Abstract
Purpose
Studies have identified several estrogen receptor α (ERα) ligand-binding domain (LBD) somatic mutations in endocrine therapy resistant, metastatic ER-positive breast cancers. The most common mutations, Tyr537Ser (Y537S) and Asp538Gly (D538G), are detected in ~ 30% of endocrine resistant metastatic breast cancer patients. These ESR1 mutations induce the agonist conformation of ERα, confer an estrogen-independent phenotype, and promote drug resistance to antiestrogens.
Methods
ER-positive, estrogen-dependent MCF-7 cells were engineered to express either the Y537S or D538G mutants using CRISPR knock-in (cY537S and cD538G). These cells were used to screen several estrogen receptor degrader (ERD) compounds synthesized using the Proteolysis Targeting Chimeras (PROTAC) method to induce degradation of ERα via the ubiquitin–proteasome pathway.
Results
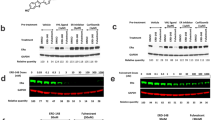
Wild-type MCF-7 and ERα LBD mutant cells were treated with ERD-148 (10 pM–1 µM) and assayed for cellular proliferation using the PrestoBlue cell viability assay. ERD-148 attenuated ER-dependent growth with IC50 values of 0.8, 10.5, and 6.1 nM in MCF-7, cY537S, and cD538G cells, respectively. Western blot analysis showed that MCF-7 cells treated with 1 nM ERD-148 for 24 h exhibited reduced ERα protein expression as compared to the mutants. The ER-regulated gene, GREB1, demonstrated significant downregulation in parental and mutant cells after 24 h of ERD-148 treatment at 10 nM. Growth of the ER-negative, estrogen-independent MDA-MB-231 breast cancer cells was not inhibited by ERD-148 at the ~ IC90 observed in the ER-positive cells.
Conclusion
ERD-148 inhibits the growth of ER-positive breast cancer cells via downregulating ERα with comparable potency to Fulvestrant with marginal non-specific toxicity.






Similar content being viewed by others
Change history
16 March 2020
In the original publication of the article, the spelling of the sixth author’s given name was incorrect. The corrected author name should read as “Wadie David”. The original article has been corrected.
References
Siegel RL, Miller KD Jemal A (2018) Cancer statistics, 2018. CA Cancer J Clin 68(1):7–30. https://doi.org/10.3322/caac.21442
Dowsett M, Cuzick J, Ingle J, Coates A, Forbes J, Bliss J, Buyse M, Baum M, Buzdar A, Colleoni M, Coombes C, Snowdon C, Gnant M, Jakesz R, Kaufmann M, Boccardo F, Godwin J, Davies C, Peto R (2010) Meta-analysis of breast cancer outcomes in adjuvant trials of aromatase inhibitors versus tamoxifen. J Clin Oncol 28(3):509–518. https://doi.org/10.1200/JCO.2009.23.1274
Cuzick J, Sestak I, Baum M, Buzdar A, Howell A, Dowsett M, Forbes JF (2010) Effect of anastrozole and tamoxifen as adjuvant treatment for early-stage breast cancer: 10-year analysis of the ATAC trial. Lancet Oncol 11(12):1135–1141. https://doi.org/10.1016/s1470-2045(10)70257-6
Pan H, Gray R, Braybrooke J, Davies C, Taylor C, McGale P, Peto R, Pritchard KI, Bergh J, Dowsett M, Hayes DF (2017) 20-year risks of breast-cancer recurrence after stopping endocrine therapy at 5 years. N Engl J Med 377(19):1836–1846. https://doi.org/10.1056/NEJMoa1701830
Jeng M, Shupnik MA, Bender TP, Westin EH, Bandyopadhyay D, Kumar R, Masamura S, Santen RJ (1998) Estrogen receptor expression and function in long-term estrogen-deprived human breast cancer cells. Endocrinology 139(10):4164–4174. https://doi.org/10.1210/endo.139.10.6229
