Abstract
Purpose
The optimal time interval from neoadjuvant chemotherapy (NAC) to surgery in patients with breast cancer has not been established. We investigated whether different time intervals impact the rate of pathologic complete response (pCR), disease free survival (DFS), overall survival (OS), surgical complications, and rates of conversion from mastectomy to breast conserving surgery (BCS) in this population.
Methods
We identified patients who received NAC at the BC Cancer Agency followed by surgery from May 2012 to April 2018. Patients were grouped based on time interval between NAC and surgery: < 4 weeks, 4–8 weeks, and > 8 weeks. Kaplan Meier method was used to estimate DFS and OS. Rates of pCR between the time intervals were also compared.
Results
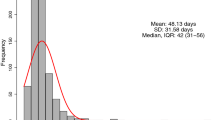
Of the 343 patients, 78 (22.8%) received surgery < 4 weeks, 233 (67.9%) received surgery between 4–8 weeks, and 32 (9.3%) received surgery > 8 weeks after NAC, with a median time to surgery (TTS) of 5.0 weeks. pCR was observed in 32.1%, 32.2%, and 28.1%, respectively (p = 0.90). Median follow-up time was 3.3 years. The 5-year DFS was 76%, 78%, and 70% (p = 0.89), respectively. The 5-year OS was 83%, 82%, and 78% (p = 0.33), respectively. No statistically significant differences were seen in surgical complications (p = 0.90), or rates of conversion from mastectomy to BCS (p = 0.19).
Conclusions
There were no statistically significant differences in pCR, DFS, OS, surgical complications, and rates of conversion from mastectomy to BCS, among breast cancer patients receiving surgery < 4 weeks, 4–8 weeks, or > 8 weeks after the last dose of NAC.



Similar content being viewed by others
References
Wolmark N, Wang J, Mamounas E, Bryant J, Fisher B (2001) Preoperative chemotherapy in patients with operable breast cancer: nine-year results from National surgical adjuvant breast and bowel project B-18. J Natl Cancer Inst Monogr 30:96–102
van der Hage JA, van de Velde CJ, Julien JP et al (2001) Peroperative chemotherapy in primary operatble breast cancer: results from the European organization for research and treatment of cancer trial 10902. J Clin Oncol 19(22):4224
Simmons CE, Hogeveen S, Leonard R et al (2015) A Canadian national expert consensus on neoadjuvant therapy for breast cancer: linking practice to evidence and beyond. Curr Oncol 22(Suppl 1):S43–53
Mauri D, Pavlidis N, Ioannidis JPA (2005) Neoadjuvant versus adjuvant systemic treatment in breast cancer: a meta-analysis. J Natl Cancer Inst 97(3):188–194
Gralow JR, Burstein HJ et al (2008) Peroperative therapy in invasive breast cancer: pathologic assessment and systemic therapy issues in operable disease. J Clin Oncol 26(5):814
Shannon C, Smith I (2003) Is there still a role for neoadjuvant therapy in breast cancer? Crit Rev Oncol Hematol 45(1):77
Cortazar P, Zhang L, Untch M et al (2014) Pathological complete response and long-term clinical benefit in breast cancer: the CTNeoBC pooled anaylsis. Lancet 384(9938):164–172
Spring LM, Fell G, Arfe A, et al. Pathological complete response (pCR) after neoadjuvant chemotherapy and impact on breast cancer recurrence and survival, stratified by breast cancer subtypes and adjuvant chemotherapy usage: patient-level meta-analyses of over 27, 000 patients. In: Proceedings from the 2018 San Antonio Breast Cancer Symposium (SABCS); Dec. 4–8; San Antonio, Texas
