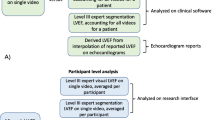
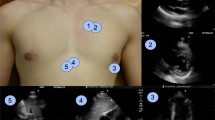
Abstract
We developed a machine learning model for efficient analysis of echocardiographic image quality in hospitalized patients. This study applied a machine learning model for automated transthoracic echo (TTE) image quality scoring in three inpatient groups. Our objectives were: (1) Assess the feasibility of a machine learning model for echo image quality analysis, (2) Establish the comprehensiveness of real-world TTE reporting by clinical group, and (3) Determine the relationship between machine learning image quality and comprehensiveness of TTE reporting. A machine learning model was developed and applied to TTEs from three matched cohorts for image quality of nine standard views. Case TTEs were comprehensive studies in mechanically ventilated patients between 01/01/2010 and 12/31/2015. For each case TTE, there were two matched spontaneously breathing controls (Control 1: Inpatients scanned in the lab and Control 2: Portable studies). We report the overall mean maximum and view specific quality scores for each TTE. The comprehensiveness of an echo report was calculated as the documented proportion of 12 standard parameters. An inverse probability weighted regression model was fit to determine the relationship between machine learning quality score and the completeness of a TTE report. 175 mechanically ventilated TTEs were included with 350 non-intubated samples (175 Control 1: Lab and 175 Control 2: Portable). In total, the machine learning model analyzed 14,086 echo video clips for quality. The overall accuracy of the model with regard to the expert ground truth for the view classification was 87.0%. The overall mean maximum quality score was lower for mechanically ventilated TTEs (0.55 [95% CI 0.54, 0.56]) versus 0.61 (95% CI 0.59, 0.62) for Control 1: Lab and 0.64 (95% CI 0.63, 0.66) for Control 2: Portable; p = 0.002. Furthermore, mechanically ventilated TTE reports were the least comprehensive, with fewer reported parameters. The regression model demonstrated the correlation of echo image quality and completeness of TTE reporting regardless of the clinical group. Mechanically ventilated TTEs were of inferior quality and clinical utility compared to spontaneously breathing controls and machine learning derived image quality correlates with completeness of TTE reporting regardless of the clinical group.





Similar content being viewed by others
Data availability
The data will not be published publicly but can be made available for facilitation of review.
Code availability
The code will not be published publicly.
References
Abdi A, Luong C, Tsang T, Jue J, Gin K, Yeung D, Hawley D, Rohling R, Abolmaesumi P (2017) Quality assessment of echocardiographic cine using recurrent neural networks: feasibility on five standard view planes. Paper presented at the Medical Image Computing and Computer-Assisted Intervention—MICCAI 2017, Lecture Notes in Computer Science, Springer, Cham
Abdi AH, Luong C, Tsang T, Allan G, Nouranian S, Jue J, Hawley D, Fleming S, Gin K, Swift J, Rohling R, Abolmaesumi P (2017) Automatic quality assessment of echocardiograms using convolutional neural networks: feasibility on the apical four-chamber view. IEEE Trans Med Imaging 36(6):1221–1230. https://doi.org/10.1109/TMI.2017.2690836
Narula S, Shameer K, Salem Omar AM, Dudley JT, Sengupta PP (2016) Machine-learning algorithms to automate morphological and functional assessments in 2D echocardiography. J Am Coll Cardiol 68(21):2287–2295. https://doi.org/10.1016/j.jacc.2016.08.062
Madani A, Arnaout R, Mo M, Arnaout R (2018) Fast and accurate view classification of echocardiograms using deep learning. npj Digital Med. https://doi.org/10.1038/s41746-017-0013-1
