Abstract
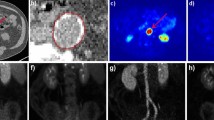
The objective was to dynamically monitor the progression of atherosclerotic plaques in ApoE−/− mice with 18F–NaF PET imaging. The ApoE−/− mice were used to develop atherosclerosis models, and the C57BL/6 J mice were used as control. 18F–NaF PET was performed when the mice were 12, 20, and 30 weeks of age. Serum lipids and lipoproteins profiles, inflammatory cytokines, and calcification factors were tested by ELISA. The lipid distribution, morphology, and calcification of plaque were evaluated by Oil Red O, HE, and alizarin red staining. The correlation between imaging and the extent of calcification was analyzed by Pearson correlation analysis. The uptake of 18F–NaF in the aorta was gradually increased with each weekly extension. Compared with the ApoE−/− mice at the age of 12 weeks and 20 weeks, the levels of lipoprotein, inflammatory cytokines, and calcification factors were higher at 30 weeks. In Oil Red O, HE, and alizarin red staining, the extent of the lipid area and calcification increased with time. The correlation analysis showed that the uptake of 18F–NaF in the aorta was related to the extent of calcification. 18F–NaF may dynamically monitor the progression of atherosclerotic plaques and ongoing microcalcification formation.






Similar content being viewed by others
Abbreviations
- PET:
-
Positron Emission Tomography
- ELISA:
-
Enzyme-linked Immunosorbent Assay
- TC:
-
Total Cholesterol
- TG:
-
Total Triglyceride
- HDL:
-
High Density Lipoprotein
- LDL:
-
Low Density Lipoprotein
- IL-1β:
-
Interleukin-1β
- TNF-α:
-
Tumor Necrosis Factor-α
- OCN:
-
Osteocalcin
- BMP:
-
Bone Morphogenetic Protein
- HE:
-
Hematoxylin–Eosin
- CA:
-
Calcium Staining Area
- TA:
-
Total Area
References
Vengrenyuk Y, Carlier S, Xanthos S, Cardoso L, Ganatos P, Virmani R et al (2006) A hypothesis for vulnerable plaque rupture due to stress-induced debonding around cellular microcalcifications in thin fibrous caps. Proc Natl Acad Sci USA 103:14678–14683
Irkle A, Vesey AT, Lewis DY, Skepper JN, Bird JLE, Dweck MR et al (2015) Identifying active vascular microcalcification by 18F–sodium fluoride positron emission tomography. Nat Commun 6:7495
Joshi NV, Vesey A, Newby DE, Dweck MR (2014) Will 18F–sodium fluoride PET-CT imaging be the magic bullet for identifying vulnerable coronary atherosclerotic plaques? Curr Cardiol Rep. https://doi.org/10.1007/s11886-014-0521-4
Ritman EL (2007) Small-animal CT–its difference from, and impact on, clinical CT. Nucl Instrum Methods Phys Res A 580:968–970
Fiz F, Morbelli S, Piccardo A, Bauckneht M, Ferrarazzo G, Pestarino E et al (2015) 18F–NaF uptake by Atherosclerotic plaque on PET/CT imaging: inverse correlation between calcification density and mineral metabolic activity. J Nucl Med 56:1019–1023
Joshi NV, Vesey AT, Williams MC, Shah ASV, Calvert PA, Craighead FHM et al (2014) 18F–fluoride positron emission tomography for identification of ruptured and high-risk coronary atherosclerotic plaques: a prospective clinical trial. Lancet 383:705–713
Dweck MR, Jenkins WSA, Vesey AT, Pringle MAH, Chin CWL, Malley TS et al (2014) 18F–sodium fluoride uptake is a marker of active calcification and disease progression in patients with aortic stenosis. Circ Cardiovasc Imag 7:371–378
Pugliese G, Iacobini C, Fantauzzi CB, Menini S (2015) The dark and bright side of atherosclerotic calcification. Atherosclerosis 238:220–230
Hop H, de Boer SA, Reijrink M et al (2019) 18F–sodium fluoride positron emission tomography assessed microcalcifications in culprit and non-culprit human carotid plaques. J Nucl Cardiol 26:1064–1075