Sikora MJ, Cordero KE, Larios JM, Johnson MD, Lippman ME, Rae JM (2009) The androgen metabolite 5alpha-androstane-3beta,17beta-diol (3betaAdiol) induces breast cancer growth via estrogen receptor: implications for aromatase inhibitor resistance. Breast Cancer Res Treat 115(2):289–296. https://doi.org/10.1007/s10549-008-0080-8
Toy W, Shen Y, Won H, Green B, Sakr RA, Will M, Li Z, Gala K, Fanning S, King TA, Hudis C, Chen D, Taran T, Hortobagyi G, Greene G, Berger M, Baselga J, Chandarlapaty S (2013) ESR1 ligand-binding domain mutations in hormone-resistant breast cancer. Nat Genet 45(12):1439–1445. https://doi.org/10.1038/ng.2822
Robinson DR, Wu YM, Vats P, Su F, Lonigro RJ, Cao X, Kalyana-Sundaram S, Wang R, Ning Y, Hodges L, Gursky A, Siddiqui J, Tomlins SA, Roychowdhury S, Pienta KJ, Kim SY, Roberts JS, Rae JM, Van Poznak CH, Hayes DF, Chugh R, Kunju LP, Talpaz M, Schott AF, Chinnaiyan AM (2013) Activating ESR1 mutations in hormone-resistant metastatic breast cancer. Nat Genet 45(12):1446–1451. https://doi.org/10.1038/ng.2823
Zhang QX, Borg A, Wolf DM, Oesterreich S, Fuqua SA (1997) An estrogen receptor mutant with strong hormone-independent activity from a metastatic breast cancer. Cancer Res 57:1244–1249
Martin LA, Ribas R, Simigdala N, Schuster E, Pancholi S, Tenev T, Gellert P, Buluwela L, Harrod A, Thornhill A, Nikitorowicz-Buniak J, Bhamra A, Turgeon MO, Poulogiannis G, Gao Q, Martins V, Hills M, Garcia-Murillas I, Fribbens C, Patani N, Li Z, Sikora MJ, Turner N, Zwart W, Oesterreich S, Carroll J, Ali S, Dowsett M (2017) Discovery of naturally occurring ESR1 mutations in breast cancer cell lines modelling endocrine resistance. Nat Commun 8(1):1865. https://doi.org/10.1038/s41467-017-01864-y
Wang P, Bahreini A, Gyanchandani R, Lucas PC, Hartmaier RJ, Watters RJ, Jonnalagadda AR, Trejo Bittar HE, Berg A, Hamilton RL, Kurland BF, Weiss KR, Mathew A, Leone JP, Davidson NE, Nikiforova MN, Brufsky AM, Ambros TF, Stern AM, Puhalla SL, Lee AV, Oesterreich S (2016) Sensitive detection of mono- and polyclonal ESR1 mutations in primary tumors, metastatic lesions, and cell-free DNA of breast cancer patients. Clin Cancer Res. https://doi.org/10.1158/1078-0432.ccr-15-1534
Harrod A, Fulton J, Nguyen VTM, Periyasamy M, Ramos-Garcia L, Lai CF, Metodieva G, de Giorgio A, Williams RL, Santos DB, Gomez PJ, Lin ML, Metodiev MV, Stebbing J, Castellano L, Magnani L, Coombes RC, Buluwela L, Ali S (2017) Genomic modelling of the ESR1 Y537S mutation for evaluating function and new therapeutic approaches for metastatic breast cancer. Oncogene 36(16):2286–2296. https://doi.org/10.1038/onc.2016.382
Li S, Shen D, Shao J, Crowder R, Liu W, Prat A, He X, Liu S, Hoog J, Lu C, Ding L, Griffith OL, Miller C, Larson D, Fulton RS, Harrison M, Mooney T, McMichael JF, Luo J, Tao Y, Goncalves R, Schlosberg C, Hiken JF, Saied L, Sanchez C, Giuntoli T, Bumb C, Cooper C, Kitchens RT, Lin A, Phommaly C, Davies SR, Zhang J, Kavuri MS, McEachern D, Dong YY, Ma C, Pluard T, Naughton M, Bose R, Suresh R, McDowell R, Michel L, Aft R, Gillanders W, DeSchryver K, Wilson RK, Wang S, Mills GB, Gonzalez-Angulo A, Edwards JR, Maher C, Perou CM, Mardis ER, Ellis MJ (2013) Endocrine-therapy-resistant ESR1 variants revealed by genomic characterization of breast-cancer-derived xenografts. Cell Rep 4(6):1116–1130. https://doi.org/10.1016/j.celrep.2013.08.022
