Abstract
Background
Deleterious BRCA mutations confer a significant lifetime risk of breast cancer (BC) as well as contralateral BC (CBC) in patients who do not undergo prophylactic mastectomy. Prior reports have suggested that tamoxifen reduces the risk of CBC in BRCA mutation carriers. Whether aromatase inhibitors (AI) have the same effect is unknown.
Methods
This is a retrospective review of patients diagnosed with non-metastatic ER+ BC between 2004 and 2014 with known BRCA mutation status. Patients were followed from primary diagnosis until CBC diagnosis or death. Median follow-up was 11.5 years. Risk of CBC was evaluated as time to event.
Results
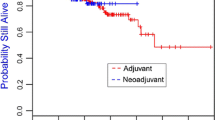
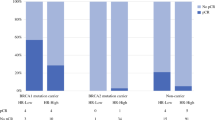
935 subjects were included in this analysis, with 53 BRCA1 mutation carriers, and 94 BRCA2 mutation carriers. Median age at diagnosis was 42.7 years. Seventy-two percent (676) received tamoxifen and 43% (405) received AI. A total of 66 CBCs occurred, of which 10% (15/147) occurred in BRCA mutation carriers vs 6.5% (51/788) in BRCA wild type. Multivariate analyses indicated that BRCA status and AI use were significantly associated with CBC risk. AI use resulted in a significant reduction in risk of CBC (HR 0.44, p = 0.004) regardless of the BRCA mutation status. Tamoxifen use was not associated with reduced risk of CBC.
Conclusions
This is the first report showing that AIs reduce the risk of CBC in BRCA mutation carriers. The potential role of AIs as chemoprevention should be validated in larger independent cohorts.








Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are not publicly available as the database is actively updated and is the list of active and prior patients receiving care at MD Anderson Cancer Center.
References
Ford D et al (1998) Genetic heterogeneity and penetrance analysis of the BRCA1 and BRCA2 genes in breast cancer families. The Breast Cancer Linkage Consortium. Am J Hum Genet 62(3):676–89
Metcalfe KA, Narod SA (2007) Breast cancer prevention in women with a BRCA1 or BRCA2 mutation. Open Med 1(3):e184–e190
Cortesi L, Rugo HS, Jackisch C (2021) An overview of PARP inhibitors for the treatment of breast cancer. Target Oncol 16(3):255–282
Kotsopoulos J (2018) Mutations and breast cancer prevention. Cancers (Basel) 10(12):524
Metcalfe K et al (2004) Contralateral breast cancer in BRCA1 and BRCA2 mutation carriers. J Clin Oncol 22(12):2328–2335
Verhoog LC et al (2000) Contralateral breast cancer risk is influenced by the age at onset in BRCA1-associated breast cancer. Br J Cancer 83(3):384–386
Pierce LJ et al (2006) Ten-year multi-institutional results of breast-conserving surgery and radiotherapy in BRCA1/2-associated stage I/II breast cancer. J Clin Oncol 24(16):2437–2443
Kuchenbaecker KB et al (2017) Risks of breast, ovarian, and contralateral breast cancer for BRCA1 and BRCA2 mutation carriers. JAMA 317(23):2402–2416
Rebbeck TR et al (2004) Bilateral prophylactic mastectomy reduces breast cancer risk in BRCA1 and BRCA2 mutation carriers: the PROSE Study Group. J Clin Oncol 22(6):1055–1062
NCC Guidelines Version 4.2021—Breast Cancer. 2021: NCCN.org.