Sanford RA, Lei X, Vvarcenas CH, Mittendorf EA, Caudle AS, Valero V, Tripathy D, Giodrano SH, Chavez-MacGregor M (2016) Impact of time from completion of neoadjuvant chemotherapy to surgery on survival outcomes in breast cancer patients. Ann Surg Oncol 23:1515–1521
Wildiers H, Neven P, Christiaens MR, Squifflet P, Amant F, Weltens C, Smeets A, van Limbergen E, Debrock G, Renard V, Van Eeno L, Wynendaele W, Paridaens R (2010) Neoadjuvant capecitabine and docetaxel (plus trastuzumab): an effective non-anthracycline-based chemotherapy regimen for patients with locally advanced breast cancer. Ann Oncol 22(3):588–594
Sparano JA, Moulder S, Kazi A, Coppola D, Negassa A, Vahdat L, Li T, Pellegrino C, Fineberg S, Munster P, Malafa M, Lee D, Hoschander S, Hopkins U, Hershman D, Wright JJ, Kleer C, Merajver S, Sebti SM (2009) Phase II trial of the farnesyl transferase inhibitor tipifarnib plus neoadjuvant doxorubicin-cyclophosphamide in patients with clinical stage IIB-IIIC breast cancer. Clin Cancer Res 15(8):2942–2948
Honkoop AH, Luykx-de Bakker SA, Hoekman K, Meyer S, Meyer OW, van Groeningen CJ, van Diest PJ, Boven E, van der Wall E, Giaccone G, Wagstaff J, Pinedo HM (1999) Prolonged neoadjuvant chemotherapy with GM-CSF in locally advanced breast cancer. Oncologist 4(2):106–111
Heys SD, Hutcheon AW, Sarkar TK, Ogston KN, Miller ID, Payne S, Smith I, Walker LG, Eremin O, Aberdeen Breast Group (2002) Neoadjuvant docetaxel in breast cancer: 3-year survival results from the Aberdeen trial. Clin Breast Cancer 3(Suppl 2):S69–74
Balduzzi A, Montagna E, Bagnardi V, Torrisi R, Bertolini F, Mancuso P, Scarano E, Viale G, Veronesi P, Cardillo A, Orlando L, Goldhirsch A, Colleoni M (2009) Infusional fluorouracil, epirubicin, and cisplatin followed by weekly paclitaxel plus bevacizumab in locally advanced breast cancer with unfavorable prognostic features. Anticancer Drugs 20(3):197–203
Azzawi K, Ismail A, Earl H, Forouhi P, Malata CM (2010) Influence of neoadjuvant chemotherapy on outcomes of immediate breast reconstruction. Plast Reconstr Surg 126(1):1–11
Yoo TK, Moon HG, Han W, Noh DY (2017) Time interval of neoadjuvant chemotherapy to surgery in breast cancer: how long is acceptable? Gland Surg 6(1):1–3
Lohrisch C, Paltiel C, Gelmon K et al (2006) Impact on survival of time from definitive surgery to initiation of adjuvant chemotherapy for early-stage breast cancer. J Clin Oncol 24(30):4888–4894
Shin DW, Cho J, Kim SY, Guallar E, Hwang SS, Cho B et al (2013) Delay to curative surgery greater than 12 weeks is associated with increased mortality in patients with colorectal and breast cancer but not long or thyroid cancer. Ann Surg Oncol 20:2468–2476
Morante Z, Ruis R, De la Cruz-Ku G, et al. Impact of the delayed initiation of adjuvant chemotherapy in the outcomes of triple negative breast cancer: proceedings from the 2018 San Antonio Breast Cancer Symposium (SABCS); Dec. 4–8, 2018; San Antonio, Texas
Acknowledgements
We would like to acknowledge the neoadjuvant breast cancer database team at the BC Cancer Agency Vancouver Centre, and all patients undergoing neoadjuvant breast cancer treatment at the BC Cancer Agency.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None of the authors have any disclosures or conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lai, V., Hajjaj, O., Le, D. et al. Impact of wait time from neoadjuvant chemotherapy to surgery in breast cancer: Does time to surgery affect patient outcomes?. Breast Cancer Res Treat 184, 755–762 (2020). https://doi.org/10.1007/s10549-020-05894-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-020-05894-x