Toy W, Weir H, Razavi P, Lawson M, Goeppert AU, Mazzola AM, Smith A, Wilson J, Morrow C, Wong WL, De Stanchina E, Carlson KE, Martin TS, Uddin S, Li Z, Fanning S, Katzenellenbogen JA, Greene G, Baselga J, Chandarlapaty S (2017) Activating ESR1 mutations differentially affect the efficacy of ER antagonists. Cancer Discov 7(3):277–287. https://doi.org/10.1158/2159-8290.CD-15-1523
Weis KE, Ekena K, Thomas JA, Lazennec G, Katzenellenbogen BS (1996) Constitutively active human estrogen receptors containing amino acid substitutions for tyrosine 537 in the receptor protein. Mol Endocrinol 10:1388–1398
Long X, Nephew KP (2006) Fulvestrant (ICI 182,780)-dependent interacting proteins mediate immobilization and degradation of estrogen receptor-alpha. J Biol Chem 281(14):9607–9615. https://doi.org/10.1074/jbc.M510809200
Ingle JN, Suman VJ, Rowland KM, Mirchandani D, Bernath AM, Camoriano JK, Fishkin PA, Nikcevich DA, Perez EA, North Central Cancer Treatment Group Trial, N (2006) Fulvestrant in women with advanced breast cancer after progression on prior aromatase inhibitor therapy: North Central Cancer Treatment Group Trial N0032. J Clin Oncol 24(7):1052–1056. https://doi.org/10.1200/JCO.2005.04.1053
Zhou B, Hu J, Xu F, Chen Z, Bai L, Fernandez-Salas E, Lin M, Liu L, Yang CY, Zhao Y, McEachern D, Przybranowski S, Wen B, Sun D, Wang S (2017) Discovery of a small-molecule degrader of bromodomain and extra-terminal (BET) proteins with picomolar cellular potencies and capable of achieving tumor regression. J Med Chem. https://doi.org/10.1021/acs.jmedchem.6b01816
Bai L, Zhou B, Yang CY, Ji J, McEachern D, Przybranowski S, Jiang H, Hu J, Xu F, Zhao Y, Liu L, Fernandez-Salas E, Xu J, Dou Y, Wen B, Sun D, Meagher J, Stuckey J, Hayes DF, Li S, Ellis MJ, Wang S (2017) Targeted degradation of BET proteins in triple-negative breast cancer. Cancer Res 77(9):2476–2487. https://doi.org/10.1158/0008-5472.CAN-16-2622
Winter GE, Buckley DL, Paulk J, Roberts JM, Souza A, Dhe-Paganon S, Bradner JE (2015) Phthalimide conjugation as a strategy for in vivo target protein degradation. Science 348(6241):1376–1381. https://doi.org/10.1126/science.aab1433
Sakamoto KM, Kim KB, Kumagai A, Mercurio F, Crews CM, Deshaies RJ (2001) Protacs: chimeric molecules that target proteins to the Skp1-Cullin-F box complex for ubiquitination and degradation. Proc Natl Acad Sci USA 98(15):8554–8559. https://doi.org/10.1073/pnas.141230798
Gadd MS, Testa A, Lucas X, Chan KH, Chen W, Lamont DJ, Zengerle M, Ciulli A (2017) Structural basis of PROTAC cooperative recognition for selective protein degradation. Nat Chem Biol 13(5):514–521. https://doi.org/10.1038/nchembio.2329
Hu J, Hu B, Wang M, Xu F, Miao B, Yang C-Y, Wang M, Liu Z, Hayes DF, Chinnaswamy K, Delproposto J, Stuckey J, Wang S (2019) Discovery of ERD-308 as a highly potent proteolysis targeting chimera (PROTAC) degrader of estrogen receptor (ER). J Med Chem 62(3):1420–1442. https://doi.org/10.1021/acs.jmedchem.8b01572
Rae JM, Johnson MD, Scheys JO, Cordero KE, Larios JM, Lippman ME (2005) GREB 1 is a critical regulator of hormone dependent breast cancer growth. Breast Cancer Res Treat 92(2):141–149. https://doi.org/10.1007/s10549-005-1483-4
Ran FA, Hsu PD, Wright J, Agarwala V, Scott DA, Zhang F (2013) Genome engineering using the CRISPR-Cas9 system. Nat Protoc 8(11):2281–2308. https://doi.org/10.1038/nprot.2013.143
