Abstract
Purpose
The status of sentinel lymph nodes (SLN) is an important factor in determining the stage of breast cancer (BC) and the surgical procedure, and also a biomarker of the BC prognosis. This meta-analysis was performed to investigate the diagnostic value of contrast-enhanced ultrasound (CEUS) for SLN metastasis.
Methods
A systematic search was conducted for relevant articles published in the PubMed, Embase, Web of Science, OVID databases, and Cochrane Library from inception to March 2023. We calculated the sensitivity, specificity, positive and negative likelihood ratio (PLR; NLR), diagnostic odds ratio (DOR), and summary receiver operator characteristic (SROC) curve to evaluate the diagnostic efficacy of CEUS in SLN metastasis. Subgroup analysis was also performed to investigate potential sources of heterogeneity.
Results
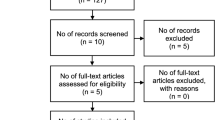
A total of 12 studies with 1525 patients were included in this meta-analysis. The overall pooled sensitivity and specificity of CEUS in the diagnosis of SLN metastasis were 0.91 (95% CI: 0.84–0.95) and 0.86 (95% CI: 0.78–0.92). The PLR, NLR, and DOR were 6.51 (95% CI: 4.09–10.36), 0.11 (95% CI: 0.07–0.18), and 59.43 (95% CI: 33.27–106.17), respectively, and the area under the SROC curve was determined to be 0.95 (95%CI: 0.92–0.96), all showing excellent diagnostic value. In the subgroup analysis, percutaneous CEUS was more sensitive than intravenous CEUS in the diagnosis of SLN metastases (0.92 versus 0.82, p < 0.05).
Conclusion
CEUS, especially percutaneous CEUS, is a reliable imaging technique for diagnosing SLN metastasis and providing important information in the stage management of breast cancer.




Similar content being viewed by others
Data availability
All data and material used in this project are publicly available in the databases listed in the manuscript.
References
Yang H, Pawitan Y, He W, Eriksson L, Holowko N, Hall P, Czene K (2019) Disease trajectories and mortality among women diagnosed with breast cancer. Breast Cancer Res 21:95. https://doi.org/10.1186/s13058-019-1181-5
Dhankhar R, Vyas SP, Jain AK, Arora S, Rath G, Goyal AK (2010) Advances in novel drug delivery strategies for breast cancer therapy. Artif Cells Blood Substit Immobil Biotechnol 38:230–249. https://doi.org/10.3109/10731199.2010.494578
Qiao J, Li J, Wang L, Guo X, Bian X, Lu Z (2021) Predictive risk factors for sentinel lymph node metastasis using preoperative contrast-enhanced ultrasound in early-stage breast cancer patients. Gland Surg 10:761–769. https://doi.org/10.21037/gs-20-867
Cserni G, Maguire A, Bianchi S, Ryska A, Kovács A (2022) Sentinel lymph node assessment in breast cancer-an update on current recommendations. Virchows Arch 480:95–107. https://doi.org/10.1007/s00428-021-03128-z
Lyman GH, Somerfield MR, Bosserman LD, Perkins CL, Weaver DL, Giuliano AE (2017) Sentinel lymph node biopsy for patients with early-stage breast cancer: american society of clinical oncology clinical practice guideline update. J Clin Oncol 35:561–564. https://doi.org/10.1200/jco.2016.71.0947
Magnoni F, Galimberti V, Corso G, Intra M, Sacchini V, Veronesi P (2020) Axillary surgery in breast cancer: An updated historical perspective. Semin Oncol 47:341–352. https://doi.org/10.1053/j.seminoncol.2020.09.001
Qiu SQ, Zhang GJ, Jansen L, de Vries J, Schröder CP, de Vries EGE, van Dam GM (2018) Evolution in sentinel lymph node biopsy in breast cancer. Crit Rev Oncol Hematol 123:83–94. https://doi.org/10.1016/j.critrevonc.2017.09.010
