Abstract
Purpose
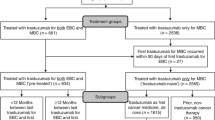
Patients treated with trastuzumab for HER2-positive metastatic breast cancer (HER2+MBC) are living longer, but there is little information on their outcomes and treatment experience beyond the median survival from clinical trials and real-world observational studies. We aim to describe the real-world treatment patterns and overall survival (OS) for women surviving five or more years from initiation of trastuzumab for HER2+MBC.
Methods
This is a retrospective, whole-of-population cohort study of women initiating trastuzumab for HER2+MBC between 2001 and 2011, followed to 2016. We defined long-term survivors (LTS) as those patients surviving ≥ 5 years from trastuzumab initiation. We used dispensing claims to describe timing of cancer treatments used by LTS and to estimate time on and off HER2-targeted therapies, and OS from trastuzumab initiation for HER2+MBC.
Results
Of 4177 women initiating trastuzumab for HER2+MBC, 1082 (26%) survived ≥ 5 years. Median age for LTS was 54 years (IQR 46–63). At a median follow-up of 9.4 years, 36% of LTS died; their conditional probability of surviving an additional 5 years was 55%. Median time on trastuzumab and all HER2-targeted therapy was 58.9 months (27.6–88.1) and 69.1 months (35.6–124.5), respectively. 85% of LTS had a period off HER2 therapy, lasting a median of 30.4 months (8.2–NR).
Conclusions
LTS generally receive HER2-targeted therapies for periods of time longer than in clinical trials, but most LTS also had breaks in treatment. More research is needed to understand the effects of long-term treatment and to identify patients who may be able to safely discontinue HER2-targeted therapy.


Similar content being viewed by others
References
Dawood S, Broglio K, Buzdar AU, Hortobagyi GN, Giordano SH (2010) Prognosis of women with metastatic breast cancer by HER2 status and trastuzumab treatment: an institutional-based review. J Clin Oncol 28(1):92–98. https://doi.org/10.1200/JCO.2008.19.9844
Slamon DJ, Leyland-Jones B, Shak S, Fuchs H, Paton V, Bajamonde A, Fleming T, Eiermann W, Wolter J, Pegram M, Baselga J, Norton L (2001) Use of chemotherapy plus a monoclonal antibody against HER2 for metastatic breast cancer that overexpresses HER2. N Engl J Med 344(11):783–792. https://doi.org/10.1056/NEJM200103153441101
Swain SM, Baselga J, Kim SB, Ro J, Semiglazov V, Campone M, Ciruelos E, Ferrero JM, Schneeweiss A, Heeson S, Clark E, Ross G, Benyunes MC, Cortes J, Group CS (2015) Pertuzumab, trastuzumab, and docetaxel in HER2-positive metastatic breast cancer. N Engl J Med 372(8):724–734. https://doi.org/10.1056/NEJMoa1413513
Daniels B, Kiely BE, Lord SJ, Houssami N, Lu CY, Ward RL, Pearson S-A (2017) Trastuzumab for metastatic breast cancer: Real world outcomes from an Australian whole-of-population cohort (2001–2016). Breast 38:7–13. https://doi.org/10.1016/j.breast.2017.11.007
Harano K, Lei X, Gonzalez-Angulo AM, Murthy RK, Valero V, Mittendorf EA, Ueno NT, Hortobagyi GN, Chavez-MacGregor M (2016) Clinicopathological and surgical factors associated with long-term survival in patients with HER2-positive metastatic breast cancer. Breast Cancer Res Treat 159(2):367–374. https://doi.org/10.1007/s10549-016-3933-6