Johnson MD, Zuo H, Lee KH, Trebley JP, Rae JM, Weatherman RV, Desta Z, Flockhart DA, Skaar TC (2004) Pharmacological characterization of 4-hydroxy-N-desmethyl tamoxifen, a novel active metabolite of tamoxifen. Breast Cancer Res Treat 85(2):151–159. https://doi.org/10.1023/B:BREA.0000025406.31193.e8
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 25(4):402–408. https://doi.org/10.1006/meth.2001.1262
Mohammed H, D'Santos C, Serandour AA, Ali HR, Brown GD, Atkins A, Rueda OM, Holmes KA, Theodorou V, Robinson JL, Zwart W, Saadi A, Ross-Innes CS, Chin SF, Menon S, Stingl J, Palmieri C, Caldas C, Carroll JS (2013) Endogenous purification reveals GREB1 as a key estrogen receptor regulatory factor. Cell Rep 3(2):342–349. https://doi.org/10.1016/j.celrep.2013.01.010
Sikora MJ, Strumba V, Lippman ME, Johnson MD, Rae JM (2012) Mechanisms of estrogen-independent breast cancer growth driven by low estrogen concentrations are unique versus complete estrogen deprivation. Breast Cancer Res Treat 134(3):1027–1039. https://doi.org/10.1007/s10549-012-2032-6
Johnston SR, Kilburn LS, Ellis P, Dodwell D, Cameron D, Hayward L, Im YH, Braybrooke JP, Brunt AM, Cheung KL, Jyothirmayi R, Robinson A, Wardley AM, Wheatley D, Howell A, Coombes G, Sergenson N, Sin HJ, Folkerd E, Dowsett M, Bliss JM, Feai S (2013) Fulvestrant plus anastrozole or placebo versus exemestane alone after progression on non-steroidal aromatase inhibitors in postmenopausal patients with hormone-receptor-positive locally advanced or metastatic breast cancer (SoFEA): a composite, multicentre, phase 3 randomised trial. Lancet Oncol 14(10):989–998. https://doi.org/10.1016/S1470-2045(13)70322-X
Mehta RS, Barlow WE, Albain KS, Vandenberg TA, Dakhil SR, Tirumali NR, Lew DL, Hayes DF, Gralow JR, Livingston RB, Hortobagyi GN (2012) Combination anastrozole and fulvestrant in metastatic breast cancer. N Engl J Med 367(5):435–444. https://doi.org/10.1056/NEJMoa1201622
Fribbens C, O'Leary B, Kilburn L, Hrebien S, Garcia-Murillas I, Beaney M, Cristofanilli M, Andre F, Loi S, Loibl S, Jiang J, Bartlett CH, Koehler M, Dowsett M, Bliss JM, Johnston SR, Turner NC (2016) Plasma ESR1 mutations and the treatment of estrogen receptor-positive advanced breast cancer. J Clin Oncol 34(25):2961–2968. https://doi.org/10.1200/JCO.2016.67.3061
Paoletti C, Cani AK, Larios JM, Hovelson DH, Aung K, Darga EP, Cannell EM, Baratta PJ, Liu CJ, Chu D, Yazdani M, Blevins AR, Sero V, Tokudome N, Thomas DG, Gersch C, Schott AF, Wu YM, Lonigro R, Robinson DR, Chinnaiyan AM, Bischoff FZ, Johnson MD, Park BH, Hayes DF, Rae JM, Tomlins SA (2018) Comprehensive mutation and copy number profiling in archived circulating breast cancer tumor cells documents heterogeneous resistance mechanisms. Cancer Res 78(4):1110–1122. https://doi.org/10.1158/0008-5472.CAN-17-2686
Schiavon G, Hrebien S, Garcia-Murillas I, Cutts RJ, Pearson A, Tarazona N, Fenwick K, Kozarewa I, Lopez-Knowles E, Ribas R, Nerurkar A, Osin P, Chandarlapaty S, Martin LA, Dowsett M, Smith IE, Turner NC (2015) Analysis of ESR1 mutation in circulating tumor DNA demonstrates evolution during therapy for metastatic breast cancer. Sci Transl Med 7(313):313–317. https://doi.org/10.1126/scitranslmed.aac7551
Jeselsohn R, Yelensky R, Buchwalter G, Frampton G, Meric-Bernstam F, Gonzalez-Angulo AM, Ferrer-Lozano J, Perez-Fidalgo JA, Cristofanilli M, Gomez H, Arteaga CL, Giltnane J, Balko JM, Cronin MT, Jarosz M, Sun J, Hawryluk M, Lipson D, Otto G, Ross JS, Dvir A, Soussan-Gutman L, Wolf I, Rubinek T, Gilmore L, Schnitt S, Come SE, Pusztai L, Stephens P, Brown M, Miller VA (2014) Emergence of constitutively active estrogen receptor-alpha mutations in pretreated advanced estrogen receptor-positive breast cancer. Clin Cancer Res 20(7):313ra182. https://doi.org/10.1158/1078-0432.CCR-13-2332