Blake GM, Park-Holohan SJ, Cook GJ, Fogelman I (2001) Quantitative studies of bone with the use of 18F–fluoride and 99mTc-methylene diphosphonate. Semin Nucl Med 31:28–49
Wootton R, Dore C (1986) The single-passage extraction of 18F in rabbit bone. Clin Phys Physiol Meas 7:333–343
Janssen T, Bannas P, Herrmann J, Veldhoen S, Busch JD, Treszl A et al (2013) Association of linear 18F–sodium fluoride accumulation in femoral arteries as a measure of diffuse calcification with cardiovascular risk factors: a PET/CT study. J Nucl Cardiol 20:569–577
New SEP, Aikawa E (2011) Molecular imaging insights into early inflammatory stages of arterial and aortic valve calcification. Circ Res 108:1381–1391
Nadra I (2005) Proinflammatory activation of macrophages by basic calcium phosphate crystals via protein kinase C and MAP kinase pathways: a vicious cycle of inflammation and arterial calcification? Circ Res 96:1248–1256
Aikawa E, Nahrendorf M, Figueiredo JL, Swirski FK, Shtatland T, Kohler RH et al (2007) Osteogenesis associates with inflammation in early-stage atherosclerosis evaluated by molecular imaging in vivo. Circulation 116:2841–2850
Dweck MR, Jones C, Joshi NV, Fletcher AM, White HRA et al (2012) Assessment of valvular calcification and inflammation by positron emission tomography in patients with aortic stenosis. Circulation 125:76–U424
Dweck MR, Chow MWL, Joshi NV, Williams MC, Alison CJ, Fletcher M et al (2012) Coronary arterial 18F–sodium fluoride uptake. J Am Coll Cardiol 59:1539–1548
Derlin T, Toth Z, Papp L, Wisotzki C, Christian IA, Habermann R et al (2011) Correlation of inflammation assessed by F-18–FDG PET, active mineral deposition assessed by F-18–Fluoride PET, and vascular calcification in Atherosclerotic plaque: a dual-tracer PET/CT study. J Nucl Med 52:1020–1027
Voight BF, Peloso GM, Orho-Melander M, Frikke-Schmidt R, Barbalic M, Jensen MK et al (2012) Plasma HDL cholesterol and risk of myocardial infarction: a mendelian randomisation study. Lancet 380:572–580
Ikenaga M, Higaki Y, Saku K, Uehara Y (2016) High-density lipoprotein mimetics: a therapeutic tool for Atherosclerotic diseases. J Atheroscler Thromb 23:385–394
Acknowledgements
This study was funded by the National Nature Science Foundation of China (81901796, 81671735, 81871407), and Shanghai Municipal Key Clinical Specialty (shslczdzk 03401), and the Scientific Research Foundation for Young Doctors of Zhongshan Hospital, Fudan University (2018ZSQN025). No potential conflicts of interest relevant to this article exist.
Author information
Authors and Affiliations
Contributions
YH and PCH participated in the experimental and data analysis. YH wrote the manuscript. BXH, and WJC dealt with the aortas. YH, DFC and HCS designed and controlled the quality of study. All authors were involved in the study and approved its final version.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Research using animal consents
The animal research protocol was approved by the medical ethics committee of Zhongshan Hospital, Fudan University. All experiments were performed following the relevant guidelines and regulations of Fudan University.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hu, Y., Hu, P., Hu, B. et al. Dynamic monitoring of active calcification in atherosclerosis by 18F–NaF PET imaging. Int J Cardiovasc Imaging 37, 731–739 (2021). https://doi.org/10.1007/s10554-020-02019-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10554-020-02019-9