Cui Q, Dai L, Li J, Xue J (2020) Accuracy of CEUS-guided sentinel lymph node biopsy in early-stage breast cancer: a study review and meta-analysis. World J Surg Oncol 18:112. https://doi.org/10.1186/s12957-020-01890-z
Du J, Wang L, Wan CF, Hua J, Fang H, Chen J, Li FH (2012) Differentiating benign from malignant solid breast lesions: combined utility of conventional ultrasound and contrast-enhanced ultrasound in comparison with magnetic resonance imaging. Eur J Radiol 81:3890–3899. https://doi.org/10.1016/j.ejrad.2012.09.004
Hwang M, Barnewolt CE, Jüngert J, Prada F, Sridharan A, Didier RA (2021) Contrast-enhanced ultrasound of the pediatric brain. Pediatr Radiol 51:2270–2283. https://doi.org/10.1007/s00247-021-04974-4
Zhang YX, Wang XM, Kang S, Li X, Geng J (2015) Contrast-enhanced ultrasonography in qualitative diagnosis of sentinel lymph node metastasis in breast cancer: A meta-analysis. J Cancer Res Ther 11:697–703. https://doi.org/10.4103/0973-1482.146129
Fowler JC, Solanki CK, Guenther I, Barber R, Miller F, Bobrow L, Ravichandran D, Lawrence D, Ballinger JR, Douglas-Jones A, Purushotham AD, Peters AM (2009) A pilot study of dual-isotope lymphoscintigraphy for breast sentinel node biopsy comparing intradermal and intraparenchymal injection. Eur J Surg Oncol 35:1041–1047. https://doi.org/10.1016/j.ejso.2009.02.018
Luo J, Feng L, Zhou Q, Chen Q, Liu J, Wu C, Luo J, Chen J, Wu H, Deng W (2021) The value of contrast-enhanced ultrasound in determining the location of sentinel lymph nodes in breast cancer. Cancer Imaging 21:28. https://doi.org/10.1186/s40644-021-00397-4
Niu Z, Xiao M, Ma L, Qin J, Li W, Zhang J, Zhu Q, Jiang Y (2022) The value of contrast-enhanced ultrasound enhancement patterns for the diagnosis of sentinel lymph node status in breast cancer: systematic review and meta-analysis. Quant Imaging Med Surg 12:936–948. https://doi.org/10.21037/qims-21-416
Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, Shamseer L, Tetzlaff JM, Akl EA, Brennan SE, Chou R, Glanville J, Grimshaw JM, Hróbjartsson A, Lalu MM, Li T, Loder EW, Mayo-Wilson E, McDonald S, McGuinness LA, Stewart LA, Thomas J, Tricco AC, Welch VA, Whiting P, Moher D (2021) The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 372:n71. https://doi.org/10.1136/bmj.n71
Whiting PF, Rutjes AW, Westwood ME, Mallett S, Deeks JJ, Reitsma JB, Leeflang MM, Sterne JA, Bossuyt PM (2011) QUADAS-2: a revised tool for the quality assessment of diagnostic accuracy studies. Ann Intern Med 155:529–536. https://doi.org/10.7326/0003-4819-155-8-201110180-00009
Irwig L, Bossuyt P, Glasziou P, Gatsonis C, Lijmer J (2002) Designing studies to ensure that estimates of test accuracy are transferable. BMJ 324:669–671. https://doi.org/10.1136/bmj.324.7338.669
Higgins JP, Thompson SG (2002) Quantifying heterogeneity in a meta-analysis. Stat Med 21:1539–1558. https://doi.org/10.1002/sim.1186
Egger M, Davey Smith G, Schneider M, Minder C (1997) Bias in meta-analysis detected by a simple, graphical test. BMJ 315:629–634. https://doi.org/10.1136/bmj.315.7109.629
Zhuang L, Ming X, Liu J, Jia C, Jin Y, Wang J, Shi Q, Wu R, Jin L, Du L (2022) Comparison of lymphatic contrast-enhanced ultrasound and intravenous contrast-enhanced ultrasound in the preoperative diagnosis of axillary sentinel lymph node metastasis in patients with breast cancer. Br J Radiol 95:20210897. https://doi.org/10.1259/bjr.20210897