Weir HM, Bradbury RH, Lawson M, Rabow AA, Buttar D, Callis RJ, Curwen JO, de Almeida C, Ballard P, Hulse M, Donald CS, Feron LJ, Karoutchi G, MacFaul P, Moss T, Norman RA, Pearson SE, Tonge M, Davies G, Walker GE, Wilson Z, Rowlinson R, Powell S, Sadler C, Richmond G, Ladd B, Pazolli E, Mazzola AM, D'Cruz C, De Savi C (2016) AZD9496: An oral estrogen receptor inhibitor that blocks the growth of ER-positive and ESR1-mutant breast tumors in preclinical models. Cancer Res 76(11):3307–3318. https://doi.org/10.1158/0008-5472.CAN-15-2357
Wang Y, Lonard DM, Yu Y, Chow DC, Palzkill TG, Wang J, Qi R, Matzuk AJ, Song X, Madoux F, Hodder P, Chase P, Griffin PR, Zhou S, Liao L, Xu J, O'Malley BW (2014) Bufalin is a potent small-molecule inhibitor of the steroid receptor coactivators SRC-3 and SRC-1. Cancer Res 74(5):1506–1517. https://doi.org/10.1158/0008-5472.CAN-13-2939
Reinert T, Saad ED, Barrios CH, Bines J (2017) Clinical implications of ESR1 mutations in hormone receptor-positive advanced breast cancer. Front Oncol 7:26. https://doi.org/10.3389/fonc.2017.00026
Wardell SE, Ellis MJ, Alley HM, Eisele K, VanArsdale T, Dann SG, Arndt KT, Primeau T, Griffin E, Shao J, Crowder R, Lai JP, Norris JD, McDonnell DP, Li S (2015) Efficacy of SERD/SERM hybrid-CDK4/6 inhibitor combinations in models of endocrine therapy-resistant breast cancer. Clin Cancer Res 21(22):5121–5130. https://doi.org/10.1158/1078-0432.CCR-15-0360
Acknowledgements
The authors would like to thank the University of Michigan Biomedical Research Sequencing Core for their support. We acknowledge the assistance provided by the GUMC Tissue Culture Shared Resource, which is supported in part by the Lombardi Comprehensive Cancer Center support grant (NIH/NCI Grant P30-CA051008).
Funding
Research reported in this publication was supported in part by the National Institute of Environmental Health Sciences of the National Institutes of Health under Award Number T32ES007062 (TLG), the Breast Cancer Research Foundation (BCRF) (N003173 to JMR), and the University of Michigan Rogel Cancer Center Breast Strategic Fund (JMR, SW, and DFH). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The University of Michigan has filed a patent application on ERD-148 and its related ER degraders. S.W. and J.H. are co-inventors on the patent application, which has been licensed to Oncopia Therapeutics, Inc. S.W. is a co-founder of Oncopia and a paid consultant. S.W. and the University of Michigan own stock in Oncopia. The University of Michigan has received a research contract from Oncopia for which S.W. serves as the principal investigator.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original version of this article was revised: The spelling of the sixth author’s given name was incorrectly published. The author name was corrected.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gonzalez, T.L., Hancock, M., Sun, S. et al. Targeted degradation of activating estrogen receptor α ligand-binding domain mutations in human breast cancer. Breast Cancer Res Treat 180, 611–622 (2020). https://doi.org/10.1007/s10549-020-05564-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-020-05564-y