Sengupta PP, Huang YM, Bansal M, Ashrafi A, Fisher M, Shameer K, Gall W, Dudley JT (2016) Cognitive machine-learning algorithm for cardiac imaging: a pilot study for differentiating constrictive pericarditis from restrictive cardiomyopathy. Circ Cardiovasc Imaging. https://doi.org/10.1161/CIRCIMAGING.115.004330
Asl BM, Setarehdan SK, Mohebbi M (2008) Support vector machine-based arrhythmia classification using reduced features of heart rate variability signal. Artif Intell Med 44(1):51–64. https://doi.org/10.1016/j.artmed.2008.04.007
Katz DH, Deo RC, Aguilar FG, Selvaraj S, Martinez EE, Beussink-Nelson L, Kim KA, Peng J, Irvin MR, Tiwari H, Rao DC, Arnett DK, Shah SJ (2017) Phenomapping for the identification of hypertensive patients with the myocardial substrate for heart failure with preserved ejection fraction. J Cardiovasc Transl Res 10(3):275–284. https://doi.org/10.1007/s12265-017-9739-z
Carneiro G, Nascimento JC, Freitas A (2012) The segmentation of the left ventricle of the heart from ultrasound data using deep learning architectures and derivative-based search methods. IEEE Trans Image Process 21(3):968–982. https://doi.org/10.1109/TIP.2011.2169273
Smistad E, Østvik A, Haugen BO, L⊘vstakken L (2017) 2D left ventricle segmentation using deep learning. In: 2017 IEEE International Ultrasonics Symposium (IUS), 6–9 September, 2017, pp. 1–4
Khamis H, Zurakhov G, Azar V, Raz A, Friedman Z, Adam D (2017) Automatic apical view classification of echocardiograms using a discriminative learning dictionary. Med Image Anal 36:15–21. https://doi.org/10.1016/j.media.2016.10.007
Zhang J, Gajjala S, Agrawal P, Tison GH, Hallock LA, Beussink-Nelson L, Lassen MH, Fan E, Aras MA, Jordan C, Fleischmann KE, Melisko M, Qasim A, Shah SJ, Bajcsy R, Deo RC (2018) Fully automated echocardiogram interpretation in clinical practice. Circulation 138(16):1623–1635. https://doi.org/10.1161/CIRCULATIONAHA.118.034338
Lang RM, Badano LP, Mor-Avi V, Afilalo J, Armstrong A, Ernande L, Flachskampf FA, Foster E, Goldstein SA, Kuznetsova T, Lancellotti P, Muraru D, Picard MH, Rietzschel ER, Rudski L, Spencer KT, Tsang W, Voigt JU (2015) Recommendations for cardiac chamber quantification by echocardiography in adults: an update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J Am Soc Echocardiogr 28(1):1–39.e14. https://doi.org/10.1016/j.echo.2014.10.003
Schiller NB, Shah PM, Crawford M, DeMaria A, Devereux R, Feigenbaum H, Gutgesell H, Reichek N, Sahn D, Schnittger I (1989) Recommendations for quantitation of the left ventricle by two-dimensional echocardiography. American Society of Echocardiography Committee on Standards, Subcommittee on Quantitation of Two-Dimensional Echocardiograms. J Am Soc Echocardiogr 2(5):358–367
Tighe DA, Rosetti M, Vinch CS, Chandok D, Muldoon D, Wiggin B, Dahlberg ST, Aurigemma GP (2007) Influence of image quality on the accuracy of real time three-dimensional echocardiography to measure left ventricular volumes in unselected patients: a comparison with gated-SPECT imaging. Echocardiography 24(10):1073–1080. https://doi.org/10.1111/j.1540-8175.2007.00525.x
Hoffmann R, Lethen H, Marwick T, Arnese M, Fioretti P, Pingitore A, Picano E, Buck T, Erbel R, Flachskampf FA, Hanrath P (1996) Analysis of interinstitutional observer agreement in interpretation of dobutamine stress echocardiograms. J Am Coll Cardiol 27(2):330–336
Kusunose K, Shibayama K, Iwano H, Izumo M, Kagiyama N, Kurosawa K, Mihara H, Oe H, Onishi T, Ota M, Sasaki S, Shiina Y, Tsuruta H, Tanaka H, Investigators J (2018) Reduced variability of visual left ventricular ejection fraction assessment with reference images: the Japanese Association of Young Echocardiography Fellows multicenter study. J Cardiol. https://doi.org/10.1016/j.jjcc.2018.01.007
Nagata Y, Kado Y, Onoue T, Otani K, Nakazono A, Otsuji Y, Takeuchi M (2018) Impact of image quality on reliability of the measurements of left ventricular systolic function and global longitudinal strain in 2D echocardiography. Echo Res Pract 5(1):27–39. https://doi.org/10.1530/ERP-17-0047
