JH, et al. Homocysteine impairs the nitric oxide synthase pathway: role of asymmetrical dimethlarginine. Circulation 2000;104:2569–75.
Simon BC, Handenschild CC, Cohen RA. Preservation of endothelium-dependent relaxation in atherosclerotic rabbit aorta by probucol. J Cardiovasc Pharmacol 1993;21:893–901.
Liu GX, Ou DM, Li LX, et al. Probucol inhibits oxidized-low density lipoprotein-induced adhesion of monocytes to endothelial cells in vitro. Acta Pharmacol Sin 2002;23:516–22.
Kielstein JT, Impraim B, Simmel S, et al. Cardiovascular effects of systemic nitric oxide synthase inhibition with asymmetrical dimethylarginine in humans. Circulation 2004;109:172–7.
Pluta RM, Jung CS, Harvey-White J, et al. In vitro and in vivo effects of probucol on hydrolysis of asymmetric dimethyl l-arginine and vasospasm in primates. J Neurosurg 2005;103:731–8.
Tagawa T, Urabe Y, Kimura Y, et al. Long-term treatment with probucol improves endothelial function in patients with coronary artery disease. Hypertens Res 2004;27:311–8.
Fleming I, Mohamed A, Galle J, et al. Oxidized low-density lipoprotein increases superoxide production by endothelial nitric oxide synthase by inhibiting PKCalpha. Cardiovasc Res 2005;65:897–906.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jiang, JL., Zhang, XH., Li, NS. et al. Probucol Decreases Asymmetrical Dimethylarginine Level by Alternation of Protein Arginine Methyltransferase I and Dimethylarginine Dimethylaminohydrolase Activity. Cardiovasc Drugs Ther 20, 281–294 (2006). https://doi.org/10.1007/s10557-006-9065-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10557-006-9065-1