Murthy P, Kidwell KM, Schott AF, Merajver SD, Griggs JJ, Smerage JD, Van Poznak CH, Wicha MS, Hayes DF, Henry NL (2016) Clinical predictors of long-term survival in HER2-positive metastatic breast cancer. Breast Cancer Res Treat 155(3):589–595. https://doi.org/10.1007/s10549-016-3705-3
Spano JP, Beuzeboc P, Coeffic D, Arnould L, Lortholary A, Andre F, Ferrero JM (2015) Long term HER2 + metastatic breast cancer survivors treated by trastuzumab: results from the French cohort study LHORA. Breast 24(4):376–383. https://doi.org/10.1016/j.breast.2015.02.035
Vaz-Luis I, Seah D, Olson EM, Wagle N, Metzger-Filho O, Sohl J, Litsas G, Burstein HJ, Krop IE, Winer EP, Lin NU (2013) Clinicopathological features among patients with advanced human epidermal growth factor-2-positive breast cancer with prolonged clinical benefit to first-line trastuzumab-based therapy: a retrospective cohort study. Clin Breast Cancer 13(4):254–263. https://doi.org/10.1016/j.clbc.2013.02.010
Witzel I, Muller V, Abenhardt W, Kaufmann M, Schoenegg W, Schneeweis A, Janicke F (2014) Long-term tumor remission under trastuzumab treatment for HER2 positive metastatic breast cancer—results from the HER-OS patient registry. BMC Cancer 14:806. https://doi.org/10.1186/1471-2407-14-806
Yardley DA, Tripathy D, Brufsky AM, Rugo HS, Kaufman PA, Mayer M, Magidson J, Yoo B, Quah C, Ulcickas Yood M (2014) Long-term survivor characteristics in HER2-positive metastatic breast cancer from registHER. Br J Cancer 110(11):2756–2764. https://doi.org/10.1038/bjc.2014.174
Yeo B, Kotsori K, Mohammed K, Walsh G, Smith IE (2015) Long-term outcome of HER2 positive metastatic breast cancer patients treated with first-line trastuzumab. Breast 24(6):751–757. https://doi.org/10.1016/j.breast.2015.09.008
Steenbruggen T, van Ramshorst M, Stouthard J, Rodenhuis S, Linn S, Sonke G, Smorenburg C (2017) Abstract P4-21-30: long-term survival in HER2-positive metastatic breast cancer: the first blow is half the battle. Can Res 77 (4 Suppl):P4-21-30. https://doi.org/10.1158/1538-7445.sabcs16-p4-21-30
Wong Y, Raghavendra AS, Hatzis C, Irizarry JP, Vega T, Barcenas CH, Gregor MC-M, Valero V, Tripathy D, Pusztai L, Murthy RK (2017) Long-term survival of de novo stage IV human epidermal growth factor receptor 2 (HER2)-positive breast cancers treated with HER2 targeted therapy. J Clin Oncol 35(15_suppl):1021–1021. https://doi.org/10.1200/JCO.2017.35.15_suppl.1021
Gamez-Pozo A, Perez Carrion RM, Manso L, Crespo C, Mendiola C, Lopez-Vacas R, Berges-Soria J, Lopez IA, Margeli M, Calero JL, Farre XG, Santaballa A, Ciruelos EM, Afonso R, Lao J, Catalan G, Gallego JV, Lopez JM, Bofill FJ, Borrego MR, Espinosa E, Vara JA, Zamora P (2014) The Long-HER study: clinical and molecular analysis of patients with HER2+ advanced breast cancer who become long-term survivors with trastuzumab-based therapy. PLoS ONE 9(10):e109611. https://doi.org/10.1371/journal.pone.0109611
Cardoso F, Costa A, Senkus E, Aapro M, Andre F, Barrios CH, Bergh J, Bhattacharyya G, Biganzoli L, Cardoso MJ, Carey L, Corneliussen-James D, Curigliano G, Dieras V, El Saghir N, Eniu A, Fallowfield L, Fenech D, Francis P, Gelmon K, Gennari A, Harbeck N, Hudis C, Kaufman B, Krop I, Mayer M, Meijer H, Mertz S, Ohno S, Pagani O, Papadopoulos E, Peccatori F, Penault-Llorca F, Piccart MJ, Pierga JY, Rugo H, Shockney L, Sledge G, Swain S, Thomssen C, Tutt A, Vorobiof D, Xu B, Norton L, Winer E (2017) 3rd ESO-ESMO international consensus guidelines for advanced breast cancer (ABC 3). Ann Oncol. https://doi.org/10.1093/annonc/mdx036