Rebbeck TR et al (1999) Breast cancer risk after bilateral prophylactic oophorectomy in BRCA1 mutation carriers. J Natl Cancer Inst 91(17):1475–1479
Tung NM et al (2020) Management of hereditary breast cancer: American Society of Clinical Oncology, American Society for Radiation Oncology, and Society of Surgical Oncology guideline. J Clin Oncol 38(18):2080–2106
Campbell AM et al (2018) Chemoprevention in BRCA1 mutation carriers (CIBRAC): protocol for an open allocation crossover feasibility trial assessing mechanisms of chemoprevention with goserelin and anastrozole versus tamoxifen and acceptability of treatment. BMJ Open 8(12):e023115
Xu L et al (2015) Tamoxifen and risk of contralateral breast cancer among women with inherited mutations in BRCA1 and BRCA2: a meta-analysis. Breast Cancer 22(4):327–334
Phillips KA et al (2013) Tamoxifen and risk of contralateral breast cancer for BRCA1 and BRCA2 mutation carriers. J Clin Oncol 31(25):3091–3099
Gronwald J et al (2014) Duration of tamoxifen use and the risk of contralateral breast cancer in BRCA1 and BRCA2 mutation carriers. Breast Cancer Res Treat 146(2):421–427
Goss PE et al (2016) Extending aromatase-inhibitor adjuvant therapy to 10 years. N Engl J Med 375(3):209–219
Cuzick J et al (2010) Effect of anastrozole and tamoxifen as adjuvant treatment for early-stage breast cancer: 10-year analysis of the ATAC trial. Lancet Oncol 11(12):1135–1141
Gierach GL et al (2016) Association of adjuvant tamoxifen and aromatase inhibitor therapy with contralateral breast cancer risk among US women with breast cancer in a general community setting. JAMA Oncol. https://doi.org/10.1001/jamaoncol.2016.3340
Foulkes WD et al (2004) Estrogen receptor status in BRCA1- and BRCA2-related breast cancer: the influence of age, grade, and histological type. Clin Cancer Res 10(6):2029–2034
Honrado E, Benitez J, Palacios J (2004) The pathology of hereditary breast cancer. Hered Cancer Clin Pract 2(3):131–138
Kawai H et al (2002) Direct interaction between BRCA1 and the estrogen receptor regulates vascular endothelial growth factor (VEGF) transcription and secretion in breast cancer cells. Oncogene 21(50):7730–7739
Tung N et al (2010) Estrogen receptor positive breast cancers in BRCA1 mutation carriers: clinical risk factors and pathologic features. Breast Cancer Res 12(1):R12
Lips EH et al (2017) BRCA1-mutated estrogen receptor-positive breast cancer shows BRCAness, suggesting sensitivity to drugs targeting homologous recombination deficiency. Clin Cancer Res 23(5):1236–1241
Bane AL et al (2007) BRCA2 mutation-associated breast cancers exhibit a distinguishing phenotype based on morphology and molecular profiles from tissue microarrays. Am J Surg Pathol 31(1):121–128
Lakhani SR, Khanna KK, Chenevix-Trench G (2010) Are estrogen receptor-positive breast cancers in BRCA1 mutation carriers sporadic? Breast Cancer Res 12(2):104
Swain SM (2001) Tamoxifen and contralateral breast cancer: the other side. J Natl Cancer Inst 93(13):963–965
Rutqvist LE et al (1995) Adjuvant tamoxifen therapy for early stage breast cancer and second primary malignancies. Stockholm Breast Cancer Study Group. J Natl Cancer Inst 87(9):645–51
Cook LS et al (1995) Population-based study of tamoxifen therapy and subsequent ovarian, endometrial, and breast cancers. J Natl Cancer Inst 87(18):1359–1364
Burstein HJ et al (2014) Adjuvant endocrine therapy for women with hormone receptor-positive breast cancer: American Society of Clinical Oncology clinical practice guideline focused update. J Clin Oncol 32(21):2255–2269
Reding KW et al (2010) Adjuvant systemic therapy for breast cancer in BRCA1/BRCA2 mutation carriers in a population-based study of risk of contralateral breast cancer. Breast Cancer Res Treat 123(2):491–498
Abderrahman B, Jordan VC (2016) Long-term adjuvant tamoxifen therapy and decreases in contralateral breast cancer. JAMA Oncol. https://doi.org/10.1001/jamaoncol.2016.3324
Mellemkjaer L et al (2014) Risk of contralateral breast cancer after tamoxifen use among Danish women. Ann Epidemiol 24(11):843–848
Ademuyiwa FO et al (2016) Managing breast cancer in younger women: challenges and solutions. Breast Cancer (Dove Med Press) 8:1–12
Chumsri S, Thompson EA (2020) Carryover effects of aromatase inhibitors in prevention. Lancet 395(10218):91–92
To C, Sporn MB, Liby KT (2014) PARP inhibitors for chemoprevention—reply. Cancer Prev Res (Phila) 7(11):1172
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript. The preliminary analysis that lead to this manuscript preparation was presented at ASCO in 2015, and the first author received an ASCO Merit award for the presentation.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection were performed by [MNS and AG], analysis was performed by [HL, MNS, MB, and BA]. The first draft of the manuscript was written by [MNS] and all authors commented on the previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Therese Bevers, the 4th author, has received research grants from Toray International, Preferred Medicine and Namida Lab. All other authors have no conflict of interest to disclose.
Ethical approval
This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Ethics Committee of UT MD Anderson Cancer Center.
Consent to participate
Waiver of informed consent was obtained for this retrospective chart review.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Nemati Shafaee, M., Goutsouliak, K., Lin, H. et al. Aromatase inhibitors and contralateral breast cancer in BRCA mutation carriers. Breast Cancer Res Treat 196, 143–152 (2022). https://doi.org/10.1007/s10549-022-06688-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-022-06688-z