Medvedofsky D, Mor-Avi V, Byku I, Singh A, Weinert L, Yamat M, Kruse E, Ciszek B, Nelson A, Otani K, Takeuchi M, Lang RM (2017) Three-dimensional echocardiographic automated quantification of left heart chamber volumes using an adaptive analytics algorithm: feasibility and impact of image quality in nonselected patients. J Am Soc Echocardiogr 30(9):879–885. https://doi.org/10.1016/j.echo.2017.05.018
Van Woudenberg N, Liao Z, Abdi AH, Girgis H, Luong C, Vaseli H, Behnami D, Zhang H, Gin K, Rohling R, Tsang T, Abolmaesumi P (2018) Quantitative echocardiography: real-time quality estimation and view classification implemented on a mobile android device. In: Stoyanov D, et al. (eds) Simulation, image processing, and ultrasound systems for assisted diagnosis and navigation. Springer, Cham, pp 74–81
Huang G, Liu Z, van der Maaten L, Weinberger KQ (2017) Densely connected convolutional networks. In Proceedings of the IEEE Conference on Computer Vision and Pattern Recognition, vol 1
Ioffe S, Szegedy C (2015) Batch normalization: accelerating deep network training by reducing internal covariate shift. ICML 37:448–456
Hochreiter S, Schmidhuber J (1997) Long short-term memory. Neural Comput 9:1735–1780
Levitov A, Frankel HL, Blaivas M, Kirkpatrick AW, Su E, Evans D, Summerfield DT, Slonim A, Breitkreutz R, Price S, McLaughlin M, Marik PE, Elbarbary M (2016) Guidelines for the appropriate use of bedside general and cardiac ultrasonography in the evaluation of critically ill patients-part ii: cardiac ultrasonography. Crit Care Med 44(6):1206–1227. https://doi.org/10.1097/ccm.0000000000001847
Lumley T, Scott AJ (2013) Two-sample rank tests under complex sampling. Biometrika 100(4):831–842
Lumley T, Scott A (2014) Tests for regression models fitted to survey data. Aust N Z J Stat 56(1):1–14
Gaudet J, Waechter J, McLaughlin K, Ferland A, Godinez T, Bands C, Boucher P, Lockyer J (2016) Focused critical care echocardiography: development and evaluation of an image acquisition assessment tool. Crit Care Med 44(6):e329–335. https://doi.org/10.1097/ccm.0000000000001620
Hensel KO, Roskopf M, Wilke L, Heusch A (2018) Intraobserver and interobserver reproducibility of M-mode and B-mode acquired mitral annular plane systolic excursion (MAPSE) and its dependency on echocardiographic image quality in children. PLoS ONE 13(5):e0196614. https://doi.org/10.1371/journal.pone.0196614
Thaden JJ, Tsang MY, Ayoub C, Padang R, Nkomo VT, Tucker SF, Cassidy CS, Bremer M, Kane GC, Pellikka PA (2017) Association between echocardiography laboratory accreditation and the quality of imaging and reporting for valvular heart disease. Circul Cardiovasc Imaging. https://doi.org/10.1161/CIRCIMAGING.117.006140
Kitzman DW (2000) Normal age-related changes in the heart: relevance to echocardiography in the elderly. Am J Geriatr Cardiol 9(6):311–320
Funding
Canadian Institutes of Health Research (CIHR). Natural Sciences and Engineering Research Council of Canada (NSERC).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors do not have any relevant conflicts of interest to disclose.
Ethical approval
This study was reviewed and approved by the University of British Columbia Clinical Review Ethics Board.
Consent to participate
This study was a retrospective review of anonymized images, therefore individual consent was not obtained.
Consent for publication
The authors of this study consent to publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Luong, C., Liao, Z., Abdi, A. et al. Automated estimation of echocardiogram image quality in hospitalized patients. Int J Cardiovasc Imaging 37, 229–239 (2021). https://doi.org/10.1007/s10554-020-01981-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10554-020-01981-8