Zhu Y, Fan X, Yang D, Dong T, Jia Y, Nie F (2021) Contrast-enhanced ultrasound for precise sentinel lymph node biopsy in women with early breast cancer: a preliminary study. Diagnostics. https://doi.org/10.3390/diagnostics11112104
Xie F, Zhang D, Cheng L, Yu L, Yang L, Tong F, Liu H, Wang S, Wang S (2015) Intradermal microbubbles and contrast-enhanced ultrasound (CEUS) is a feasible approach for sentinel lymph node identification in early-stage breast cancer. World J Surg Oncol 13:319. https://doi.org/10.1186/s12957-015-0736-x
Sun Y, Cui L, Wang S, Shi T, Hao Y, Lei Y (2021) Comparative study of two contrast agents for intraoperative identification of sentinel lymph nodes in patients with early breast cancer. Gland Surg 10:1638–1645. https://doi.org/10.21037/gs-21-87
Hao Y, Sun Y, Lei Y, Zhao H, Cui L (2021) Percutaneous Sonazoid-enhanced ultrasonography combined with in vitro verification for detection and characterization of sentinel lymph nodes in early breast cancer. Eur Radiol 31:5894–5901. https://doi.org/10.1007/s00330-020-07639-2
Li J, Lu M, Cheng X, Hu Z, Li H, Wang H, Jiang J, Li T, Zhang Z, Zhao C, Ma Y, Tan B, Liu J, Yu Y (2019) How pre-operative sentinel lymph node contrast-enhanced ultrasound helps intra-operative sentinel lymph node biopsy in breast cancer: initial experience. Ultrasound Med Biol 45:1865–1873. https://doi.org/10.1016/j.ultrasmedbio.2019.04.006
Liu J, Liu X, He J, Gou B, Luo Y, Deng S, Wen H, Zhou L (2019) Percutaneous contrast-enhanced ultrasound for localization and diagnosis of sentinel lymph node in early breast cancer. Sci Rep 9:13545. https://doi.org/10.1038/s41598-019-49736-3
Zhao J, Zhang J, Zhu QL, Jiang YX, Sun Q, Zhou YD, Wang MQ, Meng ZL, Mao XX (2018) The value of contrast-enhanced ultrasound for sentinel lymph node identification and characterisation in pre-operative breast cancer patients: A prospective study. Eur Radiol 28:1654–1661. https://doi.org/10.1007/s00330-017-5089-0
Matsuzawa F, Einama T, Abe H, Suzuki T, Hamaguchi J, Kaga T, Sato M, Oomura M, Takata Y, Fujibe A, Takeda C, Tamura E, Taketomi A, Kyuno K (2015) Accurate diagnosis of axillary lymph node metastasis using contrast-enhanced ultrasonography with Sonazoid. Mol Clin Oncol 3:299–302. https://doi.org/10.3892/mco.2014.483
Niu Z, Gao Y, Xiao M, Mao F, Zhou Y, Zhu Q, Jiang Y (2023) Contrast-enhanced lymphatic US can improve the preoperative diagnostic performance for sentinel lymph nodes in early breast cancer. Eur Radiol 33:1593–1602. https://doi.org/10.1007/s00330-022-09139-x
Cykowska A, Marano L, D’Ignazio A, Marrelli D, Swierblewski M, Jaskiewicz J, Roviello F, Polom K (2020) New technologies in breast cancer sentinel lymph node biopsy; from the current gold standard to artificial intelligence. Surg Oncol 34:324–335. https://doi.org/10.1016/j.suronc.2020.06.005
Moher D, Liberati A, Tetzlaff J, Altman DG, the PRISMA Group (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med 6:e1000097. https://doi.org/10.1371/journal.pmed.1000097
Dębiński P, Dembowski J, Kowal P, Szydełko T, Kołodziej A, Małkiewicz B, Tupikowski K, Zdrojowy R (2013) The clinical significance of lymphangiogenesis in renal cell carcinoma. Med Sci Monit 19:606–611. https://doi.org/10.12659/msm.883981
Gkegkes ID, Iavazzo C (2015) Contrast enhanced ultrasound (CEU) using microbubbles for sentinel lymph node biopsy in breast cancer: a systematic review. Acta Chir Belg 115:212–218. https://doi.org/10.1080/00015458.2015.11681099