Daniels B, Lord SJ, Kiely BE, Houssami N, Haywood P, Lu CY, Ward RL, Pearson S-A (2017) Use and outcomes of targeted therapies in early and metastatic HER2-positive breast cancer in Australia: protocol detailing observations in a whole of population cohort. BMJ Open 7(1). https://doi.org/10.1136/bmjopen-2016-014439
Hieke S, Kleber M, Konig C, Engelhardt M, Schumacher M (2015) Conditional survival: a useful concept to provide information on how prognosis evolves over time. Clin Cancer Res 21(7):1530–1536. https://doi.org/10.1158/1078-0432.CCR-14-2154
Pearson SA, Ringland CL, Ward RL (2007) Trastuzumab and metastatic breast cancer: trastuzumab use in Australia–monitoring the effect of an expensive medicine access program. J Clin Oncol 25(24):3688–3693. https://doi.org/10.1200/jco.2007.11.2516
Vitry A, Mintzes B, Lipworth W (2016) Access to new cancer medicines in Australia: dispelling the myths and informing a public debate. J Pharm Policy Pract 9(1):13. https://doi.org/10.1186/s40545-016-0062-x
Karikios DJ, Schofield D, Salkeld G, Mann KP, Trotman J, Stockler MR (2014) Rising cost of anticancer drugs in Australia. Intern Med J 44(5):458–463. https://doi.org/10.1111/imj.12399
Vu T, Claret FX (2012) Trastuzumab: updated mechanisms of action and resistance in breast cancer. Front Oncol 2:62. https://doi.org/10.3389/fonc.2012.00062
Daniels B, Kiely BE, Houssami N, Lord SJ, Dobbins T, Lu CY, Ward RL, Pearson SA (2017) Survival outcomes for Australian women receiving trastuzumab for HER2-positive metastatic breast cancer following (neo)adjuvant trastuzumab: a national population-based observational study (2006–2014). Br J Cancer. https://doi.org/10.1038/bjc.2017.405
Acknowledgements
We acknowledge the contribution of Sally Crossing (AM) (1946–2016) as the Health Consumer Advocate on this research program. We thank the Department of Human Services for providing the data for this research.
Funding
This work was supported by a Cancer Australia Priority Driven Collaborative Support Scheme (ID: 1050648) and the NHMRC Centre of Research Excellence in Medicines and Ageing (CREMA; ID: 1060407). BD is supported by an NHMRC Postgraduate Research Scholarship (ID: 1094325), the Sydney Catalyst Translational Cancer Research Centre (no grant number), and a CREMA PhD scholarship top-up (no grant number). NH receives funding through a National Breast Cancer Foundation (Australia) Breast Cancer Research Leadership Fellowship (no grant number).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
BEK has received conference support and a speaker’s honorarium from Roche. RLW is a member of the Pharmaceutical Benefits Advisory Committee (PBAC) and SAP is a member of the Drug Utilisation Sub Committee of the PBAC. The views expressed in this paper do not represent those of the either committee. The remaining authors declare no conflicts of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Daniels, B., Kiely, B.E., Lord, S.J. et al. Long-term survival in trastuzumab-treated patients with HER2-positive metastatic breast cancer: real-world outcomes and treatment patterns in a whole-of-population Australian cohort (2001–2016). Breast Cancer Res Treat 171, 151–159 (2018). https://doi.org/10.1007/s10549-018-4804-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-018-4804-0