Abstract
Purpose
We evaluated the benefit of chemotherapy in patients with ipsilateral breast tumor recurrence (IBTR) by comparing the survival outcomes between the chemotherapy and no chemotherapy groups, using propensity score matching (PSM), and analyze the survival outcomes stratified by hormone receptor status of IBTR.
Methods
We retrospectively analyzed patients who developed invasive IBTR after undergoing breast-conserving surgery at our institution between 1990 and 2013. A 1:1 PSM analysis was performed to compare the survival rates between the two study groups; additional analysis stratified by hormone receptor status was performed. The Kaplan–Meier method and Cox proportional hazards model were used to compare the second recurrence-free survival (RFS), distant metastasis-free survival (DMFS), and overall survival (OS) rates between the two groups.
Results
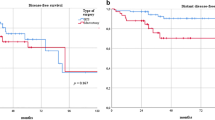
The 217 IBTR patients had a median follow-up of 125.3 months. After PSM, patients without chemotherapy and with chemotherapy (n = 35 in both groups) were included. No significant differences were observed in the 10-year second RFS [50.2% without chemotherapy vs. 39.8% with chemotherapy, hazard ratio (HR) 0.95, 95% confidence interval (CI) 0.50–1.80], DMFS (85.4% vs. 70.3%, HR 1.51, 95% CI 0.66–3.44), and OS (81.6% vs. 68.6%, HR 1.73, 95% CI 0.76–3.90) rates between the two groups. Analyses stratified by hormone receptor status showed similar findings: no significant differences were observed in the second RFS, DMFS, and OS rates between the two groups in both hormone receptor-positive and -negative groups.
Conclusion
Chemotherapy had no impact in the long-term survival outcomes of IBTR patients regardless of the hormone receptor status.


Similar content being viewed by others
References
Fisher B, Anderson S, Bryant J, Margolese RG, Deutsch M, Fisher ER, Jeong JH, Wolmark N (2002) Twenty-year follow-up of a randomized trial comparing total mastectomy, lumpectomy, and lumpectomy plus irradiation for the treatment of invasive breast cancer. N Engl J Med 347:1233–1241. https://doi.org/10.1056/NEJMoa022152
Veronesi U, Cascinelli N, Mariani L, Greco M, Saccozzi R, Luini A, Aguilar M, Marubini E (2002) Twenty-year follow-up of a randomized study comparing breast-conserving surgery with radical mastectomy for early breast cancer. N Engl J Med 347:1227–1232. https://doi.org/10.1056/NEJMoa020989
Anderson SJ, Wapnir I, Dignam JJ et al (2009) Prognosis after ipsilateral breast tumor recurrence and locoregional recurrences in patients treated by breast-conserving therapy in five national surgical adjuvant breast and bowel project protocols of node-negative breast cancer. J Clin Oncol 27:2466–2473. https://doi.org/10.1200/jco.2008.19.8424
Wapnir IL, Anderson SJ, Mamounas EP, Geyer CE Jr, Jeong JH, Tan-Chiu E, Fisher B, Wolmark N (2006) Prognosis after ipsilateral breast tumor recurrence and locoregional recurrences in five national surgical adjuvant breast and bowel project node-positive adjuvant breast cancer trials. J Clin Oncol 24:2028–2037. https://doi.org/10.1200/jco.2005.04.3273
Gentilini O, Botteri E, Rotmensz N et al (2007) When can a second conservative approach be considered for ipsilateral breast tumour recurrence? Ann Oncol 18:468–472. https://doi.org/10.1093/annonc/mdl424
Kolben T, Schwarz TM, Goess C et al (2015) Surgical management of ipsilateral breast tumor recurrence. Int J Surg 23:141–146. https://doi.org/10.1016/j.ijsu.2015.08.084
Aebi S, Gelber S, Anderson SJ et al (2014) Chemotherapy for isolated locoregional recurrence of breast cancer (CALOR): a randomised trial. Lancet Oncol 15:156–163. https://doi.org/10.1016/s1470-2045(13)70589-8