Cox K, Sever A, Jones S, Weeks J, Mills P, Devalia H, Fish D, Jones P (2013) Validation of a technique using microbubbles and contrast enhanced ultrasound (CEUS) to biopsy sentinel lymph nodes (SLN) in pre-operative breast cancer patients with a normal grey-scale axillary ultrasound. Eur J Surg Oncol 39:760–765. https://doi.org/10.1016/j.ejso.2013.03.026
Hwang M, Back SJ, Didier RA, Lorenz N, Morgan TA, Poznick L, Steffgen L, Sridharan A (2021) Pediatric contrast-enhanced ultrasound: optimization of techniques and dosing. Pediatr Radiol 51:2147–2160. https://doi.org/10.1007/s00247-020-04812-z
Greis C (2014) Technical aspects of contrast-enhanced ultrasound (CEUS) examinations: tips and tricks. Clin Hemorheol Microcirc 58:89–95. https://doi.org/10.3233/ch-141873
de Boniface J, Frisell J, Andersson Y, Bergkvist L, Ahlgren J, Rydén L, Olofsson Bagge R, Sund M, Johansson H, Lundstedt D (2017) Survival and axillary recurrence following sentinel node-positive breast cancer without completion axillary lymph node dissection: the randomized controlled SENOMAC trial. BMC Cancer 17:379. https://doi.org/10.1186/s12885-017-3361-y
Rubaltelli L, Khadivi Y, Tregnaghi A, Stramare R, Ferro F, Borsato S, Fiocco U, Adami F, Rossi CR (2004) Evaluation of lymph node perfusion using continuous mode harmonic ultrasonography with a second-generation contrast agent. J Ultrasound Med 23:829–836. https://doi.org/10.7863/jum.2004.23.6.829
Collins M, O’Donoghue C, Sun W, Zhou JM, Ma Z, Laronga C, Lee MC (2017) Use of axillary lymph node dissection (ALND) in patients with micrometastatic breast cancer. J Surg Res 215:55–59. https://doi.org/10.1016/j.jss.2017.03.039
Sato K, Tamaki K, Tsuda H, Kosuda S, Kusano S, Hiraide H, Mochizuki H (2004) Utility of axillary ultrasound examination to select breast cancer patients suited for optimal sentinel node biopsy. Am J Surg 187:679–683. https://doi.org/10.1016/j.amjsurg.2003.10.012
Mansel RE, Fallowfield L, Kissin M, Goyal A, Newcombe RG, Dixon JM, Yiangou C, Horgan K, Bundred N, Monypenny I, England D, Sibbering M, Abdullah TI, Barr L, Chetty U, Sinnett DH, Fleissig A, Clarke D, Ell PJ (2006) Randomized multicenter trial of sentinel node biopsy versus standard axillary treatment in operable breast cancer: the ALMANAC Trial. J Natl Cancer Inst 98:599–609. https://doi.org/10.1093/jnci/djj158
Chen X, He Y, Wang J, Huo L, Fan Z, Li J, Xie Y, Wang T, Ouyang T (2018) Feasibility of using negative ultrasonography results of axillary lymph nodes to predict sentinel lymph node metastasis in breast cancer patients. Cancer Med 7:3066–3072. https://doi.org/10.1002/cam4.1606
Funding
The work was supported by the Natural Science Foundation of Shandong Province [Grant Number ZR2021QH047] and the Clinical Science and Technology Innovation Development Program of Jinan [Grant Number 202134036].
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by XL, MW and QW. The first draft of the manuscript was written by XL and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, X., Wang, M., Wang, Q. et al. Diagnostic value of contrast-enhanced ultrasound for sentinel lymph node metastasis in breast cancer: an updated meta-analysis. Breast Cancer Res Treat 202, 221–231 (2023). https://doi.org/10.1007/s10549-023-07063-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-023-07063-2