Abstract
Background
Dysregulated cellular metabolism is one of the hallmarks of cancer with some tumours utilising the glutamine metabolism pathway for their sustained proliferation and survival. Glutamate dehydrogenase (GLUD1) is a key enzyme in glutaminolysis converting glutamate to α-ketoglutarate for entry into the TCA cycle. Breast cancer (BC) comprises a heterogeneous group of tumours in terms of molecular biology and clinical behaviour, and we have previously shown that altered glutamine metabolism varies substantially among the different molecular subtypes. We hypothesise that the prognostic value of GLUD1 expression will differ between the BC molecular subtypes and may act as a potential therapeutic target for BC tumours.
Methods
GLUD1 was assessed at the DNA, mRNA (n = 1980) and protein (n = 1300) levels in large, well-characterised cohorts and correlated with clinicopathological parameters, molecular subtypes, patient outcome, and treatments.
Results
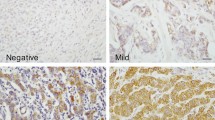
There was a correlation between GLUD1 mRNA and GLUD1 protein expression which were highly expressed in low grade luminal/ER + BC (p < 0.01). GLUD1 mRNA and protein was associated with good patient outcome but not in any specific molecular subtypes. However, high GLUD1 protein expression was associated with a better outcome in triple negative (TN) patients treated with chemotherapy (p = 0.03). High GLUD1 mRNA was associated with the glutamine transporter, SLC1A5, and leucine transporter, SLC7A8 as well as mTOR (p < 0.0001).
Conclusion
We provide comprehensive data indicating GLUD1 plays an important role in luminal/ER + BC. GLUD1 expression predicts a better patient outcome and we show that it has the potential for predicting response to chemotherapy in TNBC patients.





Similar content being viewed by others
References
Hanahan D, Weinberg RA (2011) Hallmarks of cancer: the next generation. Cell 144(5):646–674
Warburg O (1956) On the origin of cancer cells. Science 123(3191):309–314
Alberghina L, Gaglio D (2014) Redox control of glutamine utilization in cancer. Cell Death Dis 2014. 5:e1561
Yang L et al (2014) Metabolic shifts toward glutamine regulate tumor growth, invasion and bioenergetics in ovarian cancer. Mol Syst Biol 10:728
Green AR et al (2016) MYC functions are specific in biological subtypes of breast cancer and confers resistance to endocrine therapy in luminal tumours. Br J Cancer 114(8):917–928
Hu W et al (2010) Glutaminase 2, a novel p53 target gene regulating energy metabolism and antioxidant function. Proc Natl Acad Sci USA 107(16):7455–7460
Polyak K et al (1997) A model for p53-induced apoptosis. Nature 389(6648):300–305
Cao MD et al (2014) Metabolic characterization of triple negative breast cancer. BMC Cancer 14:941
Gross MI et al (2014) Antitumor activity of the glutaminase inhibitor CB-839 in triple-negative breast cancer. Mol Cancer Ther 13(4):890–901
Kim S et al (2013) Expression of glutamine metabolism-related proteins according to molecular subtype of breast cancer. Endocr Relat Cancer 20(3):339–348
Kanaan YM et al (2014) Metabolic profile of triple-negative breast cancer in African-American women reveals potential biomarkers of aggressive disease. Cancer Genom Proteom 11(6):279–294
Craze ML et al (2018) MYC regulation of glutamine-proline regulatory axis is key in luminal B breast cancer. Br J Cancer 118(2):258–265
Plaitakis A et al (2017) The glutamate dehydrogenase pathway and its roles in cell and tissue biology in health and disease. Biology (Basel) 6(1):11
Duran RV et al (2012) Glutaminolysis activates Rag-mTORC1 signaling. Mol Cell 47(3):349–358
Jin L et al (2015) Glutamate dehydrogenase 1 signals through antioxidant glutathione peroxidase 1 to regulate redox homeostasis and tumor growth. Cancer Cell 27(2):257–270
Curtis C et al (2012) The genomic and transcriptomic architecture of 2000 breast tumours reveals novel subgroups. Nature 486(7403):346–352
Silwal-Pandit L et al (2014) TP53 mutation spectrum in breast cancer is subtype specific and has distinct prognostic relevance. Clin Cancer Res 20(13):3569–3580
Camp RL, Dolled-Filhart M, Rimm DL (2004) X-tile: a new bio-informatics tool for biomarker assessment and outcome-based cut-point optimization. Clin Cancer Res 10(21):7252–7259
Jezequel P et al (2012) bc-GenExMiner: an easy-to-use online platform for gene prognostic analyses in breast cancer. Breast Cancer Res Treat 131(3):765–775
Abd El-Rehim DM et al (2005) High-throughput protein expression analysis using tissue microarray technology of a large well-characterised series identifies biologically distinct classes of breast cancer confirming recent cDNA expression analyses. Int J Cancer 116(3):340–350
McCarty KS Jr, McCarty KSS (1984) Histochemical approaches to steroid receptor analyses. Semin Diagn Pathol 1(4):297–308
McShane LM et al (2005) REporting recommendations for tumour MARKer prognostic studies (REMARK). Br J Cancer 93(4):387–391
Jerby L et al (2012) Metabolic associations of reduced proliferation and oxidative stress in advanced breast cancer. Cancer Res 72(22):5712–5720
Tyanova S et al (2016) Proteomic maps of breast cancer subtypes. Nat Commun 7:10259
El Ansari R et al (2018) Altered glutamine metabolism in breast cancer; subtype dependencies and alternative adaptations. Histopathology 72(2):183–190
Sancak Y et al (2010) Ragulator-Rag complex targets mTORC1 to the lysosomal surface and is necessary for its activation by amino acids. Cell 141(2):290–303
Sancak Y et al (2008) The Rag GTPases bind raptor and mediate amino acid signaling to mTORC1. Science 320(5882):1496–1501
Nicklin P et al (2009) Bidirectional transport of amino acids regulates mTOR and autophagy. Cell 136(3):521–534
Kim SG, Buel GR, Blenis J (2013) Nutrient regulation of the mTOR complex 1 signaling pathway. Mol Cells 35(6):463–473
Acknowledgements
We thank the Nottingham Health Science Biobank and Breast Cancer Now Tissue Bank for the provision of tissue samples. We thank the University of Nottingham (Nottingham Life Cycle 6 and Cancer Research Priority Area) for funding.
Author information
Authors and Affiliations
Contributions
MLC and ARG conceived and designed study. MLC, RE, MAA, KWC, LA, BM, MDR, CCN, IOE, EAR, ARG carried out experiments and collected data. MLC, KWC, ARG analysed data. All authors were involved in writing the paper and had final approval of the submitted and published versions.
Corresponding author
Ethics declarations
Conflict of interest
The authors confirm that they do not have any conflict of interests to declare.
Ethical approval
This study was approved by the Nottingham Research Ethics Committee 2 under the title ‘Development of a molecular genetic classification of breast cancer’. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. All tissue samples from Nottingham used in this study were pseudo-anonymised and collected prior to 1st September 2006; therefore under the Human Tissue Act informed patient consent was not needed. Release of data was also pseudo-anonymised as per Human Tissue Act regulations. This article does not contain any studies with animals performed by any of the authors.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Craze, M.L., El-Ansari, R., Aleskandarany, M.A. et al. Glutamate dehydrogenase (GLUD1) expression in breast cancer. Breast Cancer Res Treat 174, 79–91 (2019). https://doi.org/10.1007/s10549-018-5060-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-018-5060-z