Wapnir IL, Price KN, Anderson SJ et al (2018) Efficacy of chemotherapy for ER-negative and ER-positive isolated locoregional recurrence of breast cancer: final analysis of the CALOR trial. J Clin Oncol 36:1073–1079. https://doi.org/10.1200/jco.2017.76.5719
Early Breast Cancer Trialists’ Collaborative Group (EBCTCG) (2005) Effects of chemotherapy and hormonal therapy for early breast cancer on recurrence and 15-year survival: an overview of the randomised trials. Lancet 365:1687–1717. https://doi.org/10.1016/s0140-6736(05)66544-0
Rossi L, Stevens D, Pierga J-Y, Lerebours F, Reyal F, Robain M, Asselain B, Rouzier R (2015) Impact of adjuvant chemotherapy on breast cancer survival: a real-world population. PLoS ONE 10:e0132853. https://doi.org/10.1371/journal.pone.0132853
Broeckel JA, Jacobsen PB, Balducci L, Horton J, Lyman GH (2000) Quality of life after adjuvant chemotherapy for breast cancer. Breast Cancer Res Treat 62:141–150. https://doi.org/10.1023/a:1006401914682
Paik S, Tang G, Shak S et al (2006) Gene expression and benefit of chemotherapy in women with node-negative, estrogen receptor-positive breast cancer. J Clin Oncol 24:3726–3734. https://doi.org/10.1200/jco.2005.04.7985
Cardoso F, van’t Veer LJ, Bogaerts J et al (2016) 70-gene signature as an aid to treatment decisions in early-stage breast cancer. N Engl J Med 375:717–729. https://doi.org/10.1056/NEJMoa1602253
Edge SB, Byrd DR, Carducci MA, Compton CC, Fritz AG, Greene FL, Trotti A (2010) AJCC cancer staging manual, 7th edn. Springer, New York
Darby S, McGale P, Correa C et al (2011) Effect of radiotherapy after breast-conserving surgery on 10-year recurrence and 15-year breast cancer death: meta-analysis of individual patient data for 10,801 women in 17 randomised trials. Lancet 378:1707–1716. https://doi.org/10.1016/s0140-6736(11)61629-2
Spronk I, Schellevis FG, Burgers JS, de Bock GH, Korevaar JC (2018) Incidence of isolated local breast cancer recurrence and contralateral breast cancer: a systematic review. Breast 39:70–79. https://doi.org/10.1016/j.breast.2018.03.011
Clemons M, Hamilton T, Mansi J, Lockwood G, Goss P (2003) Management of recurrent locoregional breast cancer: oncologist survey. Breast 12:328–337. https://doi.org/10.1016/s0960-9776(03)00107-3
Waeber M, Castiglione-Gertsch M, Dietrich D, Thürlimann B, Goldhirsch A, Brunner KW, Borner MM (2003) Adjuvant therapy after excision and radiation of isolated postmastectomy locoregional breast cancer recurrence: definitive results of a phase III randomized trial (SAKK 23/82) comparing tamoxifen with observation. Ann Oncol 14:1215–1221. https://doi.org/10.1093/annonc/mdg347
Acknowledgements
This study was supported by a Grant (Elimination of Cancer Project Fund) from the Asan Cancer Institute of Asan Medical Center, Seoul (2017-1341).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflict of interest.
Ethical approval
This retrospective chart review study involving human participants was in accordance with the ethical standards of the institutional and national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. The Institutional Review Board of Asan Medical Center approved this study.
Informed consent
Informed consent was waived due to the retrospective nature of the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Baek, S.Y., Kim, J., Chung, I.Y. et al. Chemotherapy for ipsilateral breast tumor recurrence: a propensity score-matching study. Breast Cancer Res Treat 192, 143–152 (2022). https://doi.org/10.1007/s10549-021-06493-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-021-06493-0