Abstract
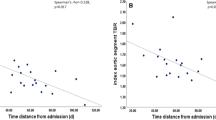
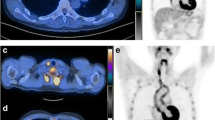
Aim of this study was to investigate the relationship between 18F-fluorodeoxyglucose (FDG) positron emission tomography-computed tomography (PET-CT) findings and serological biomarkers of inflammation and the related discriminant value of unfavourable outcome during follow-up in patients with acute aortic syndromes (AAS). Sixty patients with AAS underwent PET-CT imaging during the hospitalization along with measurement of C-reactive protein (CRP) and D-dimer (D-d) serum levels. An aortic wall pathology was considered PET-positive by a maximum standardized uptake value (SUVmax) >2.5. A combined endpoint of major adverse events (MAE) including aorta-related mortality, disease progression and re-intervention was used to compare patient subgroups at 3-year follow-up. PET-CT detected an elevated FDG uptake within the aortic wall in 25 (41.7 %) patients. PET-positive patients showed significantly increased CRP levels (10.0 ± 6.6 mg/dL) and tended to higher D-d levels (5.1 ± 3.9 mg/L), compared to PET-negative patients (5.8 ± 6.1 mg/dL and 3.1 ± 4.7 mg/L respectively; P = 0.048, P = 0.19). At 3-year follow-up, all-cause mortality and MAE were higher in the PET-positive (21.7 and 47.8 % respectively) than PET-negative group (0.0 and 13.3 % respectively; P = 0.012, P = 0.006). On Kaplan–Meier analysis, PET-positive patients were at higher risk of MAE (P = 0.031). This tendency was more evident by combining PET results with D-d levels at a cutoff value of 4.8 mg/L (P < 0.001). In patients with AAS, a pathological glucose uptake in aortic wall lesions by PET-CT was associated with high CRP levels and increased mortality and MAE at 3-year follow-up. The combination of PET results with D-d levels had the best discriminant value of MAE.




Similar content being viewed by others
References
Gallamini A, Zwarthoed C, Borra A (2014) Positron emission tomography (PET) in oncology. Cancers 6:1821–1889
Strobl FF, Rominger A, Wolpers S et al (2013) Impact of cardiovascular risk factors on vessel wall inflammation and calcified plaque burden differs across vascular beds: a PET-CT study. Int J Cardiovasc Imaging 29:1899–1908
Hetterich H, Rominger A, Walter L et al (2015) Natural history of atherosclerotic disease progression as assessed by 18F-FDG PET/CT. Int J Cardiovasc Imaging. doi:10.1007/s10554-015-0660-8
Goldstein SA, Evangelista A, Abbara S et al (2015) Multimodality imaging of diseases of the thoracic aorta in adults: from the american society of echocardiography and the European association of cardiovascular imaging: endorsed by the society of cardiovascular computed tomography and society for cardiovascular magnetic resonance. J Am Soc Echocardiogr 28:119–182
Erbel R, Aboyans V, Boileau C et al (2014) 2014 ESC Guidelines on the diagnosis and treatment of aortic diseases: document covering acute and chronic aortic diseases of the thoracic and abdominal aorta of the adult. Eur Heart J 35:2873–2926
Eggebrecht H, Naber CK, Bruch C et al (2004) Value of plasma fibrin D-dimers for detection of acute aortic dissection. J Am Coll Cardiol 44:804–809
Ohlmann P, Faure A, Morel O et al (2006) Diagnostic and prognostic value of circulating D-dimers in patients with acute aortic dissection. Crit Care Med 34:1358–1364
Tian L, Fan X, Zhu J et al (2014) Plasma D-dimer and in-hospital mortality in patients with Stanford type A acute aortic dissection. Blood Coagul Fibrinolysis 25:161–166
Wen D, Du X, Dong JZ et al (2013) Value of D-dimer and C reactive protein in predicting in-hospital death in acute aortic dissection. Heart 99:1192–1197
Kuehl H, Eggebrecht H, Boes T et al (2008) Detection of inflammation in patients with acute aortic syndrome: comparison of FDG-PET/CT imaging and serological markers of inflammation. Heart 94:1472–1477
Boellaard R, Delgado-Bolton R, Oyen WJ et al (2015) FDG PET/CT: EANM procedure guidelines for tumour imaging: version 2.0. Eur J Nucl Med Mol Imaging 42:328–354
Antoch G, Freudenberg LF, Stattaus J et al (2002) Whole-body positron emission tomography-CT: optimized CT using oral and IV contrast materials. Am J Roentgenol 179:1555–1560
Kobayashi Y, Ishii K, Oda K et al (2005) Aortic wall inflammation due to Takayasu arteritis imaged with 18F-FDG PET coregistered with enhanced CT. J Nucl Med 46:917–922
Huptas S, Mehta RH, Kühl H et al (2009) Aortic remodeling in type B aortic dissection: effects of endovascular stent-graft repair and medical treatment on true and false lumen volumes. J Endovasc Ther 16:28–38
Reeps C, Pelisek J, Bundschuh RA et al (2010) Imaging of acute and chronic aortic dissection by 18F-FDG PET/CT. J Nucl Med 51:686–691
Sakalihasan N, Nienaber CA, Hustinx R et al (2015) (Tissue PET) Vascular metabolic imaging and peripheral plasma biomarkers in the evolution of chronic aortic dissections. Eur Heart J Cardiovasc Imaging 6:626–633
Kato K, Nishio A, Kato N et al (2010) Uptake of 18F-FDG in acute aortic dissection: a determinant of unfavorable outcome. J Nucl Med 51:674–681
Shimizu K, Mitchell RN, Libby P (2006) Inflammation and cellular immune responses in abdominal aortic aneurysms. Arterioscler Thromb Vasc Biol 26:987–994
Reeps C, Essler M, Pelisek J et al (2008) Increased 18F-fluorodeoxyglucose uptake in abdominal aortic aneurysms in positron emission/computed tomography is associated with inflammation, aortic wall instability, and acute symptoms. J Vasc Surg 48:417–423
Reeps C, Bundschuh RA, Pellisek J et al (2013) Quantitative assessment of glucose metabolism in the vessel wall of abdominal aortic aneurysms: correlation with histology and role of partial volume correction. Int J Cardiovasc Imaging 29:505–512
Weber T, Rammer M, Auer J et al (2006) Plasma concentrations of D-dimer predict mortality in acute type A aortic dissection. Heart 92:836–837
Gorla R, Erbel R, Kahlert P et al (2015) Diagnostic role and prognostic implications of D-dimer in different classes of acute aortic syndromes. Eur Heart J Acute Cardiovasc Care. doi:10.1177/2048872615594500
Serino F, Abeni D, Galvagni E et al (2002) Noninvasive diagnosis of incomplete endovascular aneurysm repair: D-dimer assay to detect type I endoleaks and nonshrinking aneurysms. J Endovasc Ther 9:90–97
Acknowledgments
The authors would like to thank Dr. Luca Carannante for his technical support during the study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical standard
For this type of study formal consent is not required.
Rights and permissions
About this article
Cite this article
Gorla, R., Erbel, R., Kuehl, H. et al. Prognostic value of 18F-fluorodeoxyglucose PET-CT imaging in acute aortic syndromes: comparison with serological biomarkers of inflammation. Int J Cardiovasc Imaging 31, 1677–1685 (2015). https://doi.org/10.1007/s10554-015-0725-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10554